



1. Introduction to the Mole Concept
The mole concept is one of the most important ideas in chemistry because it provides a bridge between the microscopic world of atoms and molecules and the macroscopic world that we can measure in laboratories. Atoms and molecules are extremely small and cannot be counted directly using ordinary methods. Chemists therefore use the mole as a unit to count particles indirectly.
A mole is defined as the amount of substance that contains 6.022 × 10²³ elementary entities, such as atoms, molecules, ions, or electrons. This number is known as Avogadro’s number.
The mole allows chemists to perform calculations involving the quantities of substances involved in chemical reactions. By using the mole concept, chemists can convert between mass, number of particles, and volume of substances.
For example, if we know the mass of a chemical compound, we can calculate the number of molecules present using the mole concept. Similarly, we can determine how many atoms participate in a chemical reaction.
The mole concept is essential for understanding several important topics in chemistry, including:
- Chemical reactions and stoichiometry
- Chemical equations
- Molar mass calculations
- Gas laws
- Solution concentration
- Reaction yield
Without the mole concept, it would be nearly impossible to relate the microscopic properties of atoms and molecules to measurable quantities in the laboratory.
2. Historical Development of the Mole Concept



The mole concept developed from the work of several scientists studying gases and atomic theory.
One of the key contributors was Amedeo Avogadro, an Italian scientist who proposed Avogadro’s hypothesis in 1811.
Avogadro stated that:
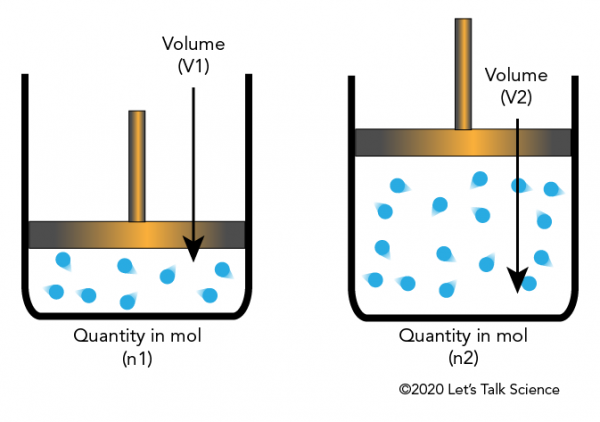
Equal volumes of gases at the same temperature and pressure contain the same number of molecules.
Although his hypothesis was initially ignored, later scientists recognized its importance. It eventually became a cornerstone of molecular theory.
The number of particles in a mole was later determined experimentally and named Avogadro’s number in his honor.
This constant provides the basis for converting between atomic-scale quantities and measurable macroscopic amounts of matter.
3. Definition of a Mole




A mole is the standard unit used in chemistry for measuring the amount of substance.
One mole of any substance contains 6.022 × 10²³ particles.
These particles can be:
- Atoms
- Molecules
- Ions
- Electrons
- Other elementary entities
Examples:
1 mole of carbon atoms contains 6.022 × 10²³ carbon atoms.
1 mole of water molecules contains 6.022 × 10²³ water molecules.
The mole functions similarly to other counting units such as a dozen.
For example:
1 dozen = 12 objects
1 mole = 6.022 × 10²³ particles
However, the mole represents an extremely large number because atoms and molecules are extremely small.
4. Avogadro’s Number

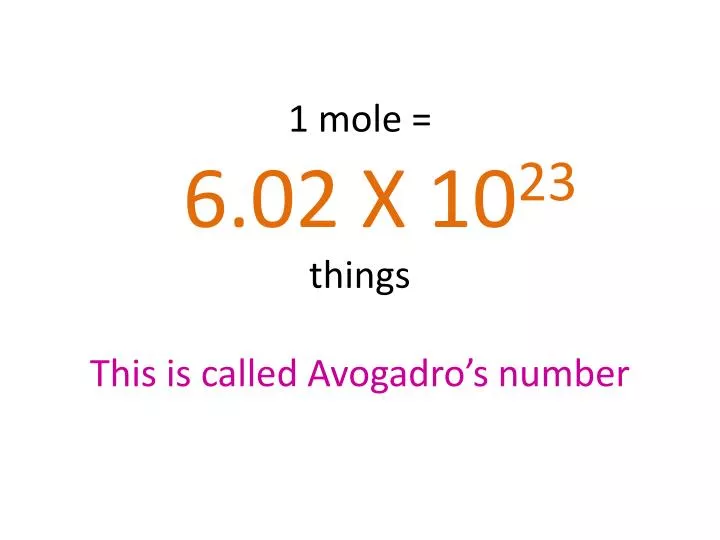


Avogadro’s number is the number of particles present in one mole of a substance.
Avogadro’s number = 6.022 × 10²³
This number represents an enormous quantity of particles.
To understand its magnitude, consider the following analogy:
If 6.022 × 10²³ grains of sand were spread across Earth, they would cover the planet in a layer several kilometers thick.
Avogadro’s number allows chemists to convert between:
- Number of particles
- Amount of substance in moles
For example:
Number of moles = Number of particles ÷ Avogadro’s number
This relationship is essential for many chemical calculations.
5. Molar Mass




Molar mass is the mass of one mole of a substance.
It is usually expressed in grams per mole (g/mol).
The molar mass of an element is numerically equal to its atomic mass from the periodic table.
Examples:
Carbon → 12 g/mol
Oxygen → 16 g/mol
Hydrogen → 1 g/mol
For compounds, molar mass is calculated by adding the atomic masses of all atoms in the molecule.
Example: Water (H₂O)
Hydrogen: 1 × 2 = 2
Oxygen: 16 × 1 = 16
Total molar mass = 18 g/mol
This means that one mole of water molecules weighs 18 grams.
6. Mole-Mass Relationship



The mole concept allows conversion between mass and moles.
Formula:
Moles = Mass ÷ Molar Mass
Example:
If we have 36 g of water:
Moles of water = 36 ÷ 18 = 2 moles
This relationship is fundamental for chemical calculations and stoichiometry.
7. Mole-Particle Relationship




The mole concept also allows conversion between moles and particles.
Formula:
Number of particles = Moles × Avogadro’s number
Example:
2 moles of oxygen molecules contain:
2 × 6.022 × 10²³ molecules
This equals:
1.2044 × 10²⁴ molecules
This calculation allows chemists to determine the number of atoms or molecules present in a sample.
8. Mole and Gas Volume




For gases, the mole concept is related to volume.
At standard temperature and pressure (STP):
1 mole of any gas occupies 22.4 liters.
This is called the molar volume of a gas.
Example:
1 mole of oxygen gas occupies 22.4 L at STP.
This relationship allows chemists to calculate the volume of gases involved in reactions.
9. Mole Concept in Chemical Reactions




The mole concept is essential for understanding stoichiometry, which deals with quantitative relationships in chemical reactions.
Balanced chemical equations represent ratios of moles of reactants and products.
Example reaction:
2H₂ + O₂ → 2H₂O
This equation means:
2 moles of hydrogen react with 1 mole of oxygen to produce 2 moles of water.
Using these mole ratios, chemists can calculate:
- Required reactant quantities
- Amount of product formed
- Limiting reagents
10. Importance of the Mole Concept
The mole concept is central to many areas of chemistry.
It allows scientists to:
- Measure amounts of substances accurately
- Perform chemical calculations
- Predict reaction outcomes
- Determine chemical formulas
- Study reaction mechanisms
Without the mole concept, quantitative chemistry would not be possible.
11. Applications in Science and Industry
The mole concept has many practical applications.
Pharmaceutical Chemistry
Drug doses are determined based on precise quantities of molecules.
Chemical Manufacturing
Industrial reactions rely on mole calculations to produce desired amounts of products.
Environmental Science
Pollutant concentrations are measured using mole-based calculations.
Biochemistry
Molecular concentrations in cells are often expressed in moles.
Materials Science
The mole concept helps determine atomic ratios in materials.
12. Conclusion
The mole concept provides a fundamental link between the microscopic world of atoms and molecules and the macroscopic quantities that chemists measure in laboratories. By defining a mole as 6.022 × 10²³ particles, chemists can convert between mass, number of particles, and volume of substances.
The mole concept enables accurate calculations in chemical reactions, helps determine molecular composition, and plays a crucial role in stoichiometry and quantitative analysis.
From laboratory experiments to industrial manufacturing and biological research, the mole concept is an essential tool that underpins much of modern chemistry. Understanding this concept allows scientists to analyze and predict chemical behavior with remarkable precision.
