



1. Introduction to Ionic Bonding
Ionic bonding is one of the fundamental types of chemical bonding that explains how atoms combine to form compounds. It occurs when atoms transfer electrons from one atom to another, resulting in the formation of charged particles called ions. These oppositely charged ions attract each other through electrostatic forces, forming an ionic bond.
Ionic bonding is commonly observed between metals and nonmetals. Metals tend to lose electrons easily, forming positively charged ions called cations, while nonmetals tend to gain electrons, forming negatively charged ions called anions.
The attraction between these ions results in the formation of ionic compounds, which often exhibit characteristic properties such as high melting points, crystalline structures, and the ability to conduct electricity when dissolved in water.
Ionic bonding plays an essential role in many natural and industrial processes. Common substances such as table salt, minerals, and many inorganic compounds are formed through ionic bonding.
Understanding ionic bonding helps scientists explain the structure and properties of many materials, predict chemical reactions, and design new compounds for technological and industrial applications.
2. Formation of Ions



Ionic bonding begins with the formation of ions. An ion is an atom or molecule that carries an electric charge due to the loss or gain of electrons.
Atoms form ions to achieve a stable electron configuration, usually resembling the configuration of noble gases.
Cations
Cations are positively charged ions formed when atoms lose one or more electrons.
Metals commonly form cations because they have relatively low ionization energies.
Examples:
Sodium → Na⁺
Magnesium → Mg²⁺
Aluminum → Al³⁺
These ions form when metal atoms lose electrons from their outermost shells.
Anions
Anions are negatively charged ions formed when atoms gain electrons.
Nonmetals typically form anions because they have high electron affinity and electronegativity.
Examples:
Chlorine → Cl⁻
Oxygen → O²⁻
Nitrogen → N³⁻
By gaining electrons, these atoms achieve stable electron configurations.
3. Electron Transfer and Ionic Bond Formation




The formation of an ionic bond involves electron transfer from a metal atom to a nonmetal atom.
A classic example is the formation of sodium chloride.
Step 1: Sodium Atom Loses an Electron
Sodium has one valence electron.
It loses this electron to achieve a stable configuration.
Na → Na⁺ + e⁻
Step 2: Chlorine Atom Gains an Electron
Chlorine requires one electron to complete its outer shell.
Cl + e⁻ → Cl⁻
Step 3: Electrostatic Attraction
The oppositely charged ions attract each other.
Na⁺ + Cl⁻ → NaCl
This attraction between ions forms the ionic bond.
Unlike covalent bonds, electrons are not shared but transferred completely.
4. Ionic Crystal Lattice Structure




Ionic compounds do not exist as individual molecules. Instead, they form crystal lattices, which are repeating three-dimensional arrangements of ions.
In these lattices:
- Each positive ion is surrounded by negative ions.
- Each negative ion is surrounded by positive ions.
This arrangement maximizes attractive forces and minimizes repulsive forces.
Lattice Energy
Lattice energy is the energy released when ions combine to form an ionic crystal.
Higher lattice energy means stronger ionic bonds.
Factors affecting lattice energy include:
- Charge of ions
- Distance between ions
Ions with higher charges and smaller sizes produce stronger lattice energies.
5. Properties of Ionic Compounds



Ionic compounds exhibit several distinctive properties.
High Melting and Boiling Points
Ionic bonds are strong electrostatic attractions.
Large amounts of energy are required to break these bonds, resulting in high melting and boiling points.
Crystalline Structure
Ionic compounds form highly ordered crystal lattices.
These structures give ionic solids a characteristic geometric shape.
Electrical Conductivity
Solid ionic compounds do not conduct electricity because ions are fixed in place.
However, when molten or dissolved in water, ions become mobile and conduct electricity.
Brittleness
Ionic crystals are brittle.
When pressure is applied, layers of ions shift, causing like charges to align and repel each other, leading to fracture.
Solubility in Water
Many ionic compounds dissolve in polar solvents such as water.
Water molecules surround ions in a process called hydration, separating them from the lattice.
6. Factors Affecting Ionic Bond Strength
Several factors influence the strength of ionic bonds.
Charge of Ions
Greater charges produce stronger electrostatic attraction.
Example:
Mg²⁺ and O²⁻ form stronger ionic bonds than Na⁺ and Cl⁻.
Ionic Radius
Smaller ions can approach each other more closely, increasing electrostatic attraction.
Thus, smaller ions form stronger ionic bonds.
Lattice Structure
The arrangement of ions within the crystal lattice also affects bond strength.
More compact lattices produce stronger interactions.
7. Ionic Bonding vs Covalent Bonding

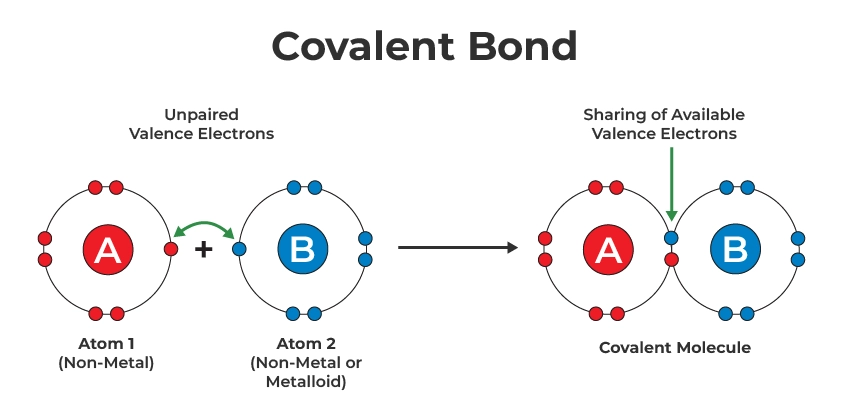


Although ionic and covalent bonds both hold atoms together, they differ significantly.
Ionic Bonding
- Electron transfer occurs.
- Forms ions.
- Usually occurs between metals and nonmetals.
- Forms crystal lattices.
Examples include NaCl and MgO.
Covalent Bonding
- Electrons are shared.
- Usually occurs between nonmetals.
- Forms discrete molecules.
Examples include water and carbon dioxide.
8. Polyatomic Ions in Ionic Compounds
Some ionic compounds contain polyatomic ions, which are groups of atoms bonded together that carry a charge.
Examples include:
Sulfate → SO₄²⁻
Nitrate → NO₃⁻
Ammonium → NH₄⁺
These ions behave as single units in ionic compounds.
Example compound:
Ammonium nitrate (NH₄NO₃)
Polyatomic ions are common in many chemical and biological systems.
9. Importance of Ionic Bonding in Nature
Ionic bonding plays a major role in many natural processes.
Examples include:
- Formation of minerals and rocks
- Electrolytes in biological systems
- Salt balance in living organisms
- Ocean chemistry
In the human body, ions such as sodium, potassium, and calcium are essential for nerve function, muscle contraction, and cellular processes.
10. Applications of Ionic Compounds
Ionic compounds are widely used in various industries and technologies.
Food Industry
Table salt (sodium chloride) is used for seasoning and food preservation.
Medicine
Electrolyte solutions help maintain fluid balance in the body.
Agriculture
Fertilizers often contain ionic compounds such as ammonium nitrate and potassium chloride.
Industrial Chemistry
Many industrial processes rely on ionic compounds for chemical synthesis.
Batteries
Certain batteries rely on the movement of ions to generate electrical energy.
11. Advanced Concepts in Ionic Bonding
More advanced models consider partial ionic character in bonds.
Some bonds may have both ionic and covalent characteristics depending on electronegativity differences.
Polarization effects also influence ionic bonding, especially when small cations interact with large anions.
These advanced concepts help explain complex chemical behavior in real compounds.
12. Conclusion
Ionic bonding is a fundamental type of chemical bonding that involves the transfer of electrons between atoms, leading to the formation of oppositely charged ions. The strong electrostatic attraction between these ions creates stable ionic compounds with distinctive physical and chemical properties.
Ionic compounds typically form crystal lattices, exhibit high melting points, conduct electricity in molten or dissolved states, and play essential roles in biological, geological, and industrial systems.
Understanding ionic bonding allows scientists to predict the properties of compounds, explain natural processes, and develop new technologies. From table salt in everyday life to minerals deep within the Earth, ionic bonding remains one of the most important interactions governing the behavior of matter.
