


1. Introduction to Chemical Kinetics
Chemical kinetics is the branch of physical chemistry that deals with the study of reaction rates and the mechanisms by which chemical reactions occur. While thermodynamics tells us whether a reaction is possible or spontaneous, chemical kinetics explains how fast a reaction proceeds and through what pathway it occurs.
A chemical reaction involves the transformation of reactants into products. However, different reactions occur at vastly different speeds. Some reactions, such as explosions or combustion, occur in fractions of a second, whereas others, like rusting of iron or geological transformations, may take years or centuries.
Chemical kinetics seeks to answer several important questions:
- How fast does a chemical reaction occur?
- What factors influence the speed of a reaction?
- What steps occur during the reaction process?
- What molecular events lead to product formation?
Understanding chemical kinetics is extremely important in many fields including:
- Industrial chemical production
- Pharmaceutical drug development
- Environmental chemistry
- Biochemistry and enzymatic reactions
- Materials science
- Atmospheric chemistry
Through kinetic studies, scientists can design efficient chemical processes, control reaction speeds, and optimize conditions for maximum yield.
2. Rate of Chemical Reaction
Definition of Reaction Rate
The rate of a chemical reaction is defined as the change in concentration of reactants or products per unit time.
Mathematically, the rate can be expressed as:
[
\text{Rate} = \frac{\text{Change in concentration}}{\text{Time}}
]
For a reaction:
[
A \rightarrow B
]
Rate can be written as:
[
\text{Rate} = -\frac{d[A]}{dt} = \frac{d[B]}{dt}
]
The negative sign indicates the decrease in reactant concentration.
Units of Reaction Rate
Common units include:
- mol L⁻¹ s⁻¹
- M s⁻¹
- mol dm⁻³ s⁻¹
Where:
- mol = amount of substance
- L = litre
- s = seconds
Average Rate vs Instantaneous Rate
Average Rate
Average rate is measured over a time interval.
[
\text{Average rate} = \frac{\Delta [A]}{\Delta t}
]
Instantaneous Rate
Instantaneous rate is the rate at a specific moment in time and is determined using derivatives.
[
\text{Instantaneous rate} = \frac{d[A]}{dt}
]
3. Rate Laws
The rate law expresses the relationship between the reaction rate and the concentration of reactants.
For a reaction:
[
aA + bB \rightarrow Products
]
The rate law is:
[
Rate = k[A]^m[B]^n
]
Where:
- k = rate constant
- [A], [B] = concentrations
- m, n = reaction orders
Order of Reaction
The order of a reaction indicates the power to which the concentration of a reactant is raised.
Types of Reaction Orders
- Zero-order reaction
- First-order reaction
- Second-order reaction
- Fractional order reaction
- Mixed-order reaction
Total order:
[
\text{Order} = m + n
]
4. Zero-Order Reactions
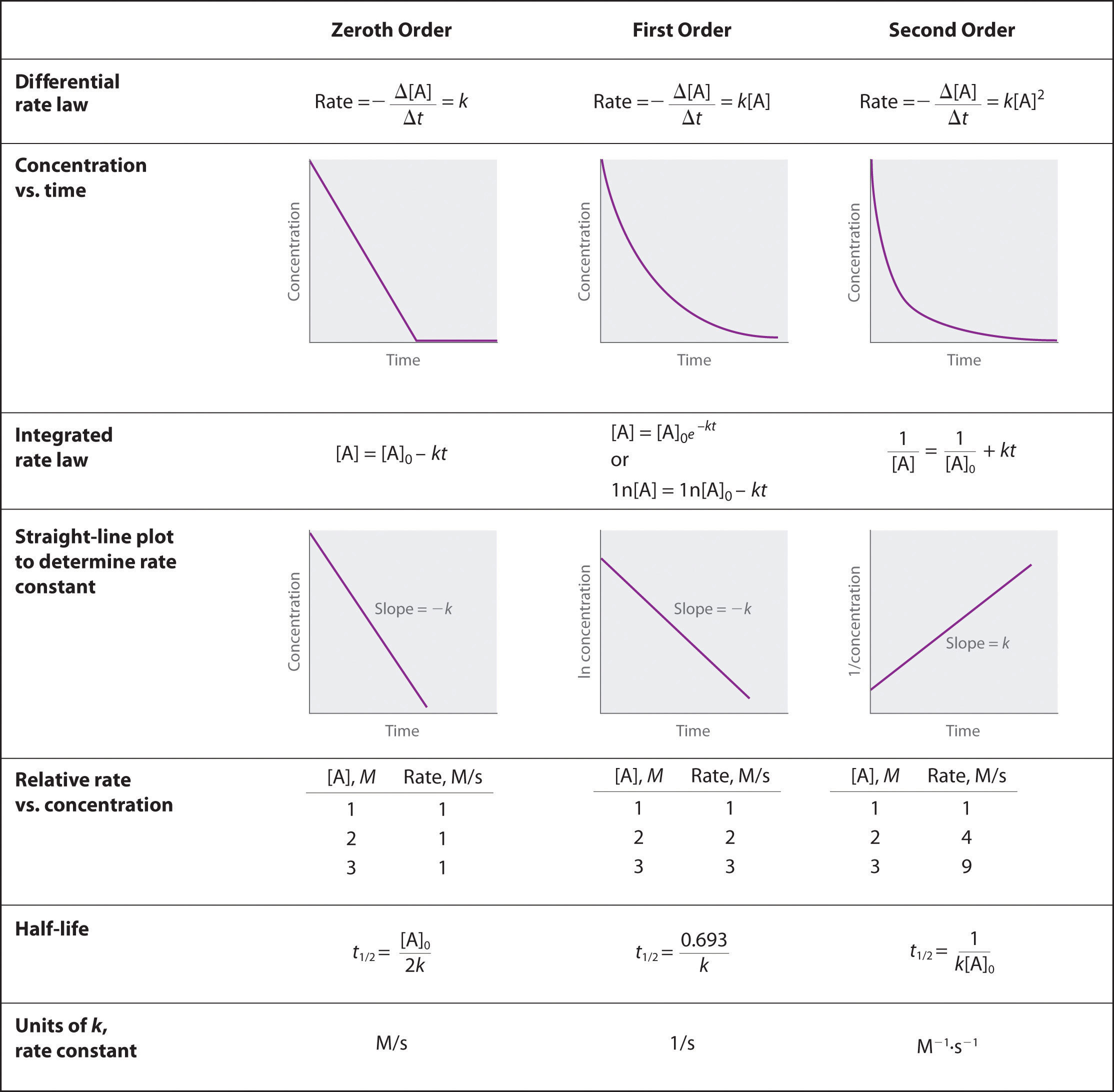
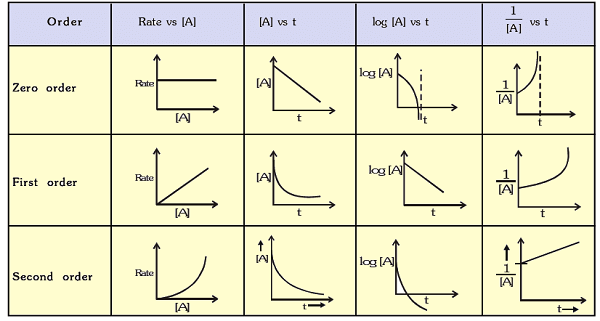
In zero-order reactions, the rate is independent of reactant concentration.
Rate law:
[
Rate = k
]
Integrated form:
[
[A] = [A]_0 – kt
]
Where:
- ([A]_0) = initial concentration
- (k) = rate constant
- (t) = time
Characteristics
- Rate is constant.
- Graph of concentration vs time is linear.
- Half-life depends on initial concentration.
Half-life:
[
t_{1/2} = \frac{[A]_0}{2k}
]
Examples
- Photochemical reactions
- Surface catalyzed reactions
- Decomposition on metal surfaces
5. First-Order Reactions



In first-order reactions, the rate is proportional to the concentration of one reactant.
Rate law:
[
Rate = k[A]
]
Integrated equation:
[
\ln[A] = \ln[A]_0 – kt
]
Alternate form:
[
[A] = [A]_0 e^{-kt}
]
Half-life
[
t_{1/2} = \frac{0.693}{k}
]
Key feature: Half-life is independent of initial concentration.
Examples
- Radioactive decay
- Decomposition of hydrogen peroxide
- Conversion of N₂O₅ to NO₂ and O₂
6. Second-Order Reactions



Rate law:
[
Rate = k[A]^2
]
Integrated equation:
[
\frac{1}{[A]} = \frac{1}{[A]_0} + kt
]
Half-life
[
t_{1/2} = \frac{1}{k[A]_0}
]
Characteristics
- Half-life depends on initial concentration.
- Graph of (1/[A]) vs time is linear.
Examples
- Dimerization reactions
- Certain bimolecular reactions
7. Factors Affecting Reaction Rate




Several factors influence reaction rates.
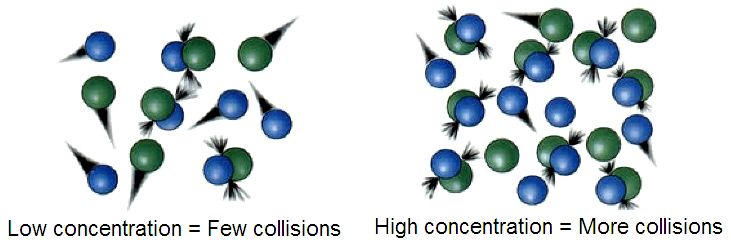
1. Concentration
Increasing reactant concentration increases collision frequency, which increases reaction rate.
2. Temperature
Higher temperature increases molecular kinetic energy, resulting in more effective collisions.
Typically:
Rate doubles for every 10°C increase.
3. Catalysts
Catalysts increase reaction rate by lowering activation energy.
Characteristics:
- Not consumed in reaction
- Provide alternative reaction pathway
- Increase efficiency
Examples:
- Platinum in hydrogenation
- Enzymes in biological reactions
4. Surface Area
In heterogeneous reactions, larger surface area increases reaction rate.
Example:
Powdered calcium carbonate reacts faster than solid marble.
5. Pressure
Important in gaseous reactions.
Increasing pressure increases concentration of gases.
6. Nature of Reactants
Some substances react faster than others depending on bond strength and molecular structure.
8. Collision Theory

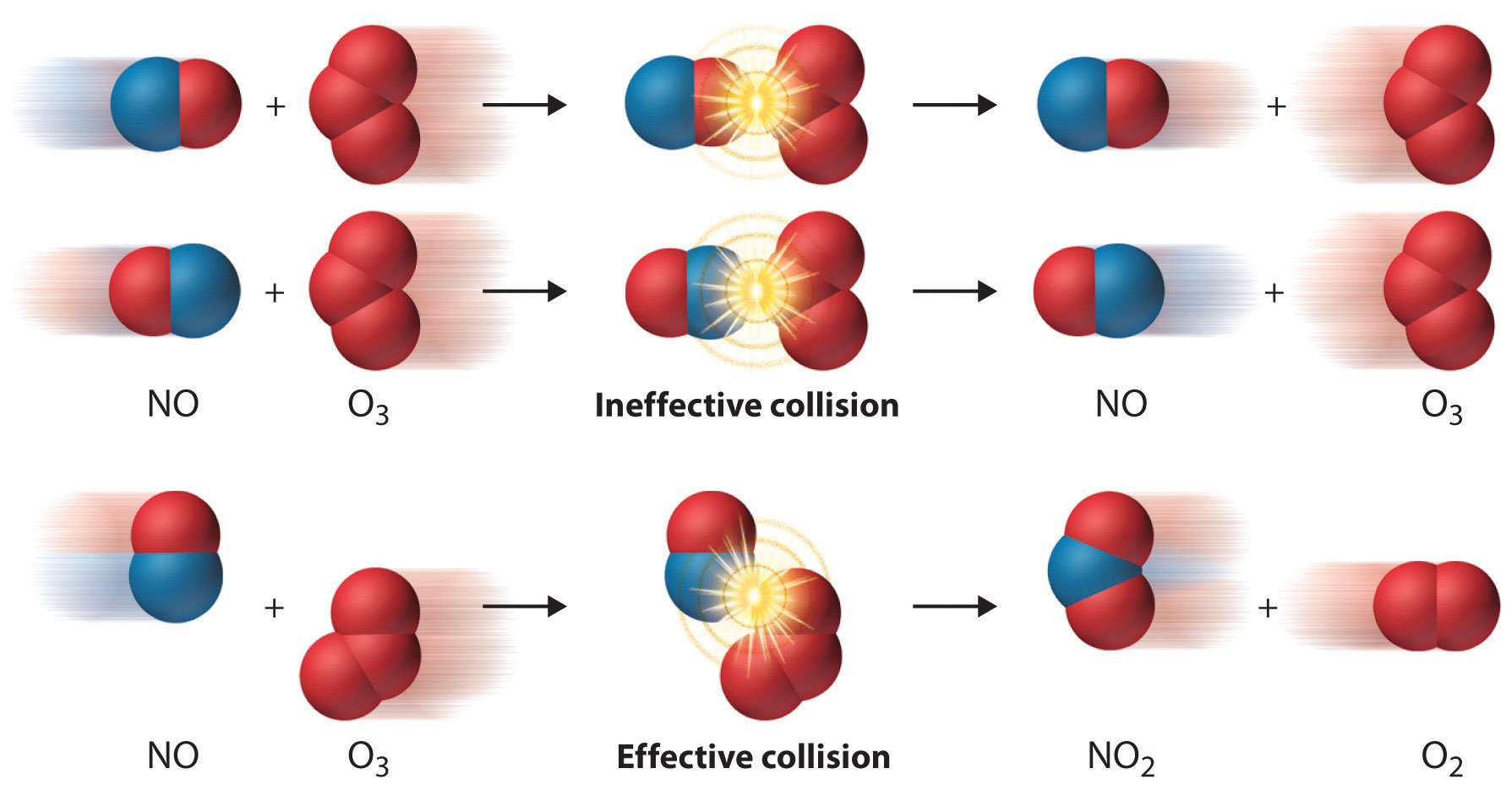


Collision theory explains how chemical reactions occur.
According to this theory:
- Molecules must collide to react.
- Collisions must have sufficient energy.
- Molecules must have correct orientation.
Effective Collisions
Only collisions meeting these conditions produce reactions.
9. Activation Energy
Activation energy is the minimum energy required for a reaction to occur.
Energy profile diagram:
- Reactants
- Transition state
- Products
Catalysts reduce activation energy.
10. Arrhenius Equation
The Arrhenius equation relates temperature and reaction rate.
[
k = Ae^{-E_a/RT}
]
Where:
- (k) = rate constant
- (A) = frequency factor
- (E_a) = activation energy
- (R) = gas constant
- (T) = temperature
Linear form:
[
\ln k = \ln A – \frac{E_a}{RT}
]
Arrhenius plots help determine activation energy.
11. Reaction Mechanisms
A reaction mechanism describes the step-by-step sequence of elementary reactions.
Example mechanism:
Step 1: Slow step
Step 2: Fast step
The slow step is called the rate-determining step.
12. Intermediate Species
Intermediates are species formed in one step and consumed in another.
Examples:
- Free radicals
- Carbocations
- Carbanions
They are unstable and short-lived.
13. Catalysis

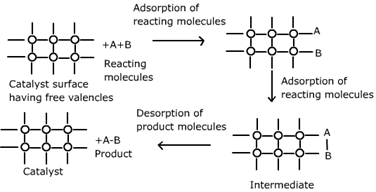


Catalysis is the acceleration of a chemical reaction using a catalyst.
Types:
Homogeneous Catalysis
Catalyst and reactants in same phase.
Example:
Acid-catalyzed ester hydrolysis.
Heterogeneous Catalysis
Catalyst in different phase.
Example:
Hydrogenation using metal catalysts.
Enzyme Catalysis
Biological catalysts.
Example:
Digestive enzymes.
14. Enzyme Kinetics
Enzymes are biological catalysts.
Reaction scheme:
[
E + S \rightarrow ES \rightarrow E + P
]
Where:
- E = enzyme
- S = substrate
- ES = enzyme-substrate complex
- P = product
Michaelis-Menten equation:
[
v = \frac{V_{max}[S]}{K_m + [S]}
]
15. Photochemical Reactions
These reactions occur due to absorption of light energy.
Example:
Photosynthesis
Ozone formation
Characteristics:
- Initiated by photons
- Often involve radicals
16. Chain Reactions
Chain reactions involve several repeating steps.
Steps:
- Initiation
- Propagation
- Termination
Example:
Chlorination of methane.
17. Steady State Approximation
Used for complex reactions.
Assumes concentration of intermediates remains constant.
18. Chemical Kinetics in Industry
Chemical kinetics is essential in industrial processes:
Examples:
- Haber process
- Petroleum refining
- Polymer manufacturing
- Pharmaceutical synthesis
Optimizing reaction rate improves:
- Production efficiency
- Energy use
- Product yield
19. Applications of Chemical Kinetics
Applications include:
- Drug development
- Environmental pollution control
- Food preservation
- Material synthesis
- Battery technology
- Atmospheric chemistry
20. Importance of Chemical Kinetics
Chemical kinetics helps scientists:
- Understand reaction mechanisms
- Predict reaction behavior
- Design catalysts
- Control industrial processes
- Develop sustainable chemical technologies
Conclusion
Chemical kinetics is a fundamental branch of chemistry that explains how and why chemical reactions occur at particular speeds. By studying reaction rates, rate laws, mechanisms, and energy changes, chemists gain insight into the microscopic processes that govern chemical transformations. From industrial production to biological systems, the principles of chemical kinetics play a vital role in modern science and technology.
