



📘 Introduction to Acid Rain
Acid rain refers to precipitation (rain, snow, sleet, or fog) that contains high levels of acidic components, primarily sulfuric acid (H₂SO₄) and nitric acid (HNO₃). It is a major environmental issue caused by the emission of sulfur dioxide (SO₂) and nitrogen oxides (NOₓ) into the atmosphere, mainly from industrial activities and the burning of fossil fuels.
Acid rain does not always fall directly near pollution sources. Pollutants can travel long distances through the atmosphere, making acid rain a global environmental problem.
🌱 Concept and Definition
📖 Definition
Acid rain is the deposition of acidic substances from the atmosphere onto the Earth’s surface, either in wet or dry form, resulting in environmental damage.
🔑 Key Features:
- Contains sulfuric and nitric acids
- Formed from air pollutants
- Can travel across regions
- Affects soil, water, plants, and structures
🌧️ Types of Acid Deposition
💧 1. Wet Deposition

- Occurs when acids mix with rain, snow, or fog
- Falls to the ground as acidic precipitation
🌫️ 2. Dry Deposition
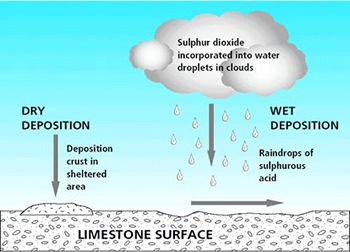

- Acidic gases and particles settle on surfaces
- Become acidic when they react with water
🔬 Formation of Acid Rain
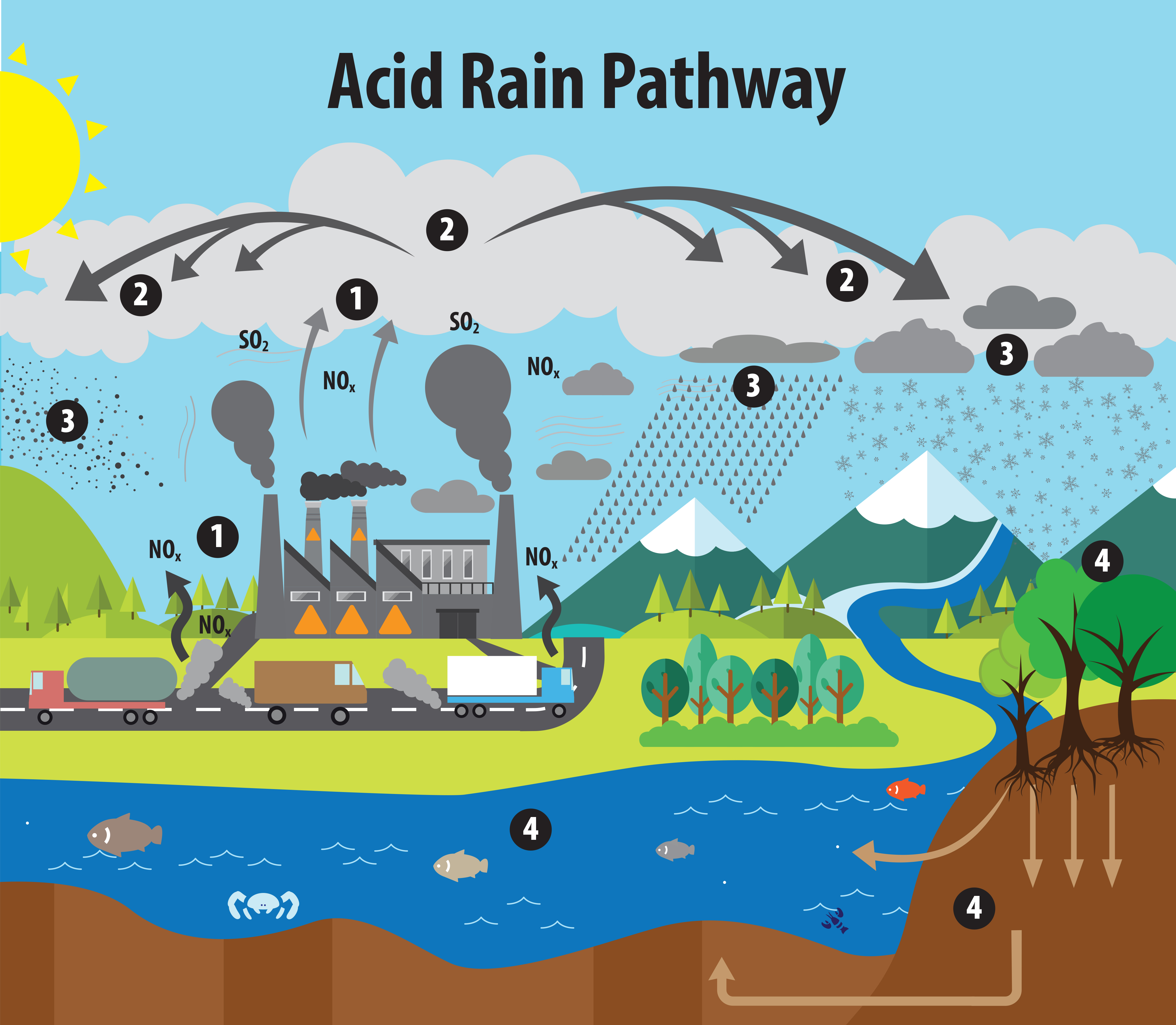

🧪 Chemical Process:
Step 1: Emission of Pollutants
- Sulfur dioxide (SO₂)
- Nitrogen oxides (NOₓ)
Step 2: Atmospheric Reactions
- SO₂ + O₂ → SO₃
- SO₃ + H₂O → H₂SO₄ (sulfuric acid)
- NOₓ + H₂O → HNO₃ (nitric acid)
Step 3: Deposition
- Acids fall as precipitation
🌍 Sources of Acid Rain
🏭 1. Anthropogenic Sources



🔥 Fossil Fuel Combustion
- Coal-fired power plants
- Major source of SO₂
🚗 Transportation
- Vehicles emit NOₓ
🏭 Industrial Activities
- Oil refineries
- Chemical industries
⚡ Power Generation
- Thermal power plants
🌋 2. Natural Sources


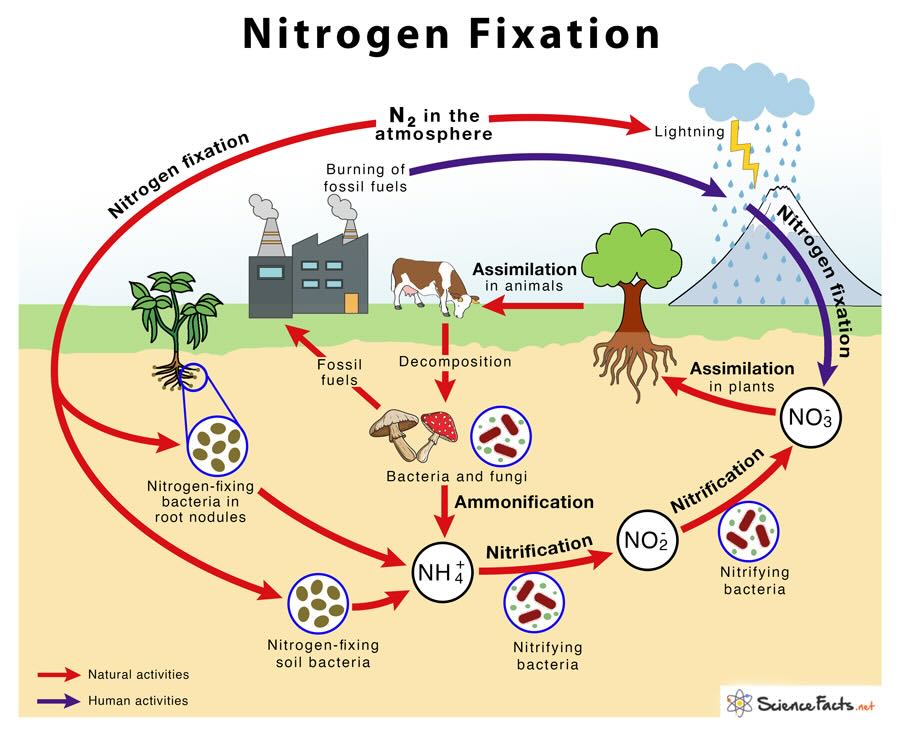
- Volcanic eruptions
- Forest fires
- Lightning
🔬 Chemistry of Acid Rain
📏 pH Scale:
- Normal rain: ~5.6 (slightly acidic)
- Acid rain: 4.0 or lower
Lower pH indicates higher acidity.
⚠️ Effects of Acid Rain
🌿 1. Effects on Vegetation


🌱 Leaf Damage
- Weakens plants
🌳 Forest Decline
- Nutrient loss in soil
🌾 Reduced Crop Yield
- Affects agriculture
🌍 2. Effects on Soil


🧪 Nutrient Leaching
- Loss of calcium, magnesium
⚠️ Toxic Metal Release
- Aluminum becomes soluble
💧 3. Effects on Water Bodies


🐟 Fish Death
- Low pH harms aquatic life
🌊 Ecosystem Disruption
- Loss of biodiversity
👨⚕️ 4. Effects on Human Health
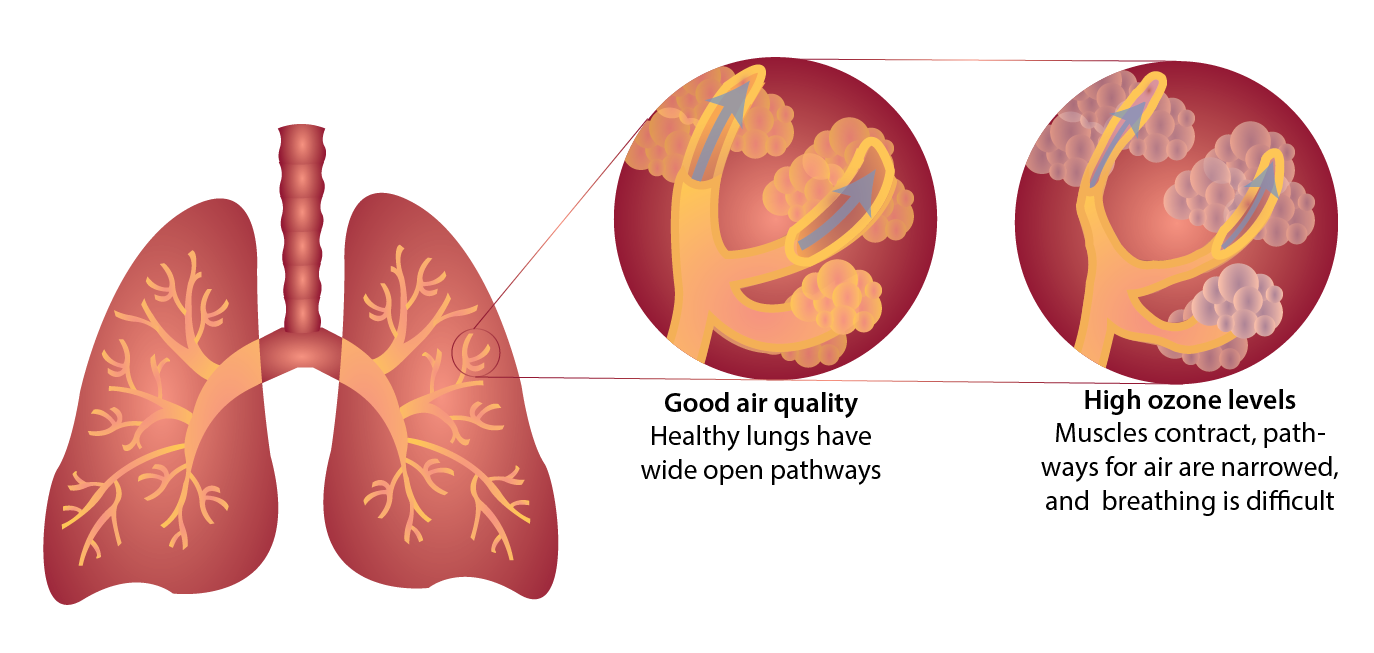

- Respiratory problems
- Skin irritation
- Indirect effects through polluted water
🏛️ 5. Effects on Buildings and Monuments


🏛️ Corrosion
- Damages marble, limestone
🏗️ Structural Damage
- Weakens buildings
🌍 Acid Rain in India


Key Issues:
- Industrial emissions
- Vehicle pollution
Example:
- Taj Trapezium Zone to protect monuments
🔬 Measurement of Acid Rain
📏 Indicators:
- pH level
- Sulfate and nitrate concentration
🌱 Control and Prevention



🏭 Industrial Control
- Use of scrubbers
- Emission reduction
🚗 Vehicle Control
- Catalytic converters
🌿 Renewable Energy
- Reduce fossil fuel use
🌳 Afforestation
- Absorbs pollutants
⚖️ Government Regulations
- Emission standards
⚖️ Global Efforts
- Clean Air Acts
- International agreements
📚 Advantages of Controlling Acid Rain
- Protect ecosystems
- Preserve monuments
- Improve health
❌ Challenges
- Industrial dependency
- Cross-border pollution
- Economic costs
🔮 Future Solutions
- Green technology
- Cleaner fuels
- International cooperation
🔄 Relationship with Other Environmental Issues
- Linked to air pollution
- Contributes to soil and water pollution
🧾 Conclusion
Acid rain is a serious environmental issue caused by air pollution, affecting ecosystems, human health, and infrastructure. Its impacts are widespread and long-lasting.
Effective solutions require:
- Reducing emissions
- Adopting clean energy
- Strengthening environmental policies
By addressing acid rain, we can protect the environment and ensure a sustainable future.
