



Introduction to the Gaseous State
Matter exists in different physical forms known as states of matter. The three classical states are solid, liquid, and gas, while modern science also recognizes additional states such as plasma and Bose–Einstein condensate. Among these, the gaseous state is the most dynamic and least structured state of matter.
In the gaseous state, particles such as atoms or molecules are widely separated and move freely in all directions. Because of this freedom of motion and the large distance between particles, gases show properties that are significantly different from solids and liquids.
The gaseous state plays an essential role in nature and technology. The air we breathe is a mixture of gases, including nitrogen, oxygen, carbon dioxide, and water vapor. Many industrial processes involve gases, including combustion, refrigeration, chemical manufacturing, and energy production.
Understanding the behavior of gases helps scientists explain atmospheric processes, weather patterns, chemical reactions, and the functioning of engines and biological systems.
1. Characteristics of Gases
1.1 Lack of Definite Shape and Volume
Unlike solids and liquids, gases do not have a fixed shape or volume. Instead, they expand to fill the entire container in which they are placed.
For example, when air is placed in a balloon, the gas spreads out and occupies the entire interior space of the balloon. Similarly, gases inside a room fill the whole room evenly.
This behavior occurs because gas molecules move independently and are not held in fixed positions.
1.2 High Compressibility
Gases are highly compressible compared with solids and liquids. When pressure is applied, gas molecules can be pushed closer together because there is a large amount of empty space between them.
This property allows gases to be stored in compressed form. For example:
- Oxygen cylinders used in hospitals
- Compressed natural gas (CNG) used as fuel
- Aerosol sprays
In contrast, liquids and solids cannot be compressed significantly because their particles are already closely packed.
1.3 Low Density
Density refers to the mass per unit volume of a substance.
Gases have much lower density than solids and liquids because their molecules are far apart. For example:
- Air density ≈ 1.2 kg/m³
- Water density ≈ 1000 kg/m³
This large difference explains why gases rise above liquids and why balloons filled with lighter gases such as helium float in the air.
1.4 Rapid Diffusion
Diffusion is the process by which particles spread from a region of high concentration to a region of low concentration.
Gases diffuse rapidly because their particles move freely and randomly. A common example is the smell of perfume spreading across a room after it is sprayed.
The rate of diffusion depends on factors such as:
- Molecular mass
- Temperature
- Pressure
Lighter gases diffuse faster than heavier gases.
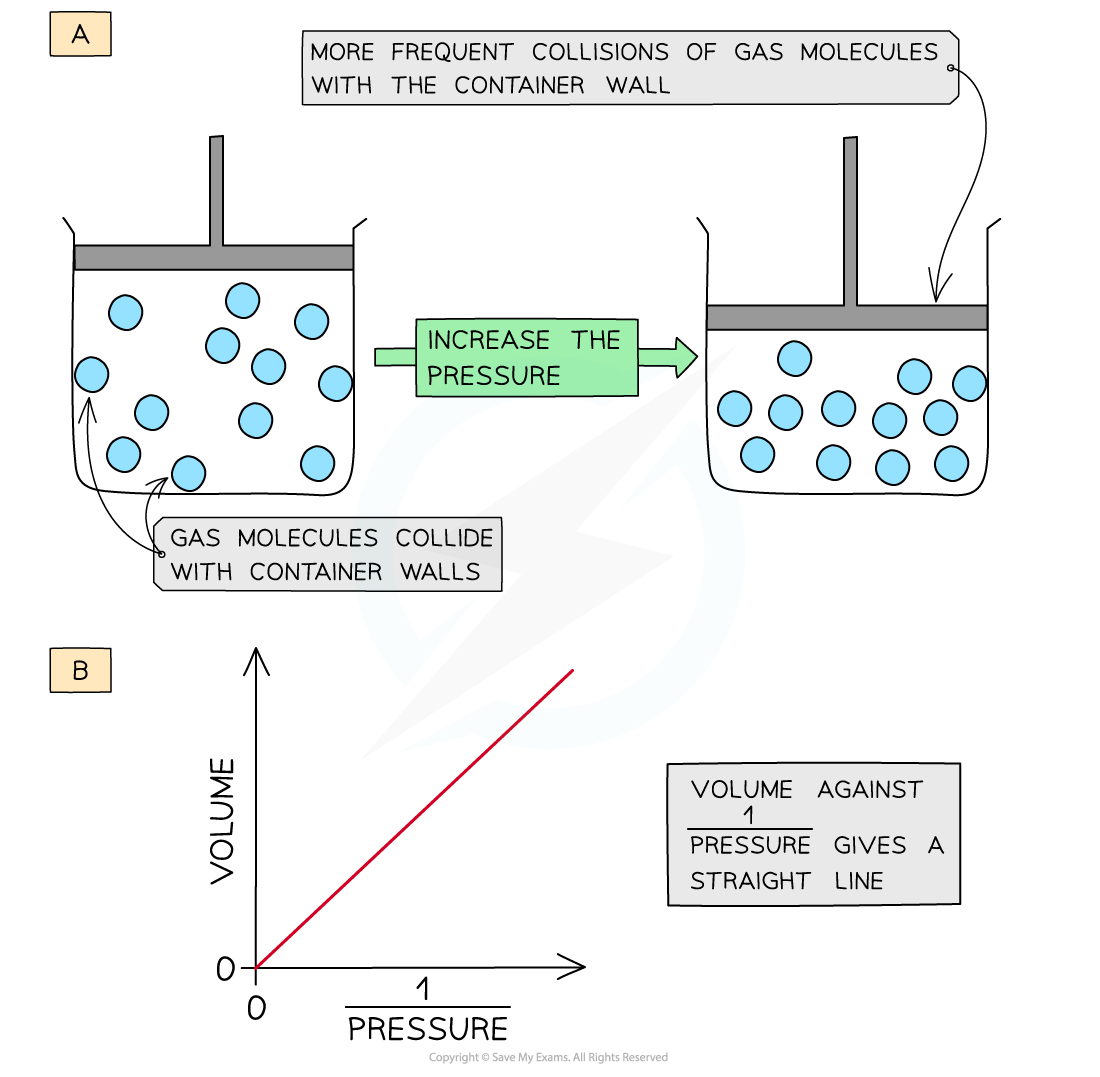
1.5 Ability to Exert Pressure
Gas molecules are constantly moving and colliding with the walls of their container. These collisions produce gas pressure.
Pressure is defined as the force exerted per unit area.
Gas pressure depends on:
- Number of molecules
- Temperature
- Volume of the container
This principle explains why increasing the temperature of a gas in a sealed container increases the pressure.
2. Molecular Nature of Gases


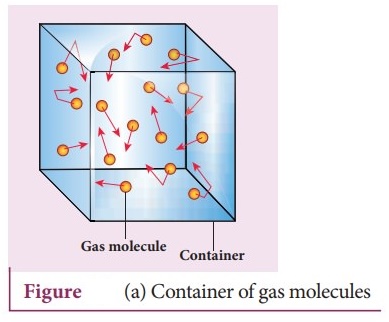
The microscopic behavior of gases is explained by the Kinetic Molecular Theory (KMT).
2.1 Basic Assumptions of Kinetic Molecular Theory
The theory is based on several fundamental assumptions:
- Gases consist of a large number of tiny particles (atoms or molecules).
- Particles move continuously in random directions.
- The volume of gas molecules is negligible compared with the volume of the container.
- No intermolecular forces act between gas molecules, except during collisions.
- Collisions between molecules and container walls are perfectly elastic.
- The average kinetic energy of molecules is proportional to absolute temperature.
These assumptions allow scientists to explain gas behavior mathematically.
2.2 Random Motion of Gas Molecules
Gas molecules move randomly and rapidly in all directions. This random motion results in frequent collisions between molecules and with the walls of the container.
These collisions are responsible for gas pressure.
2.3 Mean Free Path
The mean free path is the average distance a molecule travels between two successive collisions.
Factors affecting mean free path include:
- Temperature
- Pressure
- Size of molecules
At higher pressures, molecules are closer together, so the mean free path decreases.
2.4 Kinetic Energy of Gas Molecules
The kinetic energy of gas molecules depends on temperature.
Higher temperature means molecules move faster and possess more kinetic energy. This relationship explains many gas laws.
3. Gas Laws
Gas laws describe the mathematical relationships between pressure, volume, temperature, and the number of gas molecules.
3.1 Boyle’s Law
PV = \text{constant}
Boyle’s Law states that the pressure of a fixed amount of gas is inversely proportional to its volume at constant temperature.
If the volume decreases, pressure increases.
Examples include:
- Compressing air in a syringe
- Breathing process in lungs
3.2 Charles’s Law
\frac{V}{T} = \text{constant}
Charles’s Law states that the volume of a gas is directly proportional to its absolute temperature at constant pressure.
As temperature increases, gas expands.
Example:
Hot air balloons rise because heated air expands and becomes less dense.
3.3 Gay-Lussac’s Law
Gay-Lussac’s Law states that pressure is directly proportional to temperature when volume is constant.
Mathematically:
P ∝ T
This explains why sealed containers can explode when heated.
3.4 Avogadro’s Law
Avogadro’s Law states:
Equal volumes of gases at the same temperature and pressure contain equal numbers of molecules.
This law introduced the concept of Avogadro’s number, approximately:
6.022 × 10²³ particles per mole.
3.5 Ideal Gas Equation
genui{“math_block_widget_always_prefetch_v2”:{“content”:”PV = nRT”}}
The ideal gas equation combines all gas laws into a single formula.
Where:
P = Pressure
V = Volume
n = Number of moles
R = Gas constant
T = Temperature (Kelvin)
This equation is widely used in chemistry and physics.
4. Real Gases




In reality, gases do not perfectly follow the ideal gas equation.
4.1 Reasons for Deviation
Real gases deviate because:
- Gas molecules occupy volume.
- Intermolecular attractions exist.
These factors become important at:
- High pressure
- Low temperature
4.2 Van der Waals Equation
To correct deviations from ideal behavior, the Van der Waals equation was proposed.
It modifies pressure and volume terms to account for molecular size and attraction forces.
4.3 Critical Temperature and Pressure
The critical temperature is the highest temperature at which a gas can be liquefied.
The critical pressure is the minimum pressure required to liquefy a gas at its critical temperature.
Above these values, gases cannot be liquefied.
5. Liquefaction of Gases
Liquefaction is the process of converting gases into liquids.
It occurs through:
- Increasing pressure
- Decreasing temperature
Important methods include:
- Linde Process
- Claude Process
Liquefied gases are widely used in industries.
Examples include:
- Liquid oxygen
- Liquid nitrogen
- Liquefied petroleum gas (LPG)
6. Diffusion and Effusion of Gases
6.1 Diffusion
Diffusion is the mixing of gases due to random molecular motion.
Factors affecting diffusion:
- Temperature
- Molecular mass
- Pressure
Higher temperature increases diffusion rate.
6.2 Effusion
Effusion is the escape of gas molecules through a tiny hole without significant collisions.
Effusion rate depends on molecular mass.
6.3 Graham’s Law
Graham’s Law states:
Rate of diffusion ∝ 1/√M
Where M is molar mass.
This means lighter gases diffuse faster.
7. Partial Pressure of Gases
In mixtures of gases, each gas contributes to the total pressure.
Dalton’s Law of Partial Pressures
Total pressure = sum of partial pressures of individual gases.
Mathematically:
Ptotal = P1 + P2 + P3 + …
This law is important in atmospheric science and respiration.
8. Gas Mixtures
The atmosphere is an example of a gas mixture.
Composition of dry air approximately:
- Nitrogen – 78%
- Oxygen – 21%
- Argon – 0.93%
- Carbon dioxide – 0.04%
Gas mixtures behave according to the same gas laws as individual gases.
9. Applications of the Gaseous State
9.1 Atmospheric Science
Understanding gas behavior helps explain:
- Weather patterns
- Wind formation
- Atmospheric pressure
9.2 Industrial Applications
Gases are widely used in industries:
- Oxygen in steel production
- Nitrogen for food preservation
- Hydrogen in fuel cells
9.3 Medical Applications
Examples include:
- Oxygen therapy
- Anesthetic gases
- Respiratory treatments
9.4 Environmental Applications
Gas laws help in understanding:
- Air pollution
- Greenhouse gases
- Climate change
10. Importance of Studying the Gaseous State
The gaseous state is fundamental to both scientific research and everyday life.
Understanding gas behavior helps in:
- Designing engines
- Predicting atmospheric changes
- Developing industrial chemical processes
- Studying biological respiration
Because gases are the most mobile form of matter, they provide valuable insights into molecular motion and energy.
Conclusion
The gaseous state represents one of the most dynamic and fundamental states of matter. Gases differ from solids and liquids in their lack of fixed shape and volume, high compressibility, low density, and rapid diffusion. These properties arise from the large separation between gas molecules and their continuous random motion.
The behavior of gases is described by various gas laws such as Boyle’s law, Charles’s law, Gay-Lussac’s law, and Avogadro’s law. These relationships are unified in the ideal gas equation, which forms the basis of many calculations in chemistry and physics.
However, real gases deviate from ideal behavior under extreme conditions, requiring more advanced models such as the Van der Waals equation. Concepts like diffusion, effusion, partial pressures, and liquefaction further explain how gases behave in real environments.
From atmospheric science and industrial processes to medical applications and environmental studies, the gaseous state plays a critical role in modern science and technology. Understanding gases not only provides insight into molecular behavior but also enables advancements in energy production, chemical manufacturing, and environmental protection.
