1. Introduction to Environmental and Green Chemistry
Environmental and green chemistry are modern branches of chemistry that focus on reducing environmental impact and promoting sustainable chemical processes. These fields aim to minimize pollution, conserve natural resources, and design chemical products and processes that are safer for humans and the environment.
Environmental chemistry studies the chemical processes occurring in the environment, including interactions between air, water, soil, and living organisms. It investigates how pollutants are produced, transported, transformed, and removed in natural systems.
Green chemistry, on the other hand, focuses on designing chemical processes and products that reduce or eliminate hazardous substances. The concept of green chemistry emerged in the late 20th century as scientists became increasingly concerned about pollution, waste generation, and environmental degradation caused by industrial activities.
Together, environmental and green chemistry contribute to solving many global challenges such as:
- Climate change
- Water pollution
- Air pollution
- Waste management
- Sustainable energy production
These fields are essential for developing technologies that protect ecosystems while supporting economic development.
2. Development of Environmental Chemistry
Environmental chemistry developed as a response to growing concerns about pollution during the industrial revolution and the 20th century.
Rapid industrialization led to significant environmental problems including:
- Air pollution from factories
- Water contamination from chemical waste
- Soil degradation
- Toxic chemical exposure
Scientists began studying the chemical composition of environmental systems to understand how pollutants affect ecosystems and human health.
Environmental chemistry now focuses on several areas:
- Atmospheric chemistry
- Aquatic chemistry
- Soil chemistry
- Biogeochemical cycles
3. Development of Green Chemistry
Green chemistry was formally introduced in the 1990s by chemists Paul Anastas and John Warner. They proposed a framework for designing chemical processes that minimize environmental harm.
Traditional chemistry often produced large amounts of hazardous waste and relied heavily on toxic chemicals. Green chemistry seeks to change this by promoting safer alternatives.
The goal of green chemistry is not simply to treat pollution after it occurs, but to prevent pollution at the source.
This approach involves:
- Designing environmentally friendly chemicals
- Using renewable resources
- Reducing energy consumption
- Minimizing waste production
4. The Twelve Principles of Green Chemistry


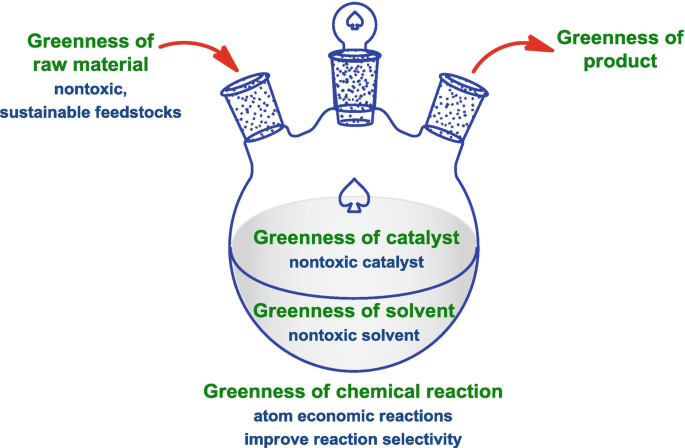
Green chemistry is guided by twelve fundamental principles.
1. Prevention
It is better to prevent waste than to treat or clean it up after it is formed.
2. Atom Economy
Chemical reactions should maximize incorporation of all materials into the final product.
3. Less Hazardous Chemical Syntheses
Synthetic methods should minimize toxicity.
4. Designing Safer Chemicals
Products should perform their function while minimizing toxicity.
5. Safer Solvents and Auxiliaries
Avoid unnecessary solvents and use safer alternatives.
6. Energy Efficiency
Chemical processes should minimize energy consumption.
7. Use of Renewable Feedstocks
Use renewable raw materials rather than depleting resources.
8. Reduce Derivatives
Avoid unnecessary chemical modifications.
9. Catalysis
Catalysts should be used instead of stoichiometric reagents.
10. Design for Degradation
Products should break down into harmless substances after use.
11. Real-Time Analysis for Pollution Prevention
Monitor chemical processes to prevent hazardous by-products.
12. Inherently Safer Chemistry
Design processes that minimize the potential for accidents.
5. Environmental Chemistry of the Atmosphere



Atmospheric chemistry studies chemical processes occurring in the Earth’s atmosphere.
Important topics include:
Air Pollution
Air pollution occurs when harmful substances accumulate in the atmosphere.
Major pollutants include:
- Carbon monoxide
- Sulfur dioxide
- Nitrogen oxides
- Particulate matter
These pollutants can cause respiratory problems and environmental damage.
Ozone Layer Depletion
The ozone layer protects Earth from harmful ultraviolet radiation.
Certain chemicals such as chlorofluorocarbons (CFCs) destroy ozone molecules.
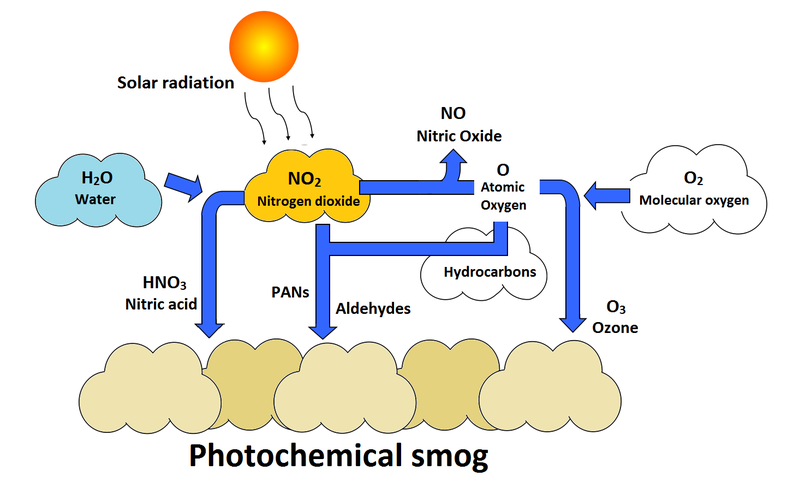
Photochemical Smog
Smog forms when nitrogen oxides react with hydrocarbons in the presence of sunlight.
It produces harmful compounds such as ozone and peroxyacyl nitrates.
6. Environmental Chemistry of Water



Water chemistry studies chemical processes occurring in rivers, lakes, oceans, and groundwater.
Water Pollution
Sources include:
- Industrial waste
- Agricultural runoff
- Domestic sewage
Common pollutants include:
- Heavy metals
- Pesticides
- Nutrients
Eutrophication
Excess nutrients cause rapid growth of algae in water bodies.
This reduces oxygen levels and harms aquatic life.
Water Treatment
Water purification involves several steps:
- Filtration
- Coagulation
- Disinfection
- Chemical treatment
7. Environmental Chemistry of Soil




Soil chemistry studies chemical processes occurring in soil systems.
Soil contains:
- Minerals
- Organic matter
- Water
- Microorganisms
Chemical reactions in soil influence nutrient availability for plants.
Soil Pollution
Soil contamination can result from:
- Industrial waste
- Agricultural chemicals
- Mining activities
Heavy metals such as lead and mercury can accumulate in soil and enter the food chain.
8. Biogeochemical Cycles
Biogeochemical cycles describe the movement of chemical elements through ecosystems.
Important cycles include:
Carbon Cycle
Carbon circulates between atmosphere, oceans, soil, and living organisms.
Nitrogen Cycle
Nitrogen moves through processes such as nitrogen fixation, nitrification, and denitrification.
Phosphorus Cycle
Phosphorus circulates through soil, water, and organisms.
These cycles maintain ecological balance.
9. Green Chemistry in Industry
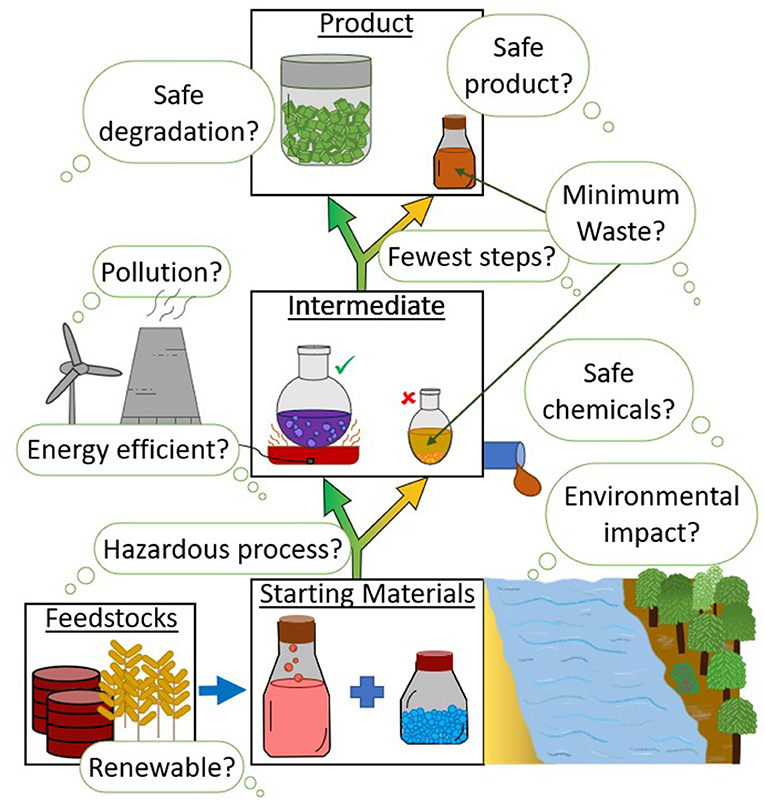

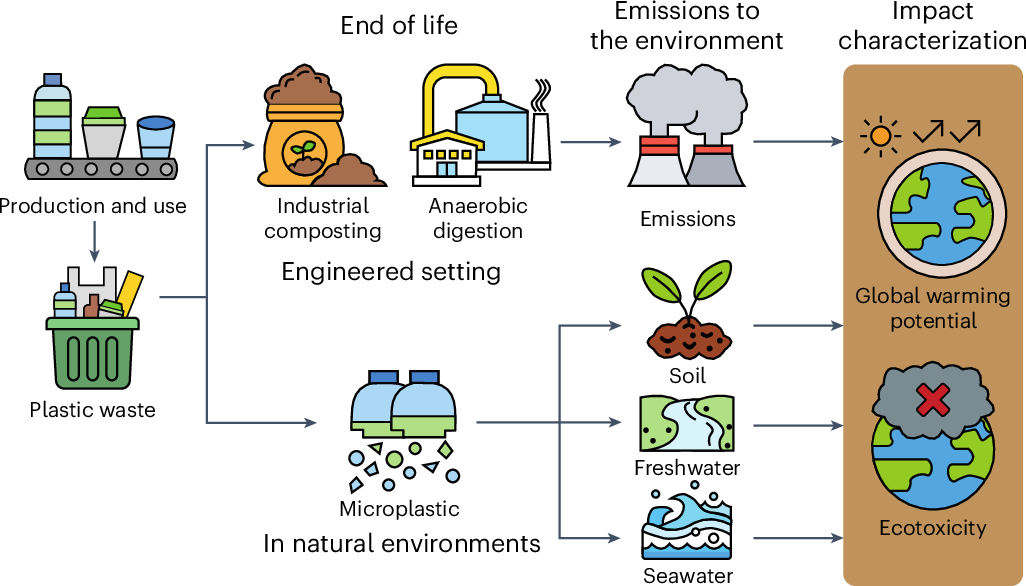
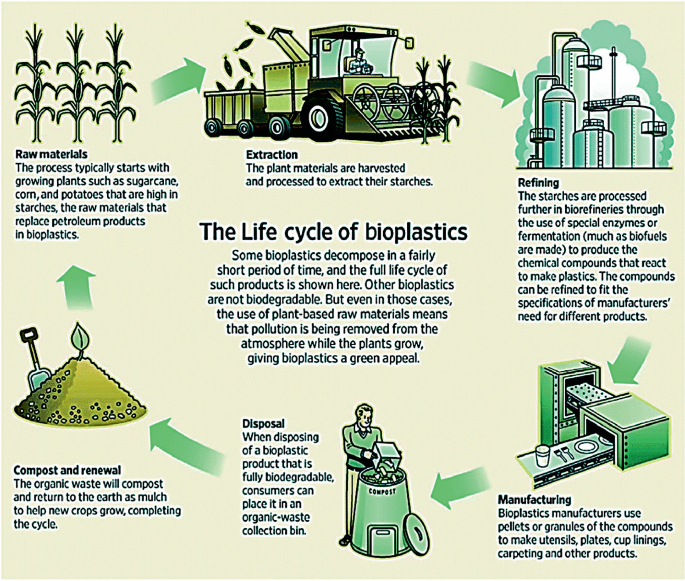
Green chemistry is transforming industrial practices.
Examples include:
Biodegradable Plastics
Development of environmentally friendly polymers.
Renewable Energy
Use of biomass and solar energy in chemical production.
Catalysis
Using catalysts to increase efficiency and reduce waste.
10. Green Solvents
Traditional solvents often pose environmental hazards.
Green solvents include:
- Water
- Supercritical carbon dioxide
- Ionic liquids
These solvents reduce environmental impact.
11. Sustainable Energy and Chemistry
Green chemistry supports sustainable energy technologies such as:
- Solar energy systems
- Hydrogen fuel cells
- Biofuels
These technologies reduce reliance on fossil fuels.
12. Importance of Environmental and Green Chemistry
Environmental and green chemistry play crucial roles in:
- Protecting ecosystems
- Preventing pollution
- Conserving natural resources
- Developing sustainable technologies
These fields help ensure that chemical science contributes positively to society and the environment.
Conclusion
Environmental and green chemistry represent a modern approach to chemical science focused on sustainability and environmental protection. Environmental chemistry studies the chemical processes occurring in air, water, soil, and living organisms, while green chemistry emphasizes the design of chemical processes that minimize waste and reduce environmental harm. The principles of green chemistry guide scientists in developing safer chemicals, renewable materials, and energy-efficient technologies. By integrating environmental awareness into chemical research and industry, these fields contribute to solving global challenges such as pollution, climate change, and resource depletion. Environmental and green chemistry therefore play a vital role in building a sustainable future.