



1. Introduction to Electrochemistry
Electrochemistry is a branch of chemistry that studies the relationship between chemical reactions and electrical energy. It focuses on processes in which chemical energy is converted into electrical energy and vice versa through redox (oxidation–reduction) reactions.
Electrochemical reactions involve the transfer of electrons between chemical species. These reactions are fundamental to many natural and technological processes such as batteries, fuel cells, electrolysis, corrosion, and electroplating.
Electrochemistry plays a vital role in modern science and technology. It has applications in:
- Energy storage systems
- Metal extraction and refining
- Industrial chemical production
- Corrosion prevention
- Environmental protection
- Biomedical devices
The field bridges chemistry, physics, materials science, and engineering, making it essential for the development of renewable energy technologies and advanced materials.
2. Fundamental Concepts in Electrochemistry
To understand electrochemistry, several key concepts must be understood.
1. Redox Reactions
Electrochemical reactions involve oxidation and reduction processes.
Oxidation → loss of electrons
Reduction → gain of electrons
Because electrons move between species, electrical energy can be generated or consumed.
2. Electrodes
Electrodes are conductive materials through which electrons enter or leave a system.
Two types of electrodes exist:
- Anode – oxidation occurs
- Cathode – reduction occurs
3. Electrolytes
Electrolytes are substances that conduct electricity when dissolved in water or molten form.
Examples include:
- Sodium chloride
- Sulfuric acid
- Potassium nitrate
Electrolytes allow the movement of ions, which is necessary for electrochemical reactions.
3. Electrochemical Cells




Electrochemical cells are devices that convert chemical energy into electrical energy or electrical energy into chemical energy.
They consist of:
- Two electrodes
- Electrolyte solution
- External circuit
- Salt bridge or membrane
Electrochemical cells are classified into two main types:
- Galvanic (voltaic) cells
- Electrolytic cells
4. Galvanic Cells (Voltaic Cells)
Galvanic cells generate electricity from spontaneous redox reactions.
Example: Daniell cell
Zn + Cu²⁺ → Zn²⁺ + Cu
In this cell:
Zinc electrode → anode
Copper electrode → cathode
At the anode:
Zn → Zn²⁺ + 2e⁻
At the cathode:
Cu²⁺ + 2e⁻ → Cu
Electrons flow through the external circuit from zinc to copper.
The salt bridge maintains electrical neutrality by allowing ions to move between solutions.
5. Electrolytic Cells
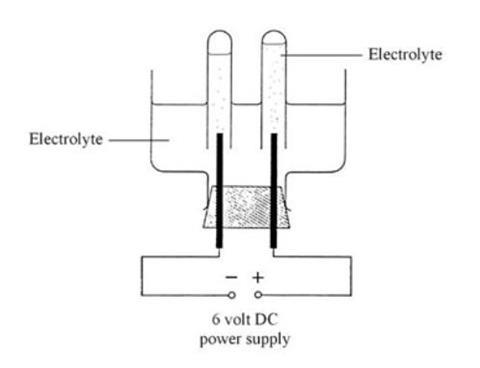


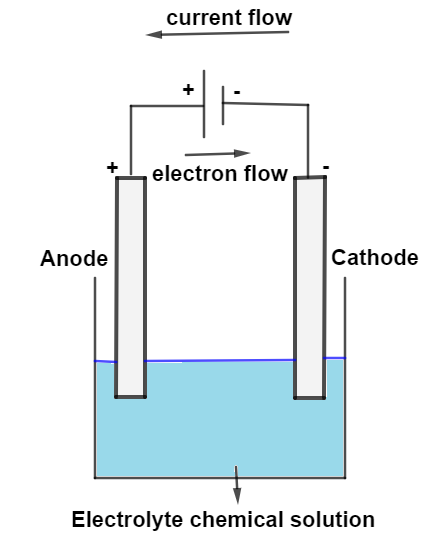
Electrolytic cells use electrical energy to drive non-spontaneous chemical reactions.
Unlike galvanic cells, these cells require an external power source.
Example: Electrolysis of water.
2H₂O → 2H₂ + O₂
In electrolysis:
- Anode → oxidation
- Cathode → reduction
Electric current forces the reaction to occur.
6. Differences Between Galvanic and Electrolytic Cells
| Feature | Galvanic Cell | Electrolytic Cell |
|---|---|---|
| Reaction type | Spontaneous | Non-spontaneous |
| Energy conversion | Chemical → electrical | Electrical → chemical |
| External power source | Not required | Required |
| Electron flow | Produced by reaction | Driven by power supply |
7. Electrode Potentials
Electrode potential measures the tendency of an electrode to gain or lose electrons.
It is measured relative to the standard hydrogen electrode (SHE).
Standard electrode potentials are measured under standard conditions:
- 1 M concentration
- 1 atm pressure
- 25°C temperature
Example standard potentials:
Zn²⁺/Zn = −0.76 V
Cu²⁺/Cu = +0.34 V
8. Standard Hydrogen Electrode




The standard hydrogen electrode (SHE) is used as a reference electrode.
Characteristics:
- Platinum electrode
- Hydrogen gas bubbled at 1 atm
- Hydrogen ion concentration = 1 M
The potential of SHE is defined as 0 volts.
All other electrode potentials are measured relative to it.
9. Cell Potential (EMF)
The electromotive force (EMF) of an electrochemical cell is the voltage produced by the cell.
Cell potential is calculated as:
E°cell = E°cathode − E°anode
Positive E°cell indicates spontaneous reaction.
Negative E°cell indicates non-spontaneous reaction.
10. Nernst Equation
The Nernst equation relates electrode potential to concentration.
[
E = E^\circ – \frac{0.059}{n} \log Q
]
Where:
- E = electrode potential
- E° = standard potential
- n = number of electrons
- Q = reaction quotient
This equation helps calculate cell potential under non-standard conditions.
11. Electrolysis



Electrolysis is the process of using electrical energy to drive chemical reactions.
Examples include:
- Electrolysis of water
- Electrolysis of molten salts
- Metal extraction
Example:
Molten NaCl electrolysis produces sodium metal and chlorine gas.
12. Faraday’s Laws of Electrolysis
Michael Faraday established two laws describing electrolysis.
First Law
The amount of substance deposited is proportional to the electric charge passed.
Second Law
Different substances deposited by the same charge are proportional to their equivalent weights.
These laws are important in electroplating and metal refining.
13. Electroplating




Electroplating is the process of depositing a metal layer onto another material using electrolysis.
Example:
Silver plating on jewelry.
Benefits include:
- Corrosion resistance
- Improved appearance
- Increased durability
14. Batteries and Fuel Cells
Electrochemistry is essential for energy storage.
Batteries
Batteries convert chemical energy into electrical energy.
Examples:
- Lithium-ion batteries
- Lead-acid batteries
- Alkaline batteries
Fuel Cells
Fuel cells generate electricity through chemical reactions.
Example:
Hydrogen fuel cell.
These devices are important for renewable energy technologies.
15. Corrosion and Electrochemistry




Corrosion is an electrochemical process in which metals deteriorate due to reactions with the environment.
Example:
Rusting of iron.
Methods to prevent corrosion include:
- Protective coatings
- Galvanization
- Cathodic protection
16. Industrial Applications of Electrochemistry
Electrochemistry is used in many industrial processes.
Examples include:
- Aluminum extraction
- Chlor-alkali process
- Electroplating
- Metal purification
- Hydrogen production
17. Environmental Applications
Electrochemistry is used to address environmental issues.
Examples include:
- Water purification
- Waste treatment
- Energy storage for renewable sources
- Fuel cell technology
18. Importance of Electrochemistry
Electrochemistry is important for understanding:
- Energy conversion
- Corrosion processes
- Electrochemical sensors
- Renewable energy systems
The field is essential for developing sustainable technologies.
Conclusion
Electrochemistry is a vital branch of chemistry that studies the relationship between chemical reactions and electrical energy. Through electrochemical cells, chemical energy can be converted into electricity, and electrical energy can drive chemical reactions. Concepts such as electrode potentials, electrolysis, and Faraday’s laws form the foundation of electrochemical science. Applications of electrochemistry are widespread, including batteries, fuel cells, corrosion prevention, electroplating, and industrial chemical processes. As the demand for renewable energy and sustainable technologies increases, electrochemistry continues to play an increasingly important role in scientific and technological advancements.
