


Introduction
Cryogenics is the branch of physics and engineering that studies the production and behavior of materials at extremely low temperatures. Typically, cryogenics deals with temperatures below −150°C (123 K), where gases begin to liquefy and matter exhibits unusual physical properties.
The term cryogenics originates from the Greek words kryos (meaning cold) and genics (meaning producing). In simple terms, cryogenics refers to the science and technology of generating and maintaining very low temperatures.
At cryogenic temperatures, many materials behave differently compared with their behavior at room temperature. For example, certain metals become superconductors, gases become liquids, and chemical reactions slow down dramatically.
Cryogenics has numerous applications in science and technology, including medical imaging, space exploration, superconductivity research, particle physics, and food preservation.
The development of cryogenics began with experiments on gas liquefaction conducted by scientists such as Heike Kamerlingh Onnes, who successfully liquefied helium and studied the behavior of materials at extremely low temperatures.
Today, cryogenic technology is essential in many advanced scientific and industrial systems.
Cryogenic Temperature Range
Cryogenic temperatures are defined as temperatures significantly below ordinary environmental temperatures.
Typical cryogenic temperature ranges include:
- Liquid nitrogen: 77 K (−196°C)
- Liquid oxygen: 90 K (−183°C)
- Liquid hydrogen: 20 K (−253°C)
- Liquid helium: 4 K (−269°C)
Liquid helium represents one of the coldest substances commonly used in cryogenic experiments.
At these temperatures, materials may exhibit phenomena such as superconductivity and superfluidity.
Cryogenic Fluids




Cryogenic fluids are substances that remain liquid only at extremely low temperatures.
Common cryogenic liquids include:
Liquid Nitrogen
- Temperature: 77 K
- Widely used in laboratories and industrial applications
Liquid Oxygen
- Temperature: 90 K
- Used in rocket propulsion and medical applications
Liquid Hydrogen
- Temperature: 20 K
- Used as rocket fuel
Liquid Helium
- Temperature: 4 K
- Used for superconducting magnets and research experiments
These liquids must be stored in specially insulated containers called Dewar flasks to minimize heat transfer.
Principles of Cryogenic Cooling
Cryogenic cooling relies on several thermodynamic processes.
Gas Liquefaction
The process of converting gases into liquids requires cooling and compression.
The behavior of gases during cooling is described by thermodynamic gas laws.
genui{“math_block_widget_common_keywords”:{“content”:”PV = nRT”}}
This equation relates pressure, volume, and temperature for ideal gases.
Lowering temperature or increasing pressure can lead to condensation of gases into liquids.
Joule–Thomson Effect
The Joule–Thomson effect describes the temperature change that occurs when a gas expands without exchanging heat with its surroundings.
When certain gases expand through a valve or porous plug, they cool down.
This effect is widely used in cryogenic refrigeration systems.
Adiabatic Expansion
Adiabatic expansion occurs when a gas expands without gaining or losing heat.
During expansion, the gas performs work and its temperature decreases.
This principle is used in cryogenic cooling systems.
Cryogenic Equipment




Cryogenic systems require specialized equipment to achieve and maintain low temperatures.
Dewar Flask
A Dewar flask is a double-walled container with vacuum insulation.
It minimizes heat transfer and keeps cryogenic liquids cold.
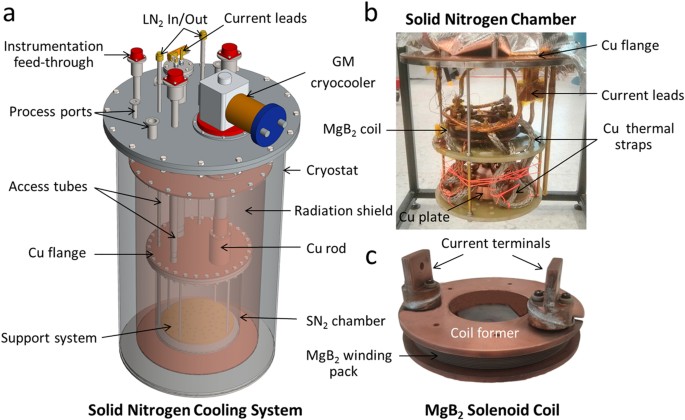
Cryostat
A cryostat is a device used to maintain low temperatures for experiments.
It allows scientists to study materials at cryogenic temperatures.
Cryogenic Refrigerators
These systems use thermodynamic cycles to produce extremely low temperatures.
Examples include:
- Stirling refrigerators
- Pulse-tube refrigerators
- Dilution refrigerators
Properties of Materials at Cryogenic Temperatures
At extremely low temperatures, materials exhibit unique physical properties.
Superconductivity

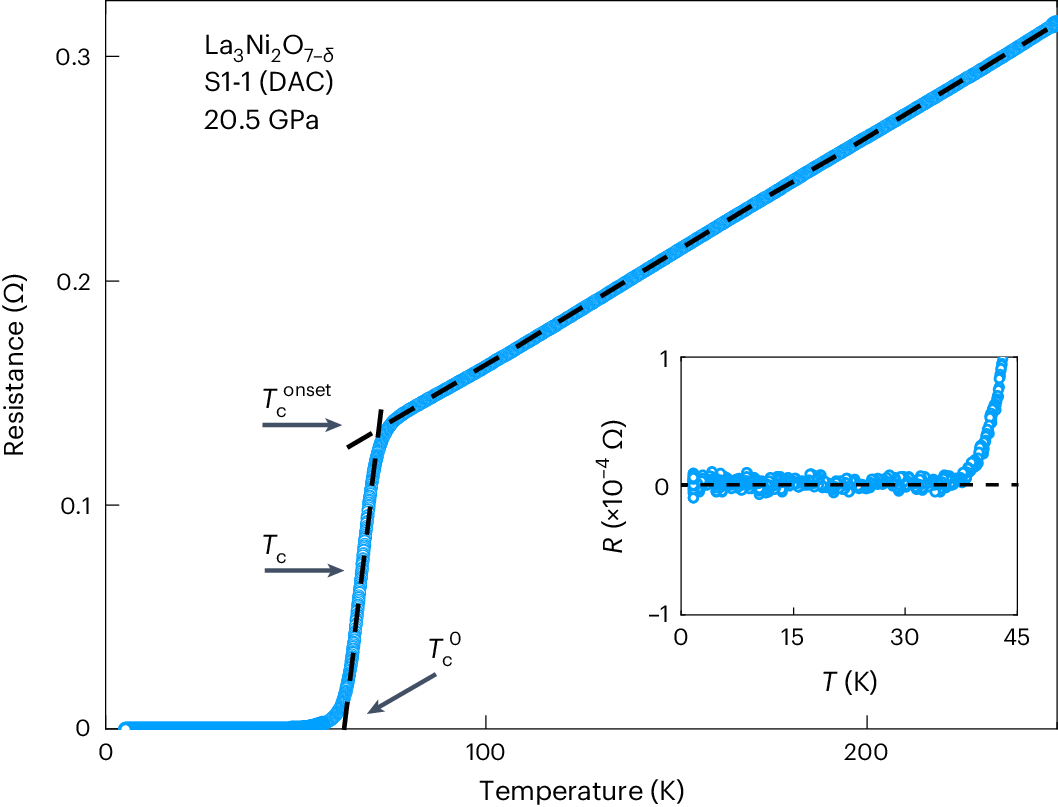
Some materials lose all electrical resistance at cryogenic temperatures.
This phenomenon is known as superconductivity.
Superconductors can carry electric current indefinitely without energy loss.
Applications include:
- MRI machines
- Particle accelerators
- Magnetic levitation trains
Superfluidity
Certain liquids, such as liquid helium, exhibit superfluidity at extremely low temperatures.
Superfluids flow without viscosity and can move through tiny pores without resistance.
Thermal Properties
At cryogenic temperatures:
- Thermal conductivity may increase or decrease depending on the material.
- Heat capacity becomes extremely small.
These properties influence cryogenic engineering designs.
Cryogenic Storage and Safety
Handling cryogenic liquids requires special safety precautions.
Potential hazards include:
Extreme Cold
Cryogenic liquids can cause severe frostbite upon contact with skin.
Oxygen Condensation
Liquid nitrogen can condense oxygen from air, creating fire hazards.
Pressure Build-Up
Cryogenic liquids expand rapidly when warmed.
Proper ventilation and pressure relief systems are necessary.
Applications of Cryogenics
Cryogenic technology has many important applications.
Medical Applications
Cryogenics is used in medicine for imaging and treatment.
Examples include:
- Magnetic resonance imaging (MRI)
- Cryosurgery for removing tumors
- Cryopreservation of biological samples
Space Exploration


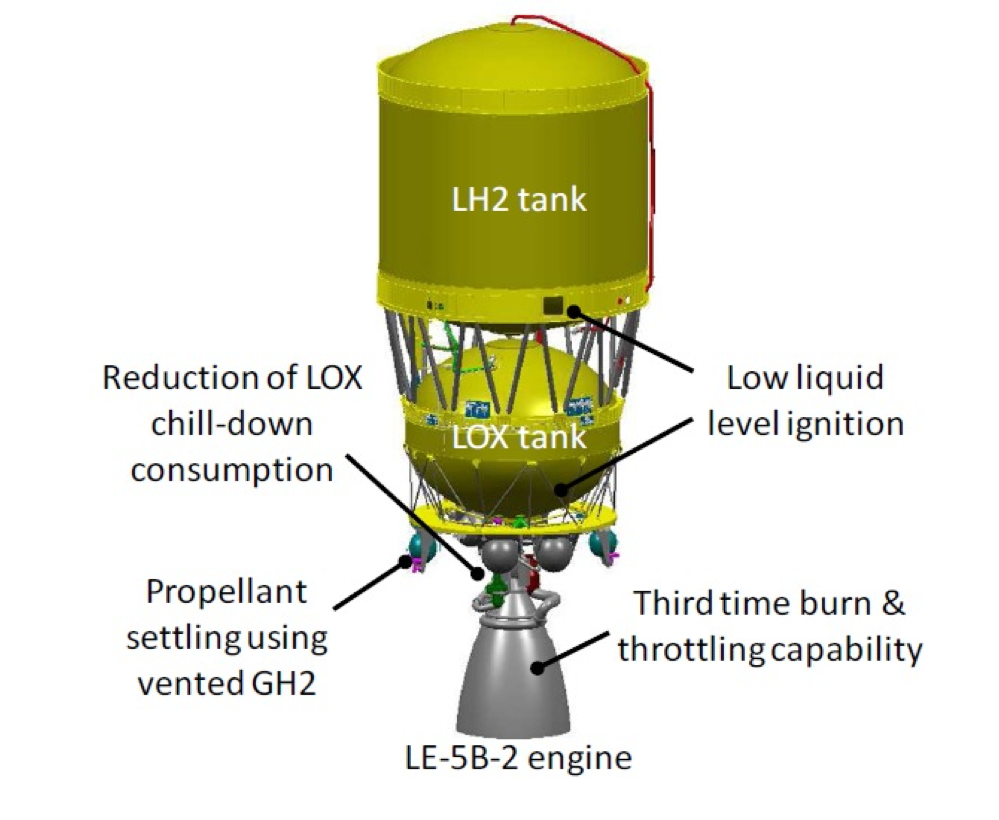

Cryogenic fuels are widely used in rocket propulsion.
Liquid hydrogen and liquid oxygen provide high energy efficiency for rockets.
These fuels must be stored at extremely low temperatures.
Particle Physics
Cryogenic systems are used in large research facilities such as the CERN.
Superconducting magnets cooled by liquid helium are essential for particle accelerators.
Food Preservation
Cryogenic freezing is used to preserve food products.
Rapid freezing maintains food quality and prevents microbial growth.
Industrial Applications
Cryogenic techniques are used in industries such as:
- Metal processing
- Electronics manufacturing
- Chemical production
Cryogenics in Scientific Research
Cryogenic environments allow scientists to study fundamental physical phenomena.
Examples include:
- Quantum behavior of materials
- Superconductivity experiments
- Low-temperature physics
Cryogenic technology is essential for many modern physics experiments.
Cryogenics and Quantum Technology
Cryogenics plays a crucial role in quantum computing.
Quantum processors often operate at temperatures close to absolute zero.
These low temperatures reduce thermal noise and allow quantum states to remain stable.
Future of Cryogenics
Advances in cryogenic technology continue to expand its applications.
Future developments may include:
- More efficient cryogenic refrigeration systems
- Advanced superconducting technologies
- Improved cryogenic fuel storage for space missions
- Enhanced quantum computing systems
Cryogenics is expected to remain a key field supporting scientific and technological progress.
Conclusion
Cryogenics is the science and technology of extremely low temperatures and their effects on materials. By cooling substances to cryogenic temperatures, scientists can observe unusual physical phenomena such as superconductivity and superfluidity.
Cryogenic technology has become essential in many fields, including medicine, aerospace engineering, particle physics, and materials science. Specialized equipment such as cryostats and Dewar flasks allow researchers to maintain extremely low temperatures for experiments and industrial processes.
As scientific research advances, cryogenics will continue to play an important role in exploring new technologies and understanding the behavior of matter under extreme conditions.
