

1. Introduction to Coordination Chemistry
Coordination chemistry is the branch of chemistry that studies coordination compounds, which are chemical species formed by the interaction of a central metal atom or metal ion with surrounding molecules or ions called ligands. These compounds are also referred to as coordination complexes.
The concept of coordination compounds was first systematically explained by the Swiss chemist Alfred Werner, who received the Nobel Prize in Chemistry in 1913 for his pioneering work in this field. Werner’s coordination theory provided a framework for understanding how metal ions bond with surrounding ligands and form complex structures.
Coordination chemistry plays an essential role in various scientific and industrial fields, including:
- Bioinorganic chemistry
- Catalysis
- Medicine
- Environmental chemistry
- Materials science
- Analytical chemistry
Many important biological molecules, such as hemoglobin, chlorophyll, and vitamin B12, are coordination compounds. In addition, coordination complexes are widely used in industrial catalysis, electrochemistry, and pharmaceuticals.
2. Basic Concepts in Coordination Chemistry
Coordination chemistry revolves around several fundamental components that define coordination complexes.
Central Metal Atom or Ion
The central metal atom is usually a transition metal capable of forming coordinate bonds with surrounding ligands.
Examples include:
- Iron (Fe)
- Copper (Cu)
- Nickel (Ni)
- Cobalt (Co)
- Platinum (Pt)
These metals typically possess empty orbitals that can accept electron pairs from ligands.
Ligands
Ligands are ions or molecules that donate electron pairs to the metal center to form coordinate covalent bonds.
Common ligands include:
- Water (H₂O)
- Ammonia (NH₃)
- Chloride ion (Cl⁻)
- Cyanide ion (CN⁻)
Ligands may be neutral molecules or negatively charged ions.
Coordination Bond
A coordination bond is a type of covalent bond in which both electrons in the bond originate from the ligand.
This bond is also called a dative covalent bond.
Coordination Number
The coordination number is the number of ligand donor atoms attached to the central metal atom.
Common coordination numbers include:
- 2
- 4
- 6
3. Types of Ligands


Ligands are classified according to the number of donor atoms that bind to the metal center.
Monodentate Ligands
Monodentate ligands donate one pair of electrons.
Examples:
- H₂O
- NH₃
- Cl⁻
Bidentate Ligands
Bidentate ligands donate two electron pairs to the metal.
Example:
Ethylenediamine.
Polydentate Ligands
Polydentate ligands donate multiple electron pairs.
Example:
EDTA (ethylenediaminetetraacetic acid).
Polydentate ligands often form very stable complexes.
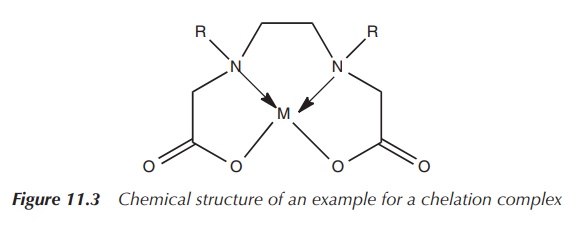
4. Chelation and Chelate Effect
Chelation occurs when a multidentate ligand forms multiple bonds with a single metal ion, creating a ring structure called a chelate ring.
The chelate effect refers to the enhanced stability of complexes containing multidentate ligands compared to those with monodentate ligands.
Chelating agents are widely used in:
- Medicine
- Metal ion removal
- Analytical chemistry
5. Nomenclature of Coordination Compounds
Coordination compounds follow specific naming rules.
General formula:
[Metal(Ligand)ₙ]
Example:
[Co(NH₃)₆]³⁺
Name:
Hexaamminecobalt(III) ion.
Rules include:
- Ligands named before metal.
- Neutral ligands keep molecular names.
- Anionic ligands end in “-o”.
- Metal oxidation state written in Roman numerals.
6. Structure and Geometry of Coordination Complexes
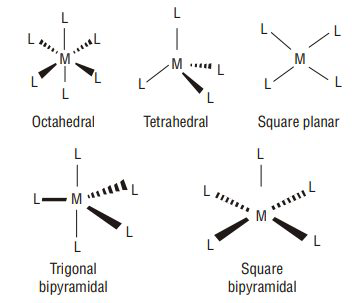



Coordination compounds can adopt several geometric structures depending on coordination number and ligand arrangement.
Octahedral Geometry
Coordination number = 6
Example:
[Co(NH₃)₆]³⁺
Ligands arranged around the metal at 90° angles.
Tetrahedral Geometry
Coordination number = 4
Example:
[NiCl₄]²⁻
Ligands positioned at the corners of a tetrahedron.
Square Planar Geometry
Coordination number = 4
Example:
[PtCl₄]²⁻
Common for d⁸ metal ions such as platinum and nickel.
7. Isomerism in Coordination Compounds



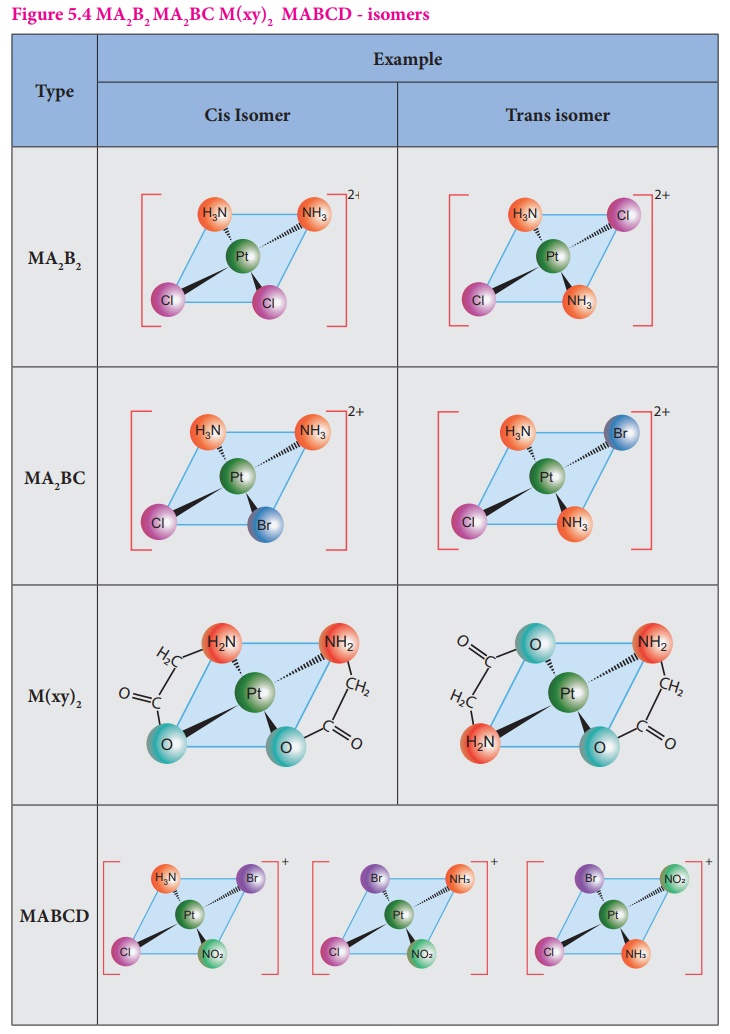
Coordination compounds often exhibit isomerism, meaning compounds have the same formula but different arrangements.
Types include:
Structural Isomerism
Different connectivity of atoms.
Examples:
- Ionization isomerism
- Hydrate isomerism
Stereoisomerism
Same bonding but different spatial arrangement.
Types include:
- Geometrical isomerism (cis/trans)
- Optical isomerism
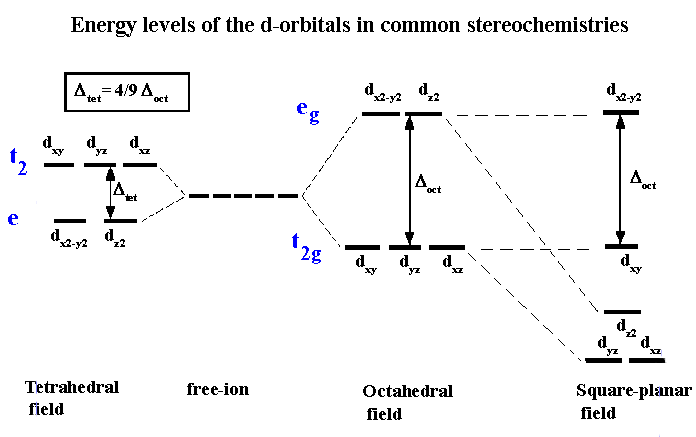
8. Crystal Field Theory
Crystal field theory explains the electronic structure of metal complexes.
It describes how ligands affect the d-orbitals of metal ions.
When ligands approach the metal ion, the five d-orbitals split into different energy levels.
In octahedral complexes:
- t₂g orbitals (lower energy)
- e_g orbitals (higher energy)
The energy difference is called crystal field splitting energy.
9. Color of Coordination Compounds
Many coordination compounds are brightly colored.
The color arises from electronic transitions between split d-orbitals.
Example:
Copper complexes appear blue due to d–d transitions.
Different ligands can change the color of the complex.
10. Magnetic Properties of Coordination Compounds
Coordination compounds may exhibit magnetic behavior depending on electron configuration.
Types include:
Paramagnetic
Contain unpaired electrons.
Example:
Fe³⁺ complexes.
Diamagnetic
All electrons paired.
Example:
Zn²⁺ complexes.
Magnetic properties help determine electronic structure.
11. Coordination Chemistry in Biological Systems




Coordination chemistry plays a vital role in biological systems.
Examples include:
Hemoglobin
Contains iron coordinated to nitrogen atoms in heme group.
Responsible for oxygen transport in blood.
Chlorophyll
Contains magnesium ion coordinated to nitrogen atoms.
Essential for photosynthesis.
Vitamin B12
Contains cobalt metal center.
Important for metabolic processes.
12. Industrial Applications of Coordination Chemistry
Coordination compounds are widely used in industry.
Examples include:
- Catalysts in chemical reactions
- Pigments and dyes
- Electroplating
- Metallurgy
Transition metal catalysts are essential in many industrial processes.
13. Coordination Compounds in Medicine
Coordination chemistry has significant medical applications.
Example:
Cisplatin, a platinum complex used in cancer treatment.
Chelating agents are used to remove toxic metals from the body.
Coordination compounds are also used in medical imaging and diagnostics.
14. Environmental Applications
Coordination chemistry helps control environmental pollution.
Applications include:
- Removal of heavy metals from water
- Environmental monitoring
- Catalytic converters
Chelating agents help bind and remove toxic metal ions.
15. Importance of Coordination Chemistry
Coordination chemistry is essential for understanding:
- Metal-ligand interactions
- Catalysis
- Biological metal complexes
- Industrial chemical processes
It bridges inorganic chemistry, biochemistry, and materials science.
Conclusion
Coordination chemistry studies compounds formed by the interaction of metal ions with ligands through coordinate covalent bonds. These complexes exhibit unique structures, properties, and chemical behaviors that are fundamental to many natural and technological processes. Coordination compounds display diverse geometries, isomerism, and electronic properties explained by crystal field theory. They play essential roles in biological systems, industrial catalysis, medicine, and environmental chemistry. Understanding coordination chemistry allows scientists to design new materials, develop pharmaceuticals, and explore the chemistry of metals in living organisms.
