



Introduction
The hydrogen atom is the simplest and most fundamental atomic system in physics and chemistry. It consists of one proton in the nucleus and one electron orbiting around it. Because of its simplicity, the hydrogen atom has played a central role in the development of atomic theory and quantum mechanics. Many of the fundamental principles of modern physics were first understood by studying hydrogen.
The hydrogen atom provides a system that can be described mathematically with great precision. Its behavior helped scientists develop theories that explain atomic structure, electron energy levels, and the interaction between matter and radiation. The study of hydrogen led to major contributions from scientists such as Niels Bohr, Erwin Schrödinger, Werner Heisenberg, and Max Planck.
Hydrogen is also the most abundant element in the universe, forming the primary component of stars and interstellar matter. Understanding hydrogen is therefore essential for astrophysics, atomic physics, spectroscopy, and quantum mechanics.
Basic Structure of the Hydrogen Atom
The hydrogen atom contains two main components:
Proton
The nucleus of the hydrogen atom contains a single proton, which carries a positive electric charge.
Properties of the proton include:
- Charge: (+1.602 \times 10^{-19}) coulombs
- Mass: (1.673 \times 10^{-27}) kg
The proton determines the identity of the element hydrogen.
Electron
The electron is a negatively charged particle that moves around the nucleus.
Properties of the electron include:
- Charge: (-1.602 \times 10^{-19}) coulombs
- Mass: (9.11 \times 10^{-31}) kg
The electron is bound to the proton through electrostatic attraction.
Classical View of the Hydrogen Atom
Before quantum theory, the hydrogen atom was often compared to a miniature solar system, where electrons orbit the nucleus just as planets orbit the Sun.
This classical picture emerged from the nuclear model proposed by Ernest Rutherford.
However, classical physics predicted that orbiting electrons would continuously emit electromagnetic radiation and eventually spiral into the nucleus. This contradiction indicated that classical physics could not fully explain atomic stability.
Bohr Model of the Hydrogen Atom




A major breakthrough came in 1913 when Niels Bohr proposed a new model for the hydrogen atom.
Bohr’s Postulates
Bohr introduced several revolutionary ideas:
- Electrons move in stable circular orbits around the nucleus.
- Each orbit corresponds to a specific quantized energy level.
- Electrons do not radiate energy while remaining in a stable orbit.
- Radiation is emitted or absorbed when electrons transition between energy levels.
Quantization of Angular Momentum
Bohr proposed that the angular momentum of the electron is quantized:
[
mvr = \frac{nh}{2\pi}
]
Where
- (n) = principal quantum number
- (h) = Planck’s constant
This condition restricts electrons to certain allowed orbits.
Energy Levels of Hydrogen
The energy levels of the hydrogen atom are given by:
[
E_n = -\frac{13.6}{n^2} , eV
]
Where:
- (n = 1, 2, 3, …)
The negative sign indicates that the electron is bound to the nucleus.
Electron Transitions
When electrons move between energy levels:
- Energy is absorbed if the electron moves to a higher orbit.
- Energy is emitted when the electron falls to a lower orbit.
The emitted radiation appears as spectral lines.
Hydrogen Spectral Series

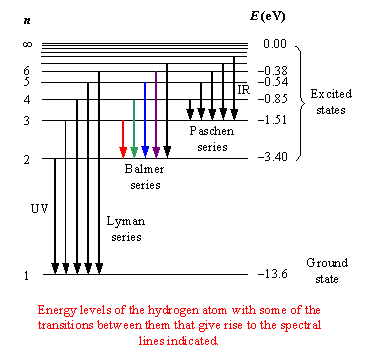

The hydrogen atom produces characteristic spectral lines when electrons transition between energy levels.
These spectral lines form several series.
Lyman Series
Transitions that end at (n = 1).
Characteristics:
- Occur in the ultraviolet region
- High-energy transitions
Balmer Series
Transitions ending at (n = 2).
Characteristics:
- Visible light spectrum
- Responsible for the bright lines observed in hydrogen emission spectra.
Paschen Series
Transitions ending at (n = 3).
Characteristics:
- Occur in the infrared region.
Rydberg Formula
The wavelengths of spectral lines are described by the Rydberg equation:
[
\frac{1}{\lambda} = R\left(\frac{1}{n_1^2} – \frac{1}{n_2^2}\right)
]
Where:
- (R) is the Rydberg constant
- (n_2 > n_1)
This formula successfully predicts hydrogen spectral lines.
Quantum Mechanical Model of Hydrogen

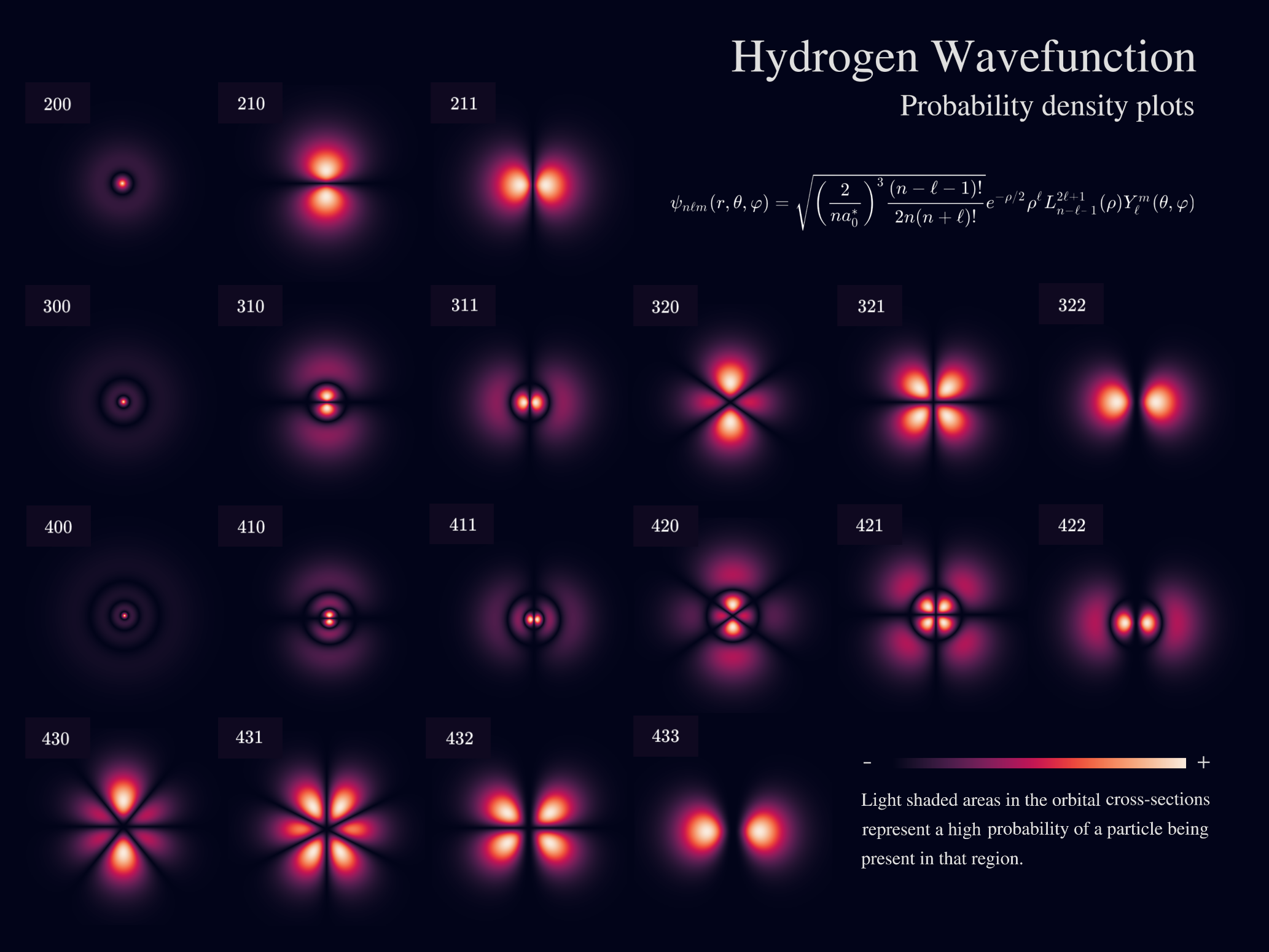


The Bohr model was eventually replaced by the quantum mechanical model, developed by Erwin Schrödinger and other physicists.
In this model:
- Electrons behave like waves.
- Their positions are described by probability distributions.
Schrödinger Equation
The hydrogen atom is described by the Schrödinger equation:
[
H\psi = E\psi
]
Solutions to this equation give wave functions representing electron probabilities.
Quantum Numbers
The hydrogen atom is described using four quantum numbers.
Principal Quantum Number (n)
Represents the main energy level.
Values:
[
n = 1,2,3,4…
]
Azimuthal Quantum Number (l)
Describes orbital shape.
[
l = 0,1,2… (n-1)
]
Types of orbitals:
- s
- p
- d
- f
Magnetic Quantum Number (m)
Determines orbital orientation.
Spin Quantum Number
Represents electron spin:
[
+\frac{1}{2}, -\frac{1}{2}
]
Hydrogen Atomic Orbitals
Orbitals represent regions where the probability of finding an electron is high.
Common orbitals include:
s Orbital
- Spherical shape
- Found in all energy levels
p Orbital
- Dumbbell-shaped
- Found from (n=2)
d and f Orbitals
More complex shapes.
Fine Structure of Hydrogen Spectrum
Small splitting of spectral lines occurs due to relativistic effects and electron spin interactions.
This phenomenon is known as fine structure.
It arises from:
- Spin–orbit coupling
- Relativistic corrections
Hyperfine Structure
Even smaller spectral splitting occurs due to interactions between the electron and nuclear spins.
This effect is called hyperfine structure.
It is important in technologies such as atomic clocks.
Hydrogen in Astrophysics
Hydrogen plays a crucial role in astronomy.
It is the most abundant element in the universe, making up about 75% of visible matter.
Hydrogen is the primary fuel for stellar nuclear fusion in stars.
Stars generate energy by converting hydrogen into helium.
Hydrogen Isotopes
Hydrogen has three isotopes.
Protium
- One proton
- One electron
- No neutrons
Deuterium
- One proton
- One neutron
Tritium
- One proton
- Two neutrons
These isotopes differ in mass but behave similarly chemically.
Hydrogen in Modern Physics
Hydrogen remains an important system in modern research.
Scientists study hydrogen to understand:
- Quantum electrodynamics
- Atomic transitions
- Precision measurements
- Fundamental constants
Because its equations can be solved exactly, hydrogen is used as a benchmark system in physics.
Applications of Hydrogen Atom Studies
Research on hydrogen atoms has led to many technological advancements.
Laser Technology
Hydrogen spectral transitions helped develop laser physics.
Atomic Clocks
Hydrogen masers are used for highly precise time measurement.
Quantum Mechanics
Hydrogen is used to test quantum theory.
Plasma Physics
Hydrogen plasma is studied in nuclear fusion experiments.
Importance in Education
The hydrogen atom is widely used in physics and chemistry education because:
- Its structure is simple
- Mathematical solutions are possible
- It demonstrates quantum principles clearly
Students often learn hydrogen before studying more complex atoms.
Conclusion
The hydrogen atom represents the simplest and most fundamental atomic system, yet it has played an extraordinary role in shaping modern physics. From early atomic theories to the development of quantum mechanics, the hydrogen atom has served as a crucial testing ground for scientific ideas.
The Bohr model provided the first successful explanation of hydrogen’s spectral lines, while quantum mechanics later offered a deeper and more accurate description of electron behavior. Modern atomic physics continues to use hydrogen as a key system for understanding fundamental interactions and testing theoretical predictions.
Because hydrogen is abundant throughout the universe and essential for stellar processes, its study also connects atomic physics with astrophysics and cosmology. The hydrogen atom therefore remains one of the most important subjects in the study of matter and the physical universe.
