1. Introduction to Catalysis
Catalysis is one of the most important concepts in chemistry, particularly in chemical kinetics and industrial chemistry. It refers to the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst. The catalyst participates in the reaction but is not consumed permanently and can be used repeatedly.
Many chemical reactions occur extremely slowly under normal conditions. Without catalysis, several industrial processes would be impractically slow or require extremely high temperatures and pressures. Catalysts allow these reactions to proceed faster and often under milder conditions.
Catalysis plays a vital role in many areas, including:
- Industrial chemical manufacturing
- Petroleum refining
- Environmental protection
- Pharmaceutical synthesis
- Biological systems
- Energy production
More than 90% of industrial chemical processes involve catalysts. Catalysis is therefore considered one of the foundations of modern chemical technology.
2. Definition of Catalysis
Catalysis is defined as:
The process in which a substance called a catalyst increases the rate of a chemical reaction without undergoing permanent chemical change.
The substance that accelerates the reaction is called a catalyst.
Example:
Hydrogen peroxide decomposes slowly:
2H₂O₂ → 2H₂O + O₂
When manganese dioxide (MnO₂) is added, the reaction becomes much faster. MnO₂ acts as a catalyst.
3. Characteristics of Catalysts
Catalysts possess several important properties.
1. Increase Reaction Rate
Catalysts speed up chemical reactions by providing an alternative reaction pathway with lower activation energy.
2. Not Consumed in Reaction
A catalyst remains chemically unchanged after the reaction.
3. Small Amount Required
Only a small quantity of catalyst is needed to significantly increase reaction rate.
4. High Specificity
Many catalysts are highly selective and promote only specific reactions.
5. Reusable
Catalysts can be used repeatedly in chemical processes.
6. Do Not Affect Reaction Equilibrium
Catalysts increase both forward and reverse reaction rates equally.
4. Energy Profile of Catalyzed Reactions



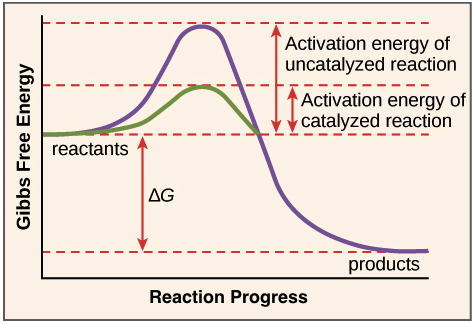
Chemical reactions require energy to initiate. This energy is called activation energy.
Activation energy represents the minimum energy required for reactant molecules to reach the transition state.
Without catalyst:
- Activation energy is high
- Reaction occurs slowly
With catalyst:
- Activation energy decreases
- Reaction becomes faster
The catalyst creates an alternative reaction pathway with lower energy requirements.
However, catalysts do not change:
- Energy of reactants
- Energy of products
- Overall enthalpy change of the reaction
5. Types of Catalysis
Catalysis is broadly classified into several categories depending on the phase of catalyst and reactants.
Major types include:
- Homogeneous catalysis
- Heterogeneous catalysis
- Enzyme catalysis
- Autocatalysis
- Positive catalysis
- Negative catalysis
6. Homogeneous Catalysis



Homogeneous catalysis occurs when catalyst and reactants are present in the same phase, usually in solution.
Because all substances are in the same phase, reactions occur uniformly throughout the mixture.
Example
Acid catalysis of ester hydrolysis:
Ester + H₂O → Acid + Alcohol
Hydrogen ions act as catalysts.
Advantages
- High selectivity
- Uniform mixing
- Easy control of reaction conditions
Disadvantages
- Difficult catalyst separation
- Catalyst recovery may be expensive
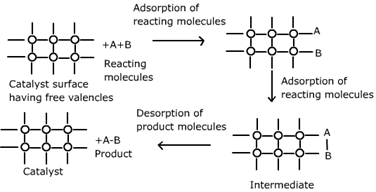
7. Heterogeneous Catalysis


In heterogeneous catalysis, the catalyst and reactants exist in different phases.
Most industrial catalysts are heterogeneous.
Example:
Hydrogenation of vegetable oils using nickel catalyst.
Here:
- Reactants = liquid or gas
- Catalyst = solid metal
Steps in Heterogeneous Catalysis
- Adsorption of reactants onto catalyst surface
- Reaction occurs on surface
- Products desorb from catalyst
Advantages
- Easy catalyst separation
- Catalyst can be reused
- Suitable for industrial processes
Examples
- Haber process (iron catalyst)
- Contact process (vanadium pentoxide)
- Catalytic converters
8. Enzyme Catalysis




Enzymes are biological catalysts found in living organisms.
They are typically proteins that accelerate biochemical reactions.
Example:
Digestive enzymes break down food molecules.
Key Features of Enzyme Catalysis
- Extremely high efficiency
- Highly specific
- Work under mild conditions
- Regulated by biological systems
Enzyme Mechanism
- Substrate binds to enzyme active site
- Enzyme-substrate complex forms
- Chemical reaction occurs
- Products released
Models of Enzyme Action
Lock and Key Model
The enzyme active site perfectly matches the substrate.
Induced Fit Model
The enzyme changes shape when substrate binds.
9. Autocatalysis
Autocatalysis occurs when one of the reaction products acts as a catalyst for the reaction itself.
As the reaction proceeds, the rate increases because more catalyst is produced.
Example:
Certain oxidation reactions.
Characteristics:
- Slow initial rate
- Rapid increase in rate later
10. Positive and Negative Catalysis
Positive Catalysis
Catalysts that increase reaction rate.
Example:
Platinum in hydrogenation.
Negative Catalysis
Substances that decrease reaction rate are called inhibitors.
Example:
Preservatives that slow food spoilage.
11. Catalytic Mechanisms
Catalytic reactions occur through multi-step mechanisms.
These involve:
- Intermediate formation
- Transition states
- Surface interactions
Catalysts may:
- Break bonds
- Form temporary complexes
- Stabilize transition states
12. Catalytic Poisoning
Catalytic poisoning occurs when a substance deactivates a catalyst.
Poison molecules bind strongly to catalyst surface and block active sites.
Example:
Lead poisoning platinum catalysts in car exhaust systems.
13. Promoters in Catalysis
Promoters are substances that enhance catalytic activity.
They do not act as catalysts themselves but improve catalyst efficiency.
Example:
Potassium oxide in Haber process catalysts.
14. Industrial Catalysis




Catalysis is essential for large-scale chemical manufacturing.
Major industrial catalytic processes include:
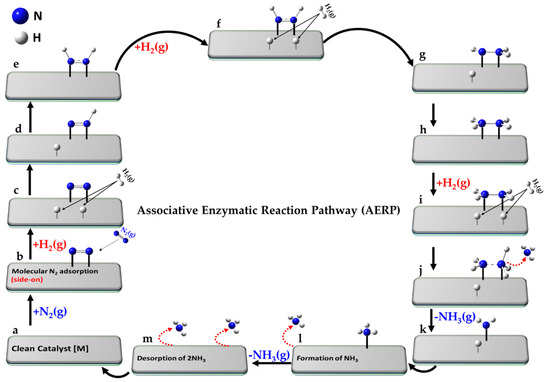
Haber Process
Production of ammonia:
N₂ + 3H₂ → 2NH₃
Catalyst: Iron
Contact Process
Production of sulfuric acid.
Catalyst: Vanadium pentoxide.
Catalytic Cracking
Used in petroleum refining to break large hydrocarbons.
Catalyst: Zeolites.
Hydrogenation
Used in food industry.
Catalyst: Nickel.
15. Environmental Catalysis
Catalysts are widely used to reduce pollution.
Catalytic Converters
Installed in automobiles.
They convert toxic gases into less harmful substances.
Reactions include:
- CO → CO₂
- NOₓ → N₂
- Hydrocarbons → CO₂ + H₂O
Catalysts used:
- Platinum
- Palladium
- Rhodium
16. Nanocatalysis
Modern research focuses on nanocatalysts.
Nanoparticles have:
- Large surface area
- High catalytic activity
- Improved efficiency
Applications include:
- Fuel cells
- Green chemistry
- Renewable energy
17. Catalysis in Green Chemistry
Green chemistry aims to minimize environmental impact.
Catalysts help by:
- Reducing energy consumption
- Minimizing waste
- Improving efficiency
Examples:
Biocatalysis and photocatalysis.
18. Photocatalysis
Photocatalysis involves catalysts activated by light.
Example:
Titanium dioxide used in:
- Water purification
- Air purification
- Self-cleaning surfaces
19. Electrocatalysis
Electrocatalysts accelerate electrochemical reactions.
Used in:
- Batteries
- Fuel cells
- Hydrogen production
Example:
Platinum catalysts in hydrogen fuel cells.
20. Importance of Catalysis
Catalysis has enormous scientific and industrial importance.
Benefits include:
- Faster chemical reactions
- Lower energy requirements
- Reduced industrial costs
- Improved product selectivity
- Environmental protection
Catalysts are critical for sustainable chemistry and energy technologies.
Conclusion
Catalysis is a cornerstone of modern chemistry and chemical engineering. By lowering activation energy and providing alternative reaction pathways, catalysts dramatically accelerate chemical reactions without being consumed. Catalysis occurs in many forms, including homogeneous, heterogeneous, enzyme, and photocatalysis. These processes are essential in industrial manufacturing, environmental protection, biochemical systems, and energy technologies. As scientific research advances, new catalytic materials such as nanocatalysts and electrocatalysts are being developed to create more efficient and sustainable chemical processes for the future.