



1. Introduction to Atomic Models
Atomic models are conceptual representations used to explain the structure and behavior of atoms. Because atoms are extremely small and cannot be observed directly with the naked eye, scientists use models to describe their internal structure and explain experimental observations.
Over the centuries, scientific understanding of atoms has evolved through a series of discoveries and experiments. Each new model improved upon earlier ones by explaining additional experimental evidence. Atomic models help scientists understand:
- The arrangement of subatomic particles
- Chemical bonding and reactions
- The periodic table of elements
- Spectral lines of atoms
- The behavior of matter at microscopic levels
The development of atomic models represents one of the most important progressions in the history of science. From early philosophical ideas to the modern quantum mechanical model, each step has contributed to a deeper understanding of matter.
Atomic models are not exact pictures of atoms but rather scientific frameworks that approximate atomic structure based on available evidence.
2. Early Philosophical Concepts of Atoms



The earliest ideas about atoms originated in ancient Greece around 400 BCE. Philosophers such as Democritus and Leucippus proposed that matter consisted of tiny indivisible particles called atoms, derived from the Greek word meaning “uncuttable.”
According to their theory:
- Matter is composed of extremely small particles.
- These particles cannot be divided further.
- Different substances are formed by different arrangements of atoms.
Although this concept was remarkably insightful, it was based on philosophical reasoning rather than experimental evidence. Therefore, it did not gain widespread acceptance in scientific circles at the time.
For many centuries afterward, the dominant view of matter was based on the four classical elements proposed by Aristotle:
- Earth
- Water
- Air
- Fire
It was not until the development of modern experimental science in the eighteenth and nineteenth centuries that atomic theory began to gain scientific support.
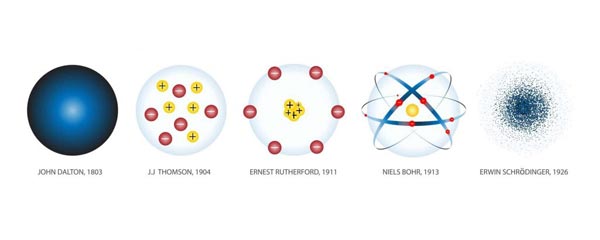
3. Dalton’s Atomic Model (Solid Sphere Model)




The first scientific atomic model was proposed by John Dalton in the early nineteenth century.
Dalton’s model was based on experimental observations related to chemical reactions and the laws of chemical combination.
Dalton’s Atomic Theory
Dalton proposed several fundamental principles:
- Matter is composed of tiny indivisible particles called atoms.
- Atoms of a given element are identical in mass and properties.
- Atoms of different elements differ in mass and properties.
- Compounds form when atoms combine in fixed whole-number ratios.
- Chemical reactions involve the rearrangement of atoms.
Characteristics of Dalton’s Model
Dalton imagined atoms as solid, indivisible spheres, similar to tiny billiard balls.
This model successfully explained several chemical laws, including:
- Law of conservation of mass
- Law of definite proportions
- Law of multiple proportions
Limitations of Dalton’s Model
Although Dalton’s theory was revolutionary, later discoveries revealed several limitations:
- Atoms are divisible and contain subatomic particles.
- Atoms of the same element can have different masses (isotopes).
- Atoms can undergo nuclear reactions.
Despite these limitations, Dalton’s model laid the foundation for modern atomic theory.
4. Thomson’s Atomic Model (Plum Pudding Model)




The discovery of the electron in 1897 by J. J. Thomson led to a new atomic model.
Through experiments with cathode ray tubes, Thomson demonstrated that atoms contain negatively charged particles.
Thomson’s Model Description
Thomson proposed the plum pudding model of the atom.
In this model:
- The atom is a positively charged sphere.
- Electrons are embedded throughout the sphere like raisins in a pudding.
This arrangement ensured that the atom remained electrically neutral.
Contributions of Thomson’s Model
The model introduced the concept of subatomic particles, proving that atoms were not indivisible.
It also explained the electrical properties of atoms.
Limitations
Thomson’s model could not explain:
- The existence of a dense nucleus
- The scattering of alpha particles observed in later experiments
These limitations led to the development of a new atomic model.
5. Rutherford’s Nuclear Model


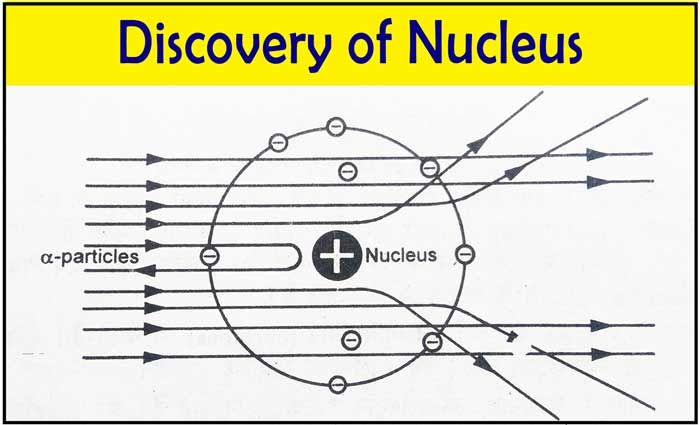

In 1911, Ernest Rutherford conducted the famous gold foil experiment, which revolutionized the understanding of atomic structure.
Gold Foil Experiment
Rutherford and his colleagues directed a beam of alpha particles at a thin sheet of gold foil.
Observations:
- Most alpha particles passed straight through the foil.
- Some particles were deflected slightly.
- A few particles bounced back at large angles.
Conclusions
From these observations, Rutherford concluded that:
- Atoms are mostly empty space.
- Positive charge is concentrated in a tiny central nucleus.
- Electrons move around the nucleus.
Rutherford’s Atomic Model
The atom consists of:
- A small, dense, positively charged nucleus
- Electrons orbiting the nucleus
- Large empty space between the nucleus and electrons
Limitations
Rutherford’s model could not explain:
- Why electrons do not spiral into the nucleus
- The discrete spectral lines observed in atomic emission spectra
These problems led to the development of the Bohr model.
6. Bohr’s Atomic Model




In 1913, Niels Bohr proposed an improved atomic model that incorporated ideas from quantum theory.
Main Features of Bohr’s Model
- Electrons move in fixed circular orbits around the nucleus.
- Each orbit corresponds to a specific energy level.
- Electrons can jump between energy levels by absorbing or emitting energy.
- Energy is quantized and exists in discrete units.
Explanation of Atomic Spectra
Bohr’s model successfully explained the spectral lines of hydrogen.
When electrons jump from higher energy levels to lower levels, they emit light of specific wavelengths.
Advantages
Bohr’s model explained:
- Atomic emission spectra
- Stability of atoms
- Quantized energy levels
Limitations
Bohr’s model works well only for hydrogen and hydrogen-like atoms.
It cannot accurately describe atoms with multiple electrons.
7. Quantum Mechanical Model of the Atom



The modern understanding of atomic structure is based on quantum mechanics.
This model was developed through the work of scientists such as:
- Erwin Schrödinger
- Werner Heisenberg
- Louis de Broglie
Key Principles
Wave-Particle Duality
Electrons behave both as particles and waves.
Uncertainty Principle
The exact position and momentum of an electron cannot be known simultaneously.
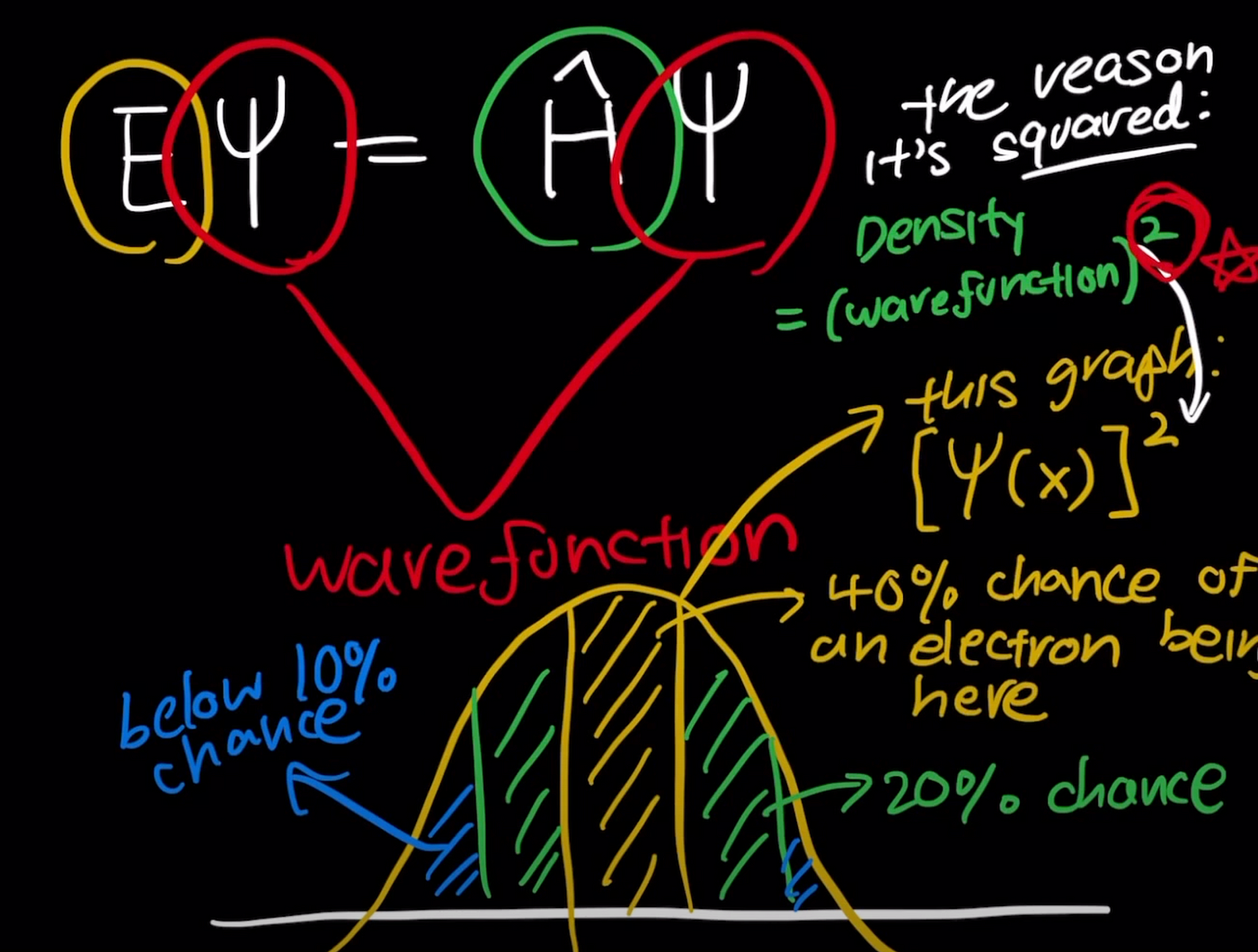
Schrödinger Wave Equation
This equation describes the probability distribution of electrons in atoms.
Orbitals
Instead of fixed orbits, electrons occupy orbitals, which are regions where electrons are most likely to be found.
Types of orbitals include:
- s orbitals (spherical)
- p orbitals (dumbbell-shaped)
- d orbitals (clover-shaped)
- f orbitals (complex shapes)
This model accurately explains atomic structure and chemical bonding.
8. Comparison of Atomic Models
Each atomic model improved upon the previous one.
Dalton Model
Atoms are indivisible solid spheres.
Thomson Model
Atoms contain electrons embedded in positive charge.
Rutherford Model
Atoms contain a dense central nucleus.
Bohr Model
Electrons occupy quantized energy levels.
Quantum Mechanical Model
Electrons exist in probability clouds called orbitals.
This progression reflects the advancement of experimental techniques and scientific understanding.
9. Importance of Atomic Models
Atomic models are essential for understanding many aspects of chemistry and physics.
Chemical Bonding
Models explain how atoms combine to form molecules.
Periodic Table
Atomic structure determines periodic trends.
Spectroscopy
Atomic models explain emission and absorption spectra.
Material Science
Understanding electron behavior helps design new materials.
Nanotechnology
Atomic-level understanding enables manipulation of matter at extremely small scales.
10. Applications in Modern Science
Atomic models play a crucial role in many technological fields.
Electronics
Semiconductors rely on understanding electron energy levels.
Nuclear Energy
Nuclear models explain fission and fusion reactions.
Medical Technology
Radiation therapy and imaging technologies rely on atomic principles.
Astronomy
Spectral analysis helps identify elements in stars and galaxies.
11. Conclusion
The development of atomic models represents a remarkable journey in scientific discovery. Starting from philosophical ideas about indivisible particles, scientists gradually uncovered the complex internal structure of atoms.
Dalton’s model introduced the concept of atoms as fundamental building blocks of matter. Thomson’s discovery of electrons revealed that atoms contain subatomic particles. Rutherford’s experiments demonstrated the existence of a dense nucleus. Bohr introduced quantized electron energy levels, and the quantum mechanical model provided a sophisticated description of electron behavior.
Today, the quantum mechanical model remains the most accurate representation of atomic structure. It continues to guide research in chemistry, physics, materials science, and nanotechnology.
The evolution of atomic models highlights the importance of experimentation, theoretical insight, and technological advancement in expanding our understanding of the natural world.
