

1. Introduction to Crystal Field Theory
Crystal Field Theory (CFT) is a theoretical model used to explain the electronic structure, magnetic properties, and colors of coordination compounds, particularly those involving transition metal ions. It describes how the presence of ligands surrounding a metal ion influences the energy levels of the metal’s d-orbitals.
The theory was developed in the early 20th century as part of efforts to understand the bonding and properties of coordination complexes. Although earlier theories such as Werner’s coordination theory explained the existence of coordination compounds, they did not fully explain properties like color and magnetism. Crystal Field Theory provided a more detailed understanding by considering the electrostatic interactions between metal ions and ligands.
In coordination compounds, ligands approach a central metal ion and interact with its d-electrons. These interactions cause the degeneracy of the five d-orbitals to be lifted, meaning that the orbitals split into groups with different energy levels. This phenomenon is known as crystal field splitting.
Crystal Field Theory plays an essential role in inorganic chemistry and helps explain many properties of coordination compounds, including:
- Color of transition metal complexes
- Magnetic properties
- Stability of complexes
- Geometry of coordination compounds
- Spectroscopic behavior
Although Crystal Field Theory treats ligands as point charges and does not consider covalent bonding effects, it remains a powerful and widely used model for understanding transition metal chemistry.
2. Basic Concepts of Crystal Field Theory
Crystal Field Theory is based on several key ideas.
Metal Ions and d-Orbitals
Transition metals possess partially filled d-orbitals, which play a crucial role in coordination chemistry.
The five d-orbitals are:
- dxy
- dyz
- dxz
- dx²−y²
- dz²
In an isolated metal atom, these orbitals have the same energy. This condition is known as degeneracy.
However, when ligands approach the metal ion, interactions occur that alter the energy levels of these orbitals.
Ligands as Point Charges
In CFT, ligands are treated as point charges or dipoles that produce an electrostatic field around the metal ion.
The repulsion between ligand electrons and metal d-electrons causes certain orbitals to increase in energy while others decrease.
This leads to splitting of d-orbitals.
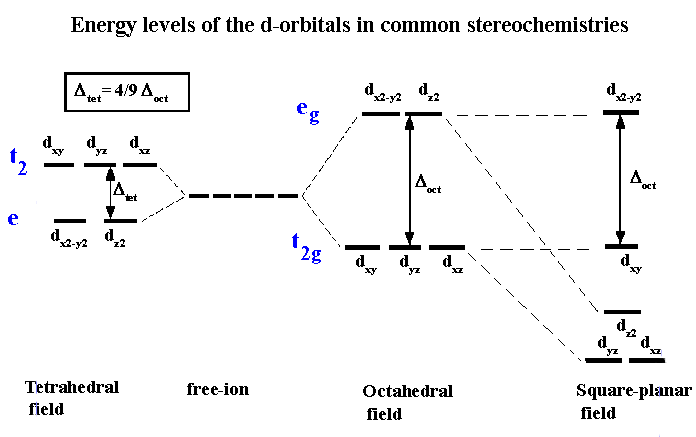
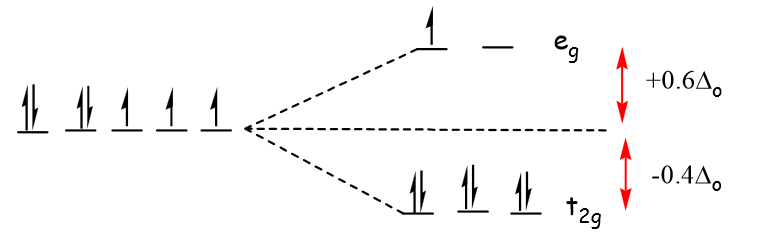
3. Crystal Field Splitting in Octahedral Complexes
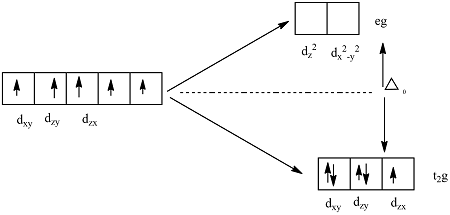
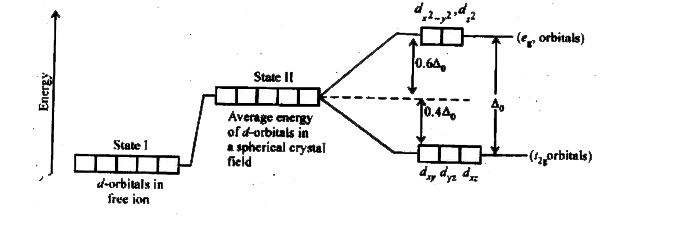
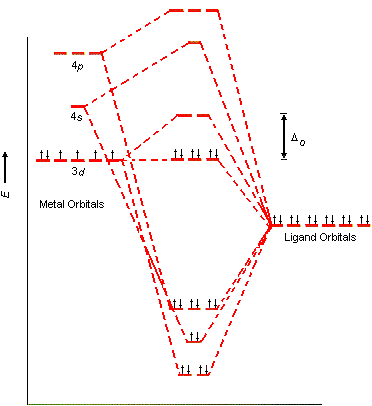
In an octahedral complex, six ligands surround the central metal ion along the x, y, and z axes.
When ligands approach the metal ion, they interact differently with the d-orbitals depending on orbital orientation.
Splitting of d-Orbitals
The five d-orbitals split into two groups:
Lower energy orbitals:
- dxy
- dyz
- dxz
These are called t₂g orbitals.
Higher energy orbitals:
- dx²−y²
- dz²
These are called e_g orbitals.
The energy difference between these two sets is called the crystal field splitting energy, represented as Δ₀ (delta octahedral).
4. Crystal Field Splitting in Tetrahedral Complexes



In tetrahedral complexes, four ligands surround the metal ion.
The orientation of ligands differs from the octahedral case, resulting in a different pattern of orbital splitting.
Splitting Pattern
The orbitals split into two groups:
Lower energy orbitals:
- dx²−y²
- dz²
Higher energy orbitals:
- dxy
- dyz
- dxz
The energy difference is represented as Δt.
In tetrahedral complexes:
Δt is smaller than Δ₀.
5. Factors Affecting Crystal Field Splitting
Several factors influence the magnitude of crystal field splitting.
Nature of the Metal Ion
Higher oxidation states increase splitting energy.
Example:
Fe³⁺ > Fe²⁺
Nature of the Ligands
Different ligands produce different field strengths.
This leads to the spectrochemical series.
Geometry of the Complex
Octahedral complexes usually have larger splitting than tetrahedral complexes.
Metal-Ligand Distance
Shorter metal–ligand distances increase splitting energy.
6. Spectrochemical Series
The spectrochemical series ranks ligands according to their ability to split d-orbitals.
Typical order:
I⁻ < Br⁻ < Cl⁻ < F⁻ < H₂O < NH₃ < CN⁻ < CO
Ligands on the left produce weak fields, while those on the right produce strong fields.
7. High-Spin and Low-Spin Complexes


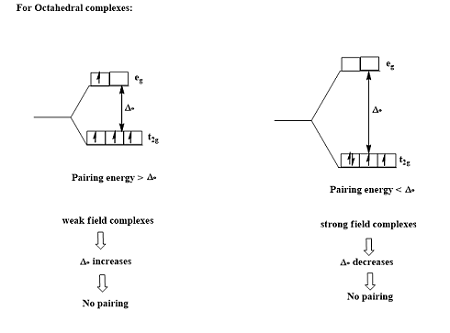
Electron arrangement in d-orbitals depends on:
- Crystal field splitting energy
- Electron pairing energy
Two possibilities exist:
High-Spin Complexes
Electrons occupy higher orbitals before pairing.
Occurs with weak-field ligands.
Example:
[Fe(H₂O)₆]²⁺
Low-Spin Complexes
Electrons pair in lower orbitals before occupying higher ones.
Occurs with strong-field ligands.
Example:
[Fe(CN)₆]⁴⁻
8. Crystal Field Stabilization Energy (CFSE)
Crystal Field Stabilization Energy measures the stabilization gained due to orbital splitting.
It depends on:
- Number of electrons in t₂g orbitals
- Number of electrons in e_g orbitals
CFSE influences the stability of coordination complexes.
9. Color of Coordination Compounds
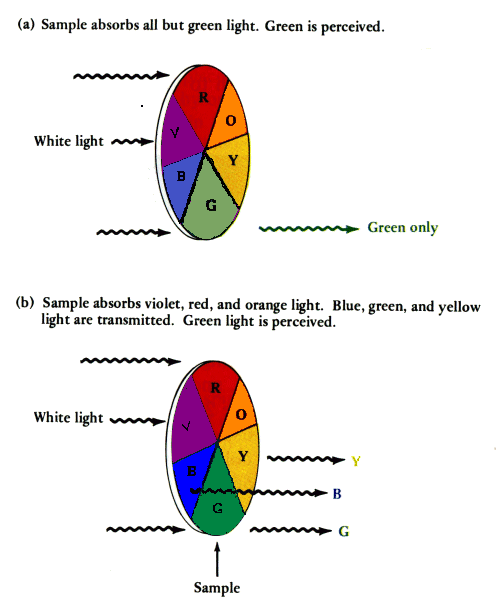


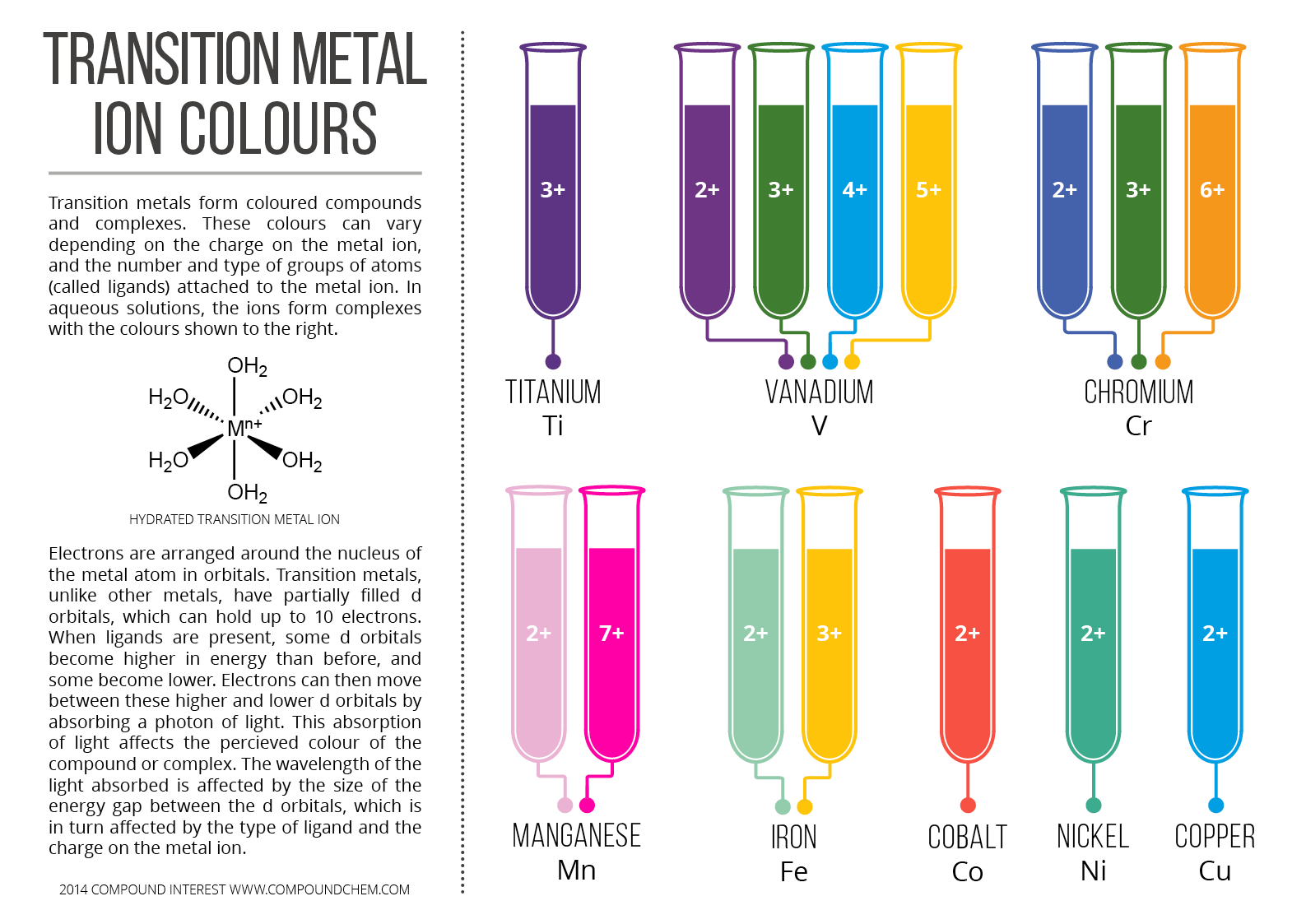
Many coordination compounds exhibit bright colors.
Color arises due to d–d electronic transitions.
When light is absorbed, electrons move from lower energy orbitals to higher energy orbitals.
The color observed corresponds to the wavelength of light not absorbed.
Example:
Copper complexes often appear blue.
10. Magnetic Properties of Complexes
Crystal Field Theory explains magnetic behavior.
Paramagnetic Complexes
Contain unpaired electrons.
Attracted by magnetic fields.
Diamagnetic Complexes
Contain only paired electrons.
Not attracted by magnetic fields.
Magnetic measurements help determine electron configuration.
11. Limitations of Crystal Field Theory
Although useful, CFT has limitations.
It assumes purely electrostatic interactions.
However, many metal–ligand bonds have covalent character.
To address this limitation, more advanced theories were developed:
- Ligand Field Theory
- Molecular Orbital Theory
12. Applications of Crystal Field Theory
Crystal Field Theory has numerous applications.
Coordination Chemistry
Explains structure and stability of complexes.
Spectroscopy
Helps interpret absorption spectra.
Bioinorganic Chemistry
Explains metal behavior in biological molecules.
Catalysis
Understanding electron configuration helps design catalysts.
13. Crystal Field Theory in Biological Systems




Crystal Field Theory helps explain metal ion behavior in biological systems.
Examples include:
- Hemoglobin
- Chlorophyll
- Metalloproteins
These molecules rely on metal–ligand interactions for biological functions.
14. Importance of Crystal Field Theory
Crystal Field Theory is a cornerstone of inorganic chemistry.
It helps scientists understand:
- Electronic structures of metal complexes
- Magnetic behavior
- Spectroscopic properties
- Chemical reactivity
The theory provides a simple yet powerful framework for analyzing coordination compounds.
Conclusion
Crystal Field Theory is an important model that explains the electronic structure and properties of coordination compounds. By describing how ligands interact with the d-orbitals of transition metal ions, the theory accounts for the splitting of orbital energies, which affects color, magnetism, and stability of complexes. Octahedral and tetrahedral geometries produce different splitting patterns, and factors such as ligand strength and metal oxidation state influence the magnitude of splitting. Concepts such as high-spin and low-spin complexes and crystal field stabilization energy further enhance the understanding of coordination chemistry. Although Crystal Field Theory has limitations, it remains a fundamental tool for interpreting the behavior of transition metal complexes in chemistry and biology.
