


Introduction
The Zeeman effect refers to the splitting of atomic spectral lines when atoms are placed in an external magnetic field. When atoms emit or absorb light, they produce characteristic spectral lines corresponding to transitions between energy levels. However, when a magnetic field is applied, these spectral lines split into multiple components.
The Zeeman effect provides direct evidence that atomic energy levels are influenced by magnetic fields and that electrons possess magnetic properties related to their angular momentum and spin.
The phenomenon was discovered in 1896 by Pieter Zeeman while studying the spectral lines of sodium in the presence of a magnetic field. The theoretical explanation was later developed by Hendrik Lorentz, who used classical electromagnetic theory to interpret the observations.
The Zeeman effect played a major role in the development of atomic physics, quantum mechanics, and spectroscopy. It also confirmed that electrons behave like tiny magnets due to their angular momentum and spin.
Today, the Zeeman effect is widely used in fields such as astrophysics, plasma physics, magnetic field measurement, and atomic spectroscopy.
Basic Concept of the Zeeman Effect
Atoms contain electrons that possess angular momentum. Associated with this angular momentum is a magnetic moment, which means that electrons behave like small magnets.
When an atom is placed in a magnetic field, the magnetic moment interacts with the external magnetic field. This interaction modifies the energy levels of the atom.
Because energy levels change slightly, transitions between these levels produce spectral lines at slightly different wavelengths.
As a result, a single spectral line splits into multiple components.
Magnetic Moment of an Electron
The magnetic moment associated with the orbital motion of an electron is given by
[
\mu = \frac{e}{2m_e}L
]
where
- (e) = electron charge
- (m_e) = electron mass
- (L) = orbital angular momentum
This magnetic moment interacts with the external magnetic field.
Energy Interaction with Magnetic Field
When a magnetic moment is placed in a magnetic field (B), the energy of interaction is
[
E = -\vec{\mu} \cdot \vec{B}
]
Because the orientation of the magnetic moment can have several discrete values, the energy levels split into multiple levels.
This splitting leads to multiple spectral lines.
Types of Zeeman Effect
The Zeeman effect can be classified into two main types:
- Normal Zeeman Effect
- Anomalous Zeeman Effect
Normal Zeeman Effect




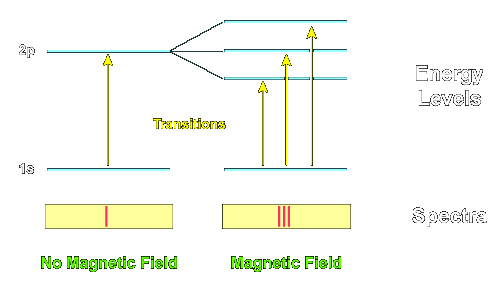
The normal Zeeman effect occurs when spectral lines split into three components in the presence of a magnetic field.
These three lines are called a triplet.
The three components include:
- One central line with unchanged frequency
- Two side lines with shifted frequencies
Explanation of Normal Zeeman Effect
In the normal Zeeman effect, the splitting occurs due to orbital angular momentum only, without considering electron spin.
The magnetic quantum number (m_l) determines the possible orientations of angular momentum.
Possible values are:
[
m_l = -l, -(l-1), …, 0, …, +(l-1), +l
]
In the presence of a magnetic field, each value of (m_l) corresponds to a slightly different energy.
Frequency Shift
The frequency change due to the magnetic field is given by
[
\Delta \nu = \frac{eB}{4\pi m_e}
]
where
- (B) = magnetic field strength
This results in symmetric splitting around the original spectral line.
Anomalous Zeeman Effect



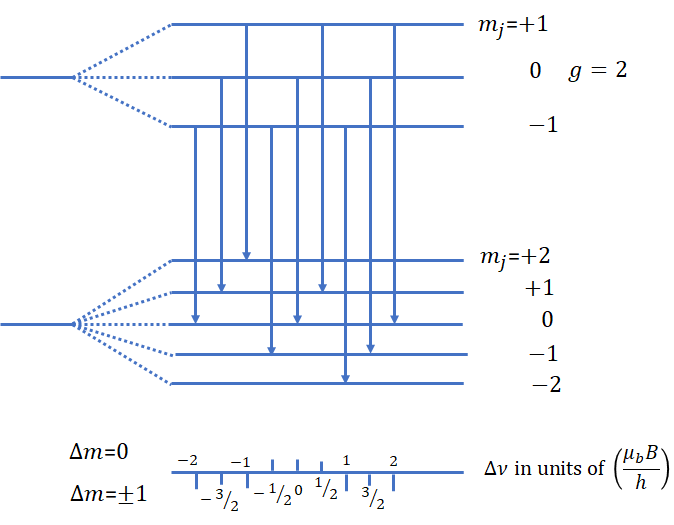
The anomalous Zeeman effect occurs when spectral lines split into more than three components.
This effect cannot be explained using classical theory alone.
It arises due to the influence of electron spin.
Role of Electron Spin
Electrons possess intrinsic spin angular momentum.
Spin contributes to the total magnetic moment of the atom.
The total angular momentum is given by
[
J = L + S
]
where
- (L) = orbital angular momentum
- (S) = spin angular momentum
Because of this interaction, energy levels split into more complicated patterns.
Landé g-Factor
The splitting of energy levels in the anomalous Zeeman effect is described using the Landé g-factor.
The Landé factor is given by
[
g = 1 + \frac{J(J+1) + S(S+1) – L(L+1)}{2J(J+1)}
]
This factor determines the magnitude of splitting in different atomic states.
Zeeman Splitting of Energy Levels
In the presence of a magnetic field, the energy of a state with magnetic quantum number (m_j) becomes
[
E = E_0 + g \mu_B B m_j
]
where
- (E_0) = original energy level
- (\mu_B) = Bohr magneton
- (B) = magnetic field strength
Each value of (m_j) corresponds to a different energy level.
Polarization of Zeeman Components
The spectral lines produced by the Zeeman effect have specific polarization properties.
Three types of components appear:
π Components
- No change in magnetic quantum number
- Linearly polarized
- Observed when viewed perpendicular to the magnetic field
σ⁺ Components
- Positive change in magnetic quantum number
- Circularly polarized
σ⁻ Components
- Negative change in magnetic quantum number
- Circularly polarized
These polarization patterns help identify Zeeman splitting experimentally.
Quantum Mechanical Explanation
Quantum mechanics provides a complete explanation of the Zeeman effect.
The interaction between magnetic field and electron magnetic moment modifies the Hamiltonian of the atom.
The perturbation term is
[
H’ = -\vec{\mu} \cdot \vec{B}
]
Using perturbation theory, the energy shifts of atomic states can be calculated.
Zeeman Effect in Hydrogen




The hydrogen atom provides a simple system for studying the Zeeman effect.
When hydrogen atoms are placed in a magnetic field:
- Energy levels split according to magnetic quantum numbers.
- Transitions between these levels produce multiple spectral lines.
Hydrogen often shows the normal Zeeman effect when spin interactions are negligible.
Experimental Observation
The Zeeman effect is observed using spectroscopic instruments such as:
- Spectroscopes
- Diffraction gratings
- Interferometers
The experimental setup usually includes:
- A light source (such as a discharge tube)
- A strong magnetic field
- A spectrometer to analyze spectral lines
When the magnetic field is applied, the spectral lines split and can be observed clearly.
Zeeman Effect vs Stark Effect
The Zeeman effect should not be confused with the Stark effect.
| Feature | Zeeman Effect | Stark Effect |
|---|---|---|
| Cause | Magnetic field | Electric field |
| Splitting | Magnetic interaction | Electric interaction |
| Discovery | Pieter Zeeman | Johannes Stark |
Both effects provide important information about atomic structure.
Applications of the Zeeman Effect
The Zeeman effect has numerous applications in science and technology.
Astronomy
Astronomers use the Zeeman effect to measure magnetic fields in stars and interstellar space.
By analyzing spectral line splitting, scientists can determine magnetic field strength in celestial objects.
Plasma Physics
The Zeeman effect helps study magnetic fields in plasma environments such as fusion reactors.
Magnetic Field Measurement
The Zeeman effect is used to measure extremely small magnetic fields with high precision.
Atomic Spectroscopy
Spectral line splitting provides detailed information about atomic energy levels and quantum states.
Laser Physics
Certain laser systems rely on transitions affected by Zeeman splitting.
Importance in Quantum Physics
The Zeeman effect played a major role in confirming the existence of electron spin and quantum angular momentum.
It provided strong experimental evidence supporting quantum mechanics and helped refine atomic models.
The phenomenon also helped scientists develop concepts such as:
- Magnetic quantum numbers
- Spin–orbit coupling
- Angular momentum quantization
Modern Research
Modern research continues to explore Zeeman splitting in various systems.
Scientists study Zeeman effects in:
- Cold atoms
- Quantum gases
- Solid-state systems
- Quantum computing devices
Advanced spectroscopic techniques allow extremely precise measurements of magnetic interactions.
Conclusion
The Zeeman effect is one of the most important phenomena in atomic physics and spectroscopy. It describes the splitting of atomic spectral lines when atoms are exposed to an external magnetic field. This splitting occurs because the magnetic moments associated with electron motion and spin interact with the magnetic field, altering atomic energy levels.
The discovery of the Zeeman effect provided strong evidence for the magnetic properties of electrons and played a crucial role in the development of quantum mechanics. The phenomenon is classified into normal and anomalous types depending on whether electron spin contributes to the splitting.
Today, the Zeeman effect is widely used in many scientific fields, including astrophysics, plasma physics, and spectroscopy. It remains an important tool for studying atomic structure, magnetic fields, and fundamental interactions in matter.
