



1. Introduction to Redox Reactions
Redox reactions, short for reduction–oxidation reactions, are among the most fundamental types of chemical reactions in chemistry. They involve the transfer of electrons between chemical species, leading to changes in oxidation states. These reactions are essential in many natural processes, biological systems, and industrial applications.
The term redox combines two processes:
- Oxidation – loss of electrons
- Reduction – gain of electrons
Because electrons are transferred from one substance to another, oxidation and reduction always occur simultaneously. This is why they are considered a pair of reactions.
Redox reactions are involved in numerous everyday and industrial processes, such as:
- Rusting of iron
- Combustion of fuels
- Photosynthesis
- Cellular respiration
- Battery operation
- Corrosion prevention
- Metallurgical processes
Understanding redox reactions is essential in fields like electrochemistry, environmental chemistry, biochemistry, and industrial chemistry.
2. Historical Development of Redox Concepts
Historically, oxidation and reduction were defined differently from modern definitions.
Originally:
- Oxidation referred to reactions involving oxygen.
- Reduction referred to the removal of oxygen.
Example:
2Mg + O₂ → 2MgO
Magnesium is oxidized because it combines with oxygen.
Later, the concept expanded to include hydrogen transfer.
Oxidation: loss of hydrogen
Reduction: gain of hydrogen.
Modern chemistry defines oxidation and reduction based on electron transfer.
3. Modern Definition of Oxidation and Reduction
According to the modern definition:
Oxidation is the loss of electrons.
Reduction is the gain of electrons.
Example reaction:
Zn + Cu²⁺ → Zn²⁺ + Cu
In this reaction:
Zn → Zn²⁺ + 2e⁻ (oxidation)
Cu²⁺ + 2e⁻ → Cu (reduction)
Thus:
- Zinc is oxidized
- Copper ions are reduced
4. Oxidizing and Reducing Agents
In redox reactions, certain substances cause oxidation or reduction.
Oxidizing Agent
An oxidizing agent is a substance that accepts electrons and causes another substance to be oxidized.
Example:
Cu²⁺ acts as oxidizing agent.
Reducing Agent
A reducing agent is a substance that donates electrons and causes another substance to be reduced.
Example:
Zn acts as reducing agent.
5. Oxidation Number Concept




The oxidation number (oxidation state) represents the apparent charge of an atom in a compound.
It helps identify oxidation and reduction processes.
Rules for Assigning Oxidation Numbers
- Elements in free state → oxidation number = 0
- Alkali metals → +1
- Alkaline earth metals → +2
- Oxygen usually → −2
- Hydrogen → +1 (with nonmetals)
- Sum of oxidation numbers equals overall charge.
Example:
H₂O
H = +1
O = −2
6. Identifying Redox Reactions
Redox reactions can be identified by observing changes in oxidation states.
Example:
Fe²⁺ → Fe³⁺
Increase in oxidation number → oxidation.
Example:
Cl₂ → 2Cl⁻
Decrease in oxidation number → reduction.
7. Types of Redox Reactions
Redox reactions can be classified into several categories.
1. Combination Reactions


Two or more substances combine to form one product.
Example:
2Mg + O₂ → 2MgO
Magnesium is oxidized.
2. Decomposition Reactions
A compound breaks down into simpler substances.
Example:
2HgO → 2Hg + O₂
Mercury oxide decomposes into mercury and oxygen.
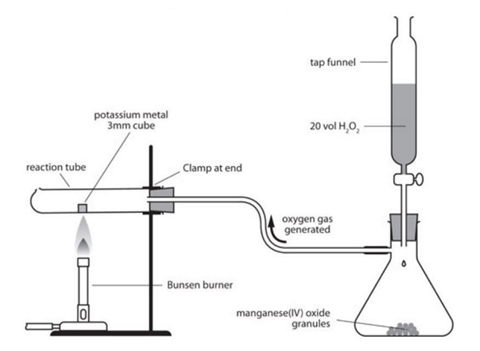
3. Displacement Reactions
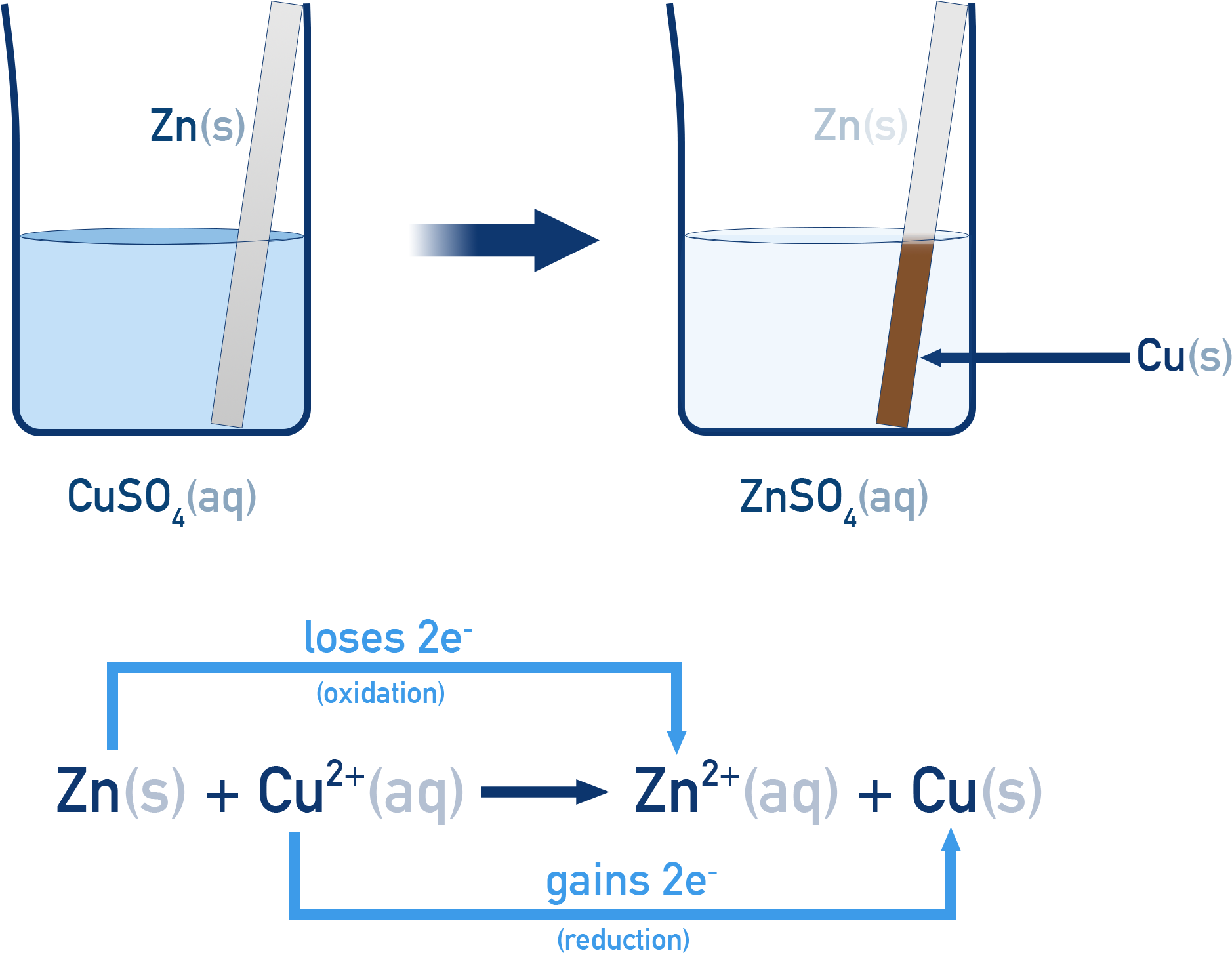


One element replaces another in a compound.
Example:
Zn + CuSO₄ → ZnSO₄ + Cu
4. Disproportionation Reactions
In disproportionation reactions, the same element undergoes both oxidation and reduction.
Example:
2H₂O₂ → 2H₂O + O₂
Hydrogen peroxide acts as both oxidizing and reducing agent.
8. Balancing Redox Reactions
Balancing redox reactions involves ensuring that both mass and charge are conserved.
Two methods are commonly used:
- Oxidation number method
- Half-reaction method
9. Half-Reaction Method
The half-reaction method separates oxidation and reduction reactions.
Example:
Zn → Zn²⁺ + 2e⁻
Cu²⁺ + 2e⁻ → Cu
Electrons lost must equal electrons gained.
10. Redox Reactions in Electrochemistry




Electrochemical cells use redox reactions to produce electricity.
Example:
Zn–Cu galvanic cell.
Oxidation occurs at anode.
Reduction occurs at cathode.
Electrons flow through external circuit.
11. Redox Reactions in Biological Systems
Redox reactions are essential for life.
Examples include:
Cellular Respiration
Glucose is oxidized to produce energy.
Photosynthesis
Carbon dioxide is reduced to form glucose.
Electron Transport Chain
Electrons are transferred through proteins to produce ATP.
12. Redox Reactions in Industrial Processes
Redox reactions are widely used in industry.
Examples include:
- Metal extraction from ores
- Chemical manufacturing
- Petroleum refining
- Corrosion control
Example:
Iron extraction in blast furnace.
Fe₂O₃ + 3CO → 2Fe + 3CO₂
13. Corrosion and Rusting



Corrosion is a destructive redox reaction involving metals.
Rusting occurs when iron reacts with oxygen and water.
Reaction:
4Fe + 3O₂ + 6H₂O → 4Fe(OH)₃
This eventually forms rust.
14. Redox Titrations
Redox titrations are analytical techniques used to determine concentrations.
Examples:
- Permanganate titration
- Dichromate titration
- Iodometric titration
These methods rely on oxidation–reduction reactions.
15. Environmental Importance of Redox Reactions
Redox reactions occur in many environmental processes.
Examples include:
- Oxygen cycling in ecosystems
- Nitrogen cycle reactions
- Decomposition of organic matter
- Atmospheric chemical reactions
Understanding redox chemistry helps scientists address pollution and environmental changes.
16. Applications of Redox Reactions
Redox reactions are used in many practical applications.
Batteries
Energy storage and electricity generation.
Fuel Cells
Clean energy technology.
Metallurgy
Extraction and purification of metals.
Water Treatment
Oxidizing agents remove pollutants.
Medicine
Redox reactions occur in metabolic pathways.
17. Importance of Redox Chemistry
Redox reactions are essential for understanding:
- Energy transformations
- Chemical reactions
- Biological metabolism
- Industrial chemical processes
They form the basis of electrochemistry and energy technologies.
Conclusion
Redox reactions are fundamental chemical processes involving the transfer of electrons between substances. These reactions involve oxidation, where electrons are lost, and reduction, where electrons are gained. Understanding redox chemistry helps scientists analyze chemical reactions, design electrochemical devices, study biological processes, and develop industrial technologies. From rusting of metals to energy generation in batteries and cellular respiration in living organisms, redox reactions play a vital role in both natural and technological systems.
