



1. Introduction to Hydrocarbons
Hydrocarbons are a fundamental class of organic compounds composed only of carbon and hydrogen atoms. They form the backbone of organic chemistry and are the simplest organic molecules. Hydrocarbons are extremely important in both natural systems and industrial applications because they are the primary components of fossil fuels such as petroleum, natural gas, and coal.
The carbon atom has the unique ability to form stable covalent bonds with itself and with other elements. Because of this property, carbon atoms can form long chains, branched structures, and rings, leading to an enormous variety of hydrocarbon compounds.
Hydrocarbons are important in many areas, including:
- Fuel production (gasoline, diesel, natural gas)
- Petrochemical industries
- Polymer manufacturing
- Pharmaceutical synthesis
- Lubricants and solvents
- Plastics and synthetic materials
Hydrocarbons serve as the starting materials for the production of thousands of organic chemicals used in everyday life.
2. Basic Structure of Hydrocarbons
Hydrocarbons consist of two elements:
- Carbon (C)
- Hydrogen (H)
Carbon atoms have four valence electrons and can form four covalent bonds. This allows carbon atoms to connect with each other in different ways, producing a wide range of structures.
Hydrocarbon structures may include:
- Straight chains
- Branched chains
- Cyclic rings
- Aromatic rings
The diversity of hydrocarbon structures is the reason organic chemistry contains millions of different compounds.
3. Classification of Hydrocarbons
Hydrocarbons are broadly classified into two major categories:
- Aliphatic hydrocarbons
- Aromatic hydrocarbons
Aliphatic hydrocarbons are further divided into:
- Alkanes
- Alkenes
- Alkynes
4. Aliphatic Hydrocarbons



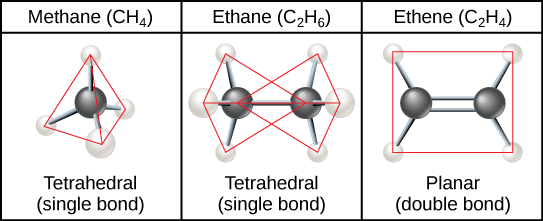
Aliphatic hydrocarbons are hydrocarbons arranged in open chains rather than rings.
They are divided into:
Saturated hydrocarbons
Contain only single carbon–carbon bonds.
Example: Alkanes
Unsaturated hydrocarbons
Contain double or triple bonds.
Examples: Alkenes and Alkynes
5. Alkanes
Alkanes are saturated hydrocarbons containing only single carbon–carbon bonds.
General formula:
[
C_nH_{2n+2}
]
Examples:
Methane – CH₄
Ethane – C₂H₆
Propane – C₃H₈
Butane – C₄H₁₀
Alkanes are relatively chemically stable because single bonds are strong and less reactive.
Properties of Alkanes
- Nonpolar molecules
- Insoluble in water
- Soluble in organic solvents
- Low chemical reactivity
Uses of Alkanes
- Fuels (natural gas, LPG)
- Lubricants
- Paraffin wax
- Petrochemical feedstocks
6. Alkenes



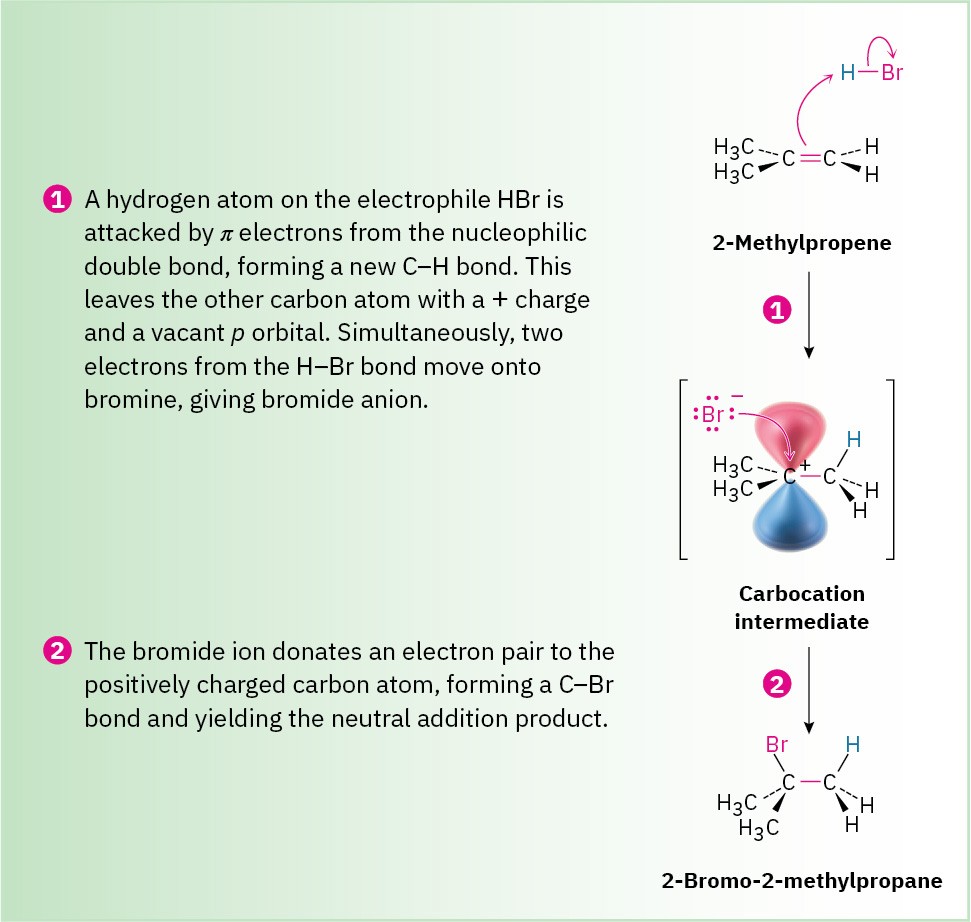
Alkenes are unsaturated hydrocarbons containing at least one carbon–carbon double bond.
General formula:
[
C_nH_{2n}
]
Example:
Ethene – C₂H₄
Double bonds make alkenes more reactive than alkanes.
Reactions of Alkenes
Common reactions include:
- Addition reactions
- Polymerization
- Hydrogenation
- Halogenation
Example:
Ethene + H₂ → Ethane
Alkenes are widely used in the plastic industry.
Example:
Polyethylene is produced from ethene.
7. Alkynes




Alkynes are unsaturated hydrocarbons containing at least one carbon–carbon triple bond.
General formula:
[
C_nH_{2n-2}
]
Example:
Ethyne (acetylene) – C₂H₂
Alkynes are highly reactive due to the presence of triple bonds.
Uses of Alkynes
- Welding and cutting metals
- Chemical synthesis
- Production of polymers
Acetylene is commonly used in oxy-acetylene welding.
8. Aromatic Hydrocarbons




Aromatic hydrocarbons contain benzene rings or similar cyclic structures.
Example:
Benzene – C₆H₆
Aromatic compounds have special stability due to delocalized electrons.
Common aromatic hydrocarbons include:
- Benzene
- Toluene
- Xylene
- Naphthalene
Applications
- Solvents
- Pharmaceutical synthesis
- Dyes and pigments
- Plastics manufacturing
9. Physical Properties of Hydrocarbons
Hydrocarbon properties depend on molecular structure.
Boiling and Melting Points
Increase with molecular size.
Solubility
Hydrocarbons are generally insoluble in water but soluble in organic solvents.
Density
Most hydrocarbons are less dense than water.
State of Matter
- Small hydrocarbons → gases
- Medium hydrocarbons → liquids
- Large hydrocarbons → solids
10. Chemical Reactions of Hydrocarbons
Hydrocarbons undergo various chemical reactions.
Combustion
Hydrocarbons burn in oxygen to produce carbon dioxide and water.
Example:
CH₄ + 2O₂ → CO₂ + 2H₂O
Combustion reactions release large amounts of energy.
Substitution Reactions
Common in alkanes.
Example:
CH₄ + Cl₂ → CH₃Cl + HCl
Addition Reactions
Common in alkenes and alkynes.
Example:
Ethene + Br₂ → Dibromoethane
Polymerization
Small molecules combine to form polymers.
Example:
Ethene → Polyethylene
11. Sources of Hydrocarbons

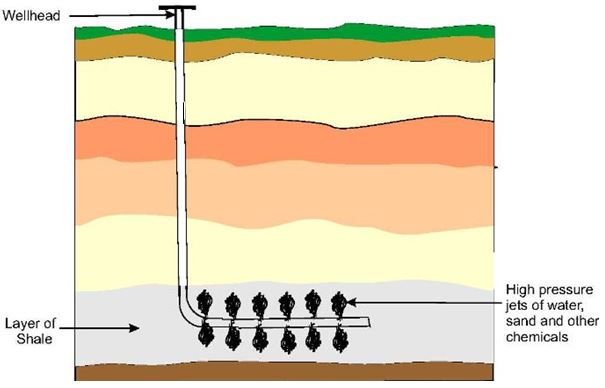


Hydrocarbons occur naturally in fossil fuels.
Major sources include:
Petroleum (Crude Oil)
A complex mixture of hydrocarbons.
Refined into:
- Gasoline
- Diesel
- Kerosene
- Lubricants
Natural Gas
Contains mainly methane.
Used as fuel and chemical feedstock.
Coal
Contains complex hydrocarbon structures.
Used to produce coal gas and coke.
12. Environmental Impact of Hydrocarbons
Hydrocarbon use has environmental consequences.
Examples include:
- Air pollution
- Greenhouse gas emissions
- Oil spills
- Climate change
Burning hydrocarbons releases carbon dioxide, contributing to global warming.
13. Industrial Applications
Hydrocarbons are essential in many industries.
Examples include:
- Fuel production
- Plastic manufacturing
- Synthetic rubber production
- Pharmaceutical synthesis
- Chemical solvents
Petrochemical industries convert hydrocarbons into thousands of products.
14. Importance of Hydrocarbons
Hydrocarbons are fundamental to modern society.
They provide:
- Energy resources
- Raw materials for chemicals
- Building blocks for organic synthesis
- Materials for plastics and fibers
Understanding hydrocarbons is essential for energy production and chemical manufacturing.
Conclusion
Hydrocarbons are organic compounds consisting solely of carbon and hydrogen atoms. They form the foundation of organic chemistry and are widely used as fuels, industrial chemicals, and raw materials for numerous products. Hydrocarbons are classified into alkanes, alkenes, alkynes, and aromatic compounds based on their structures and bonding patterns. Their physical and chemical properties depend on molecular structure and bonding. Hydrocarbons are abundant in natural resources such as petroleum, natural gas, and coal, and they play a vital role in modern energy systems and industrial processes. Despite their importance, responsible use and management are necessary to minimize environmental impacts and ensure sustainable development.
