



1. Introduction to Covalent Bonding
Covalent bonding is one of the fundamental types of chemical bonding that occurs when atoms share pairs of electrons. This type of bonding is most commonly observed between nonmetal atoms, which have similar electronegativities and prefer to share electrons rather than transfer them completely.
In covalent bonding, two atoms overlap their atomic orbitals and share electrons in order to achieve a more stable electronic configuration. By sharing electrons, atoms can complete their outer electron shells and achieve a configuration similar to that of noble gases.
Covalent bonds are responsible for forming a vast range of substances, from simple molecules such as hydrogen and oxygen to complex organic molecules like proteins, DNA, and polymers. Most compounds in biological systems are formed through covalent bonding.
Understanding covalent bonding is crucial for explaining:
- Molecular structures
- Chemical reactivity
- Properties of organic and inorganic compounds
- Molecular geometry
- Biological macromolecules
- Chemical synthesis
Unlike ionic compounds, which form crystal lattices, covalent compounds usually form discrete molecules. These molecules are held together internally by strong covalent bonds and externally by weaker intermolecular forces.
The study of covalent bonding also forms the basis of many advanced areas of chemistry, including organic chemistry, biochemistry, molecular biology, and materials science.
2. Formation of Covalent Bonds



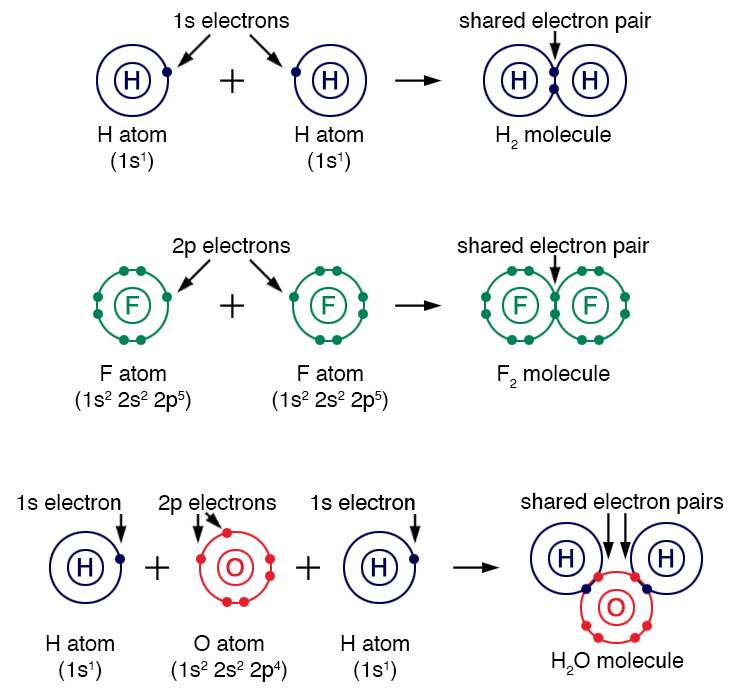
Covalent bonds form when two atoms share one or more pairs of electrons. This sharing allows each atom to achieve a stable electron configuration.
Atoms form covalent bonds when the difference in electronegativity between them is relatively small. Because neither atom strongly dominates the electron attraction, they share electrons rather than transfer them.
Example: Hydrogen Molecule
A hydrogen atom contains one electron in its outer shell. Two hydrogen atoms can share their electrons, forming a covalent bond and creating a hydrogen molecule (H₂).
Each hydrogen atom effectively has two electrons in its outer shell, satisfying the duet rule, which is a simplified version of the octet rule for hydrogen.
Orbital Overlap
Covalent bonding occurs when atomic orbitals overlap. The overlapping region contains shared electron pairs that attract both nuclei, holding the atoms together.
Orbital overlap increases the probability of finding shared electrons between the nuclei, which stabilizes the molecule.
3. The Octet Rule in Covalent Bonding




The octet rule states that atoms tend to form bonds until they are surrounded by eight electrons in their outermost shell.
This rule explains why atoms share electrons in covalent bonds.
Example:
Oxygen has six valence electrons. By sharing two electrons with another oxygen atom, each atom achieves eight electrons in its outer shell.
Although the octet rule works well for many elements, there are several exceptions:
- Hydrogen requires only two electrons.
- Some molecules have expanded octets.
- Some molecules have incomplete octets.
Despite these exceptions, the octet rule remains a useful guideline for understanding covalent bonding.
4. Types of Covalent Bonds




Covalent bonds can be classified based on the number of shared electron pairs.
Single Covalent Bonds
A single covalent bond involves the sharing of one pair of electrons.
Example:
Hydrogen molecule (H₂)
These bonds are relatively long and weaker compared to multiple bonds.
Double Covalent Bonds
A double bond involves the sharing of two pairs of electrons.
Example:
Oxygen molecule (O₂)
Double bonds are shorter and stronger than single bonds.
Triple Covalent Bonds
A triple bond involves three pairs of shared electrons.
Example:
Nitrogen molecule (N₂)
Triple bonds are the strongest and shortest covalent bonds.
5. Polar and Nonpolar Covalent Bonds




Covalent bonds can also be classified based on how electrons are shared.
Nonpolar Covalent Bonds
In nonpolar covalent bonds, electrons are shared equally between atoms.
This occurs when atoms have similar electronegativity values.
Example:
Hydrogen molecule (H₂)
Because electrons are shared equally, no partial charges develop.
Polar Covalent Bonds
In polar covalent bonds, electrons are shared unequally.
The atom with higher electronegativity attracts electrons more strongly.
Example:
Water (H₂O)
This unequal sharing produces partial positive and negative charges, creating a dipole.
Polar covalent bonds are responsible for many important chemical and biological properties.
6. Lewis Structures




Lewis structures are diagrams that represent the arrangement of valence electrons in molecules.
They show:
- Bonding electron pairs
- Lone pairs of electrons
Lewis structures help predict molecular shape and bonding patterns.
Steps for drawing Lewis structures include:
- Count total valence electrons.
- Arrange atoms around the central atom.
- Form single bonds between atoms.
- Distribute remaining electrons to satisfy the octet rule.
- Form multiple bonds if necessary.
Lewis structures are essential tools in understanding molecular structure.
7. Molecular Geometry and VSEPR Theory




The VSEPR theory (Valence Shell Electron Pair Repulsion theory) explains the shapes of molecules based on electron pair repulsion.
Electron pairs around a central atom repel each other and arrange themselves to minimize repulsion.
Common molecular shapes include:
Linear
Trigonal planar
Tetrahedral
Trigonal pyramidal
Bent
Molecular geometry influences physical properties such as polarity and reactivity.
8. Bond Length and Bond Energy
Bond length is the distance between the nuclei of two bonded atoms.
Bond energy is the amount of energy required to break a bond.
General relationships include:
- Shorter bonds are stronger.
- Triple bonds are stronger than double bonds.
- Double bonds are stronger than single bonds.
Bond energy influences chemical reaction rates and molecular stability.
9. Covalent Network Solids




Some substances form extended networks of covalent bonds rather than discrete molecules.
Examples include:
Diamond
Graphite
Silicon dioxide
These materials form covalent network solids with extremely strong bonds throughout the structure.
Properties include:
- Very high melting points
- Exceptional hardness
- Poor electrical conductivity (except graphite)
Diamond is one of the hardest known natural substances due to its strong covalent network.
10. Importance of Covalent Bonding
Covalent bonding is essential for many chemical and biological systems.
It explains:
- Structure of organic molecules
- Formation of biological macromolecules
- Chemical reactions in living organisms
- Material properties in polymers and plastics
Most compounds found in living organisms are covalent molecules.
Examples include:
Proteins
DNA
Carbohydrates
Lipids
Covalent bonding therefore plays a crucial role in life processes.
11. Applications in Modern Science
Covalent bonding is important in many technological fields.
Organic Chemistry
Most organic compounds contain covalent bonds.
Polymer Science
Plastics and synthetic fibers are formed through covalent bonding.
Pharmaceuticals
Drug molecules interact with biological targets through covalent and noncovalent interactions.
Nanotechnology
Carbon nanotubes and graphene involve covalent bonding networks.
Materials Science
Covalent bonding determines the strength and stability of many advanced materials.
12. Conclusion
Covalent bonding is one of the most important mechanisms by which atoms combine to form molecules. By sharing electrons, atoms achieve stable electronic configurations and create a vast array of chemical substances.
Covalent bonds vary in strength and character depending on the number of shared electrons and differences in electronegativity. These bonds determine molecular structure, polarity, and chemical reactivity.
From simple molecules such as hydrogen to complex biological macromolecules like DNA, covalent bonding underlies much of the chemistry of life and matter. Understanding covalent bonding is therefore essential for studying chemistry, biology, materials science, and many modern technologies.
