



1. Introduction to Colloids and Emulsions
Colloids and emulsions are important types of heterogeneous mixtures studied in surface chemistry and physical chemistry. In these systems, particles of one substance are dispersed throughout another substance, but the particles are small enough that they do not settle under gravity.
Colloidal systems are widely found in nature and everyday life. Examples include milk, fog, smoke, paints, creams, blood, and gels. Because of their unique physical and chemical properties, colloids play an essential role in many fields such as medicine, food science, environmental chemistry, nanotechnology, and industrial manufacturing.
An emulsion is a specific type of colloid in which one liquid is dispersed in another immiscible liquid. Emulsions are stabilized by substances called emulsifying agents or surfactants that prevent the droplets from merging.
The study of colloids and emulsions provides insight into:
- Particle dispersion and stability
- Surface interactions
- Optical properties of mixtures
- Industrial applications such as paints, cosmetics, and food products
Understanding colloidal systems helps scientists design stable products and control physical properties in many industrial and biological processes.
2. Definition of Colloids
A colloid is a heterogeneous mixture in which particles of one substance (called the dispersed phase) are distributed throughout another substance (called the dispersion medium).
The particle size in colloidal systems typically ranges from:
1 nanometer to 1000 nanometers
This size range is intermediate between true solutions and suspensions.
Examples of colloids include:
- Milk (fat droplets in water)
- Fog (water droplets in air)
- Smoke (solid particles in air)
- Paint (pigment particles in liquid)
Because colloidal particles are extremely small, they remain suspended for long periods and do not settle easily.
3. Components of a Colloidal System
Every colloidal system consists of two main components.
Dispersed Phase
The dispersed phase is the substance present in the form of small particles.
Examples:
- Fat droplets in milk
- Solid particles in smoke
Dispersion Medium
The dispersion medium is the continuous phase in which the dispersed particles are distributed.
Examples:
- Water in milk
- Air in smoke
4. Classification of Colloids
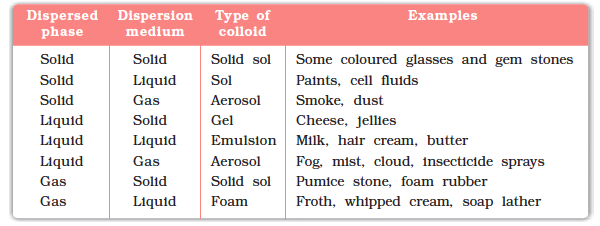


Colloids are classified based on the physical states of the dispersed phase and dispersion medium.
Sol
Solid dispersed in liquid.
Example:
Paints.
Gel
Liquid dispersed in solid.
Example:
Jelly.
Aerosol
Solid or liquid dispersed in gas.
Examples:
Smoke and fog.
Foam
Gas dispersed in liquid.
Example:
Whipped cream.
Emulsion
Liquid dispersed in another liquid.
Example:
Milk.
5. Properties of Colloids
Colloids exhibit several unique physical properties.
Tyndall Effect




The Tyndall effect is the scattering of light by colloidal particles.
When a beam of light passes through a colloidal solution, the path of light becomes visible due to scattering by particles.
Example:
Light beam visible in fog.
This effect distinguishes colloids from true solutions.
Brownian Motion
Brownian motion refers to the random movement of colloidal particles.
This motion occurs due to continuous collisions with molecules of the dispersion medium.
Brownian motion helps maintain stability of colloidal systems.
Electrical Charge on Colloidal Particles
Most colloidal particles carry an electric charge.
This charge causes repulsion between particles, preventing them from aggregating.
The presence of charge helps stabilize colloidal dispersions.
6. Preparation of Colloids
Colloids can be prepared using several methods.
Dispersion Methods
Large particles are broken into smaller colloidal particles.
Examples include:
- Mechanical grinding
- Ultrasonic dispersion
Condensation Methods
Small molecules combine to form larger colloidal particles.
Examples include:
- Chemical reactions
- Reduction reactions
7. Purification of Colloids
Impurities in colloidal solutions can be removed through purification techniques.
Common methods include:
Dialysis
Removal of small ions through a semipermeable membrane.
Ultrafiltration
Separation of colloidal particles using special filters.
Electrodialysis
Purification using an electric field.
8. Stability of Colloids




Colloids remain stable due to repulsive forces between particles.
Factors affecting stability include:
- Particle charge
- Particle size
- Temperature
- Presence of electrolytes
Coagulation
Coagulation occurs when colloidal particles aggregate and settle.
Adding electrolytes can neutralize charges and cause coagulation.
9. Emulsions



An emulsion is a colloidal system in which one liquid is dispersed in another immiscible liquid.
Because most liquids do not mix naturally, emulsions require stabilizing agents.
Examples of emulsions include:
- Milk
- Mayonnaise
- Cream
- Cosmetic lotions
10. Types of Emulsions
There are two major types of emulsions.
Oil-in-Water (O/W) Emulsion
Oil droplets dispersed in water.
Example:
Milk.
Water-in-Oil (W/O) Emulsion
Water droplets dispersed in oil.
Example:
Butter.
11. Emulsifying Agents
Emulsifying agents help stabilize emulsions.
They work by reducing surface tension between liquids.
Common emulsifiers include:
- Soaps
- Detergents
- Proteins
- Lecithin
Emulsifiers form protective layers around droplets and prevent them from merging.
12. Surfactants and Micelles
Surfactants are molecules containing:
- Hydrophilic head
- Hydrophobic tail
These molecules form micelles in water.
Micelles trap oil and grease, allowing them to be removed during cleaning.
This principle explains how soaps and detergents work.
13. Applications of Colloids and Emulsions
Colloidal systems have numerous applications.
Food Industry
Examples include:
- Milk
- Ice cream
- Butter
- Sauces
Pharmaceutical Industry
Colloids are used in:
- Drug delivery systems
- Medical suspensions
- Vaccines
Cosmetics
Examples include:
- Creams
- Lotions
- Makeup products
Environmental Applications
Colloids are used in water purification and pollution control.
14. Colloids in Biological Systems
Many biological fluids are colloidal in nature.
Examples include:
- Blood
- Cytoplasm
- Proteins in cells
Colloidal properties influence biological processes such as nutrient transport and cell communication.
15. Importance of Colloids and Emulsions
Colloidal systems are important in both natural processes and industrial applications.
They help explain:
- Stability of dispersed systems
- Behavior of nanoparticles
- Interaction between particles and surfaces
Colloid science is also essential for advances in nanotechnology and materials science.
Conclusion
Colloids and emulsions are important types of heterogeneous mixtures characterized by the dispersion of small particles within a continuous medium. Colloidal systems exhibit unique properties such as the Tyndall effect, Brownian motion, and electrical charge on particles. Emulsions are special types of colloids in which one liquid is dispersed in another immiscible liquid and stabilized by emulsifying agents. These systems are widely found in everyday life and have numerous applications in food science, medicine, cosmetics, environmental chemistry, and industrial processes. The study of colloids and emulsions provides valuable insights into particle interactions, surface chemistry, and the behavior of dispersed systems in both natural and technological environments.
