

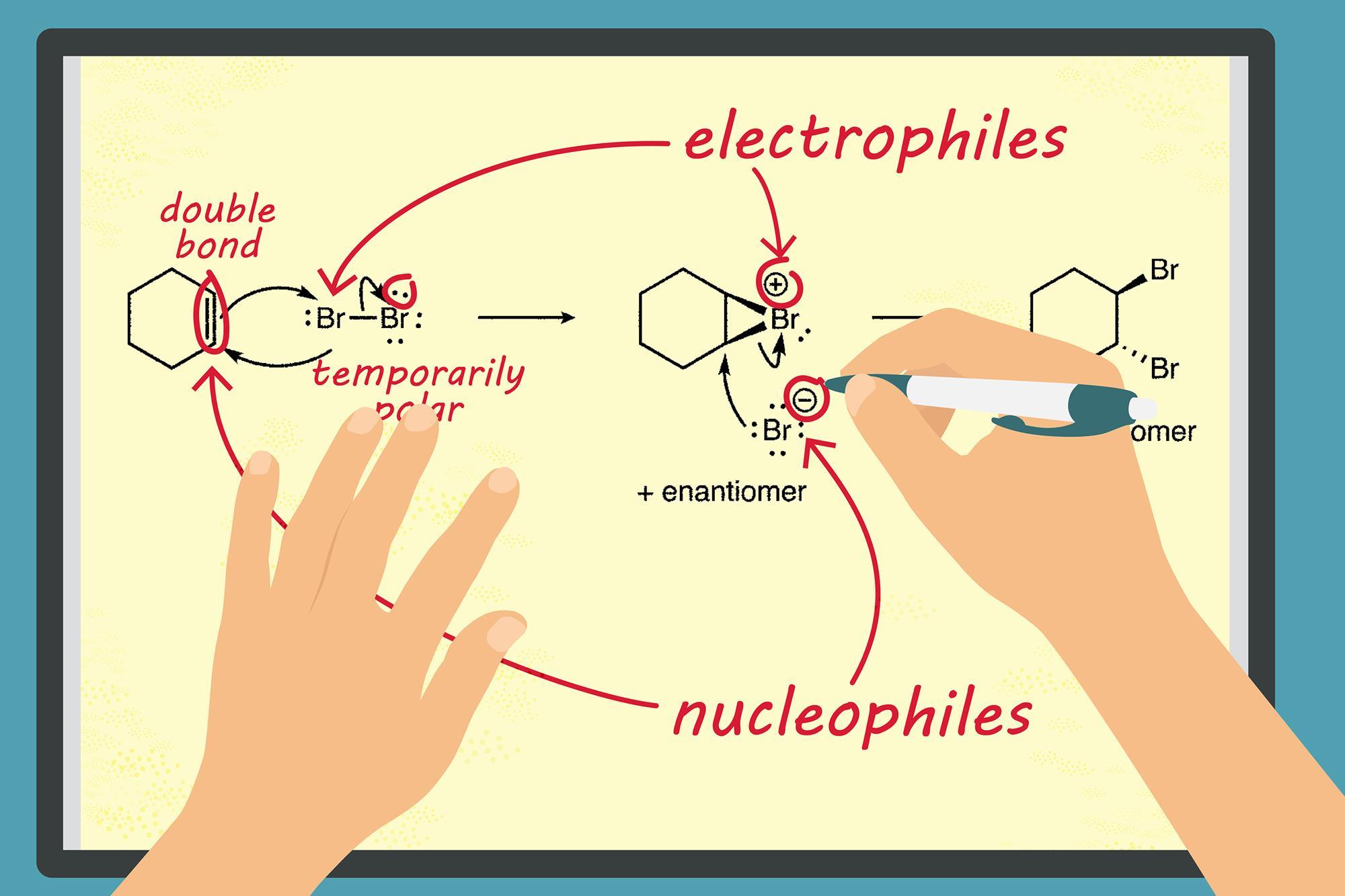
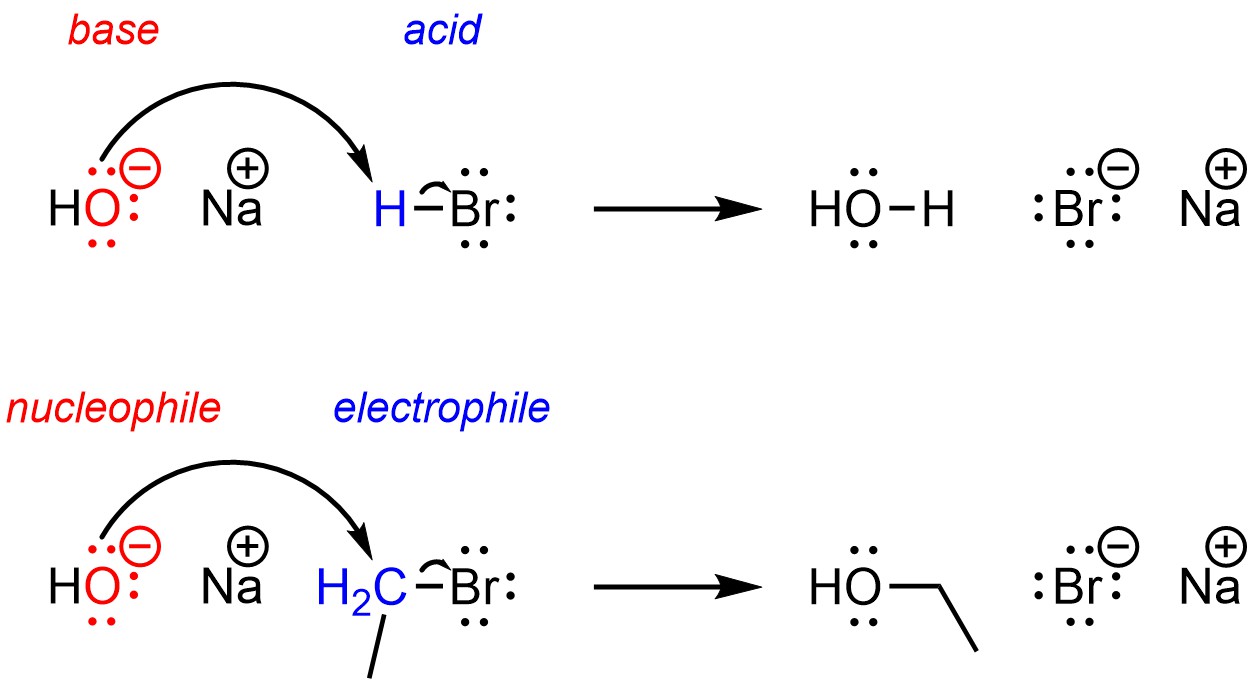
1. Introduction to Organic Reaction Mechanisms
Organic reaction mechanisms describe the step-by-step sequence of events that occur during a chemical reaction involving organic molecules. These mechanisms explain how reactants are converted into products by showing the movement of electrons, the formation and breaking of chemical bonds, and the formation of intermediate species.
In organic chemistry, simply writing the overall reaction equation is not enough to fully understand how a reaction occurs. Instead, chemists analyze the detailed pathway that molecules follow during the transformation. This pathway is called the reaction mechanism.
Organic reaction mechanisms provide insights into:
- How bonds break and form
- How electrons move between atoms
- The stability of intermediates
- The rate of reactions
- The formation of products
Understanding reaction mechanisms is essential for:
- Predicting reaction outcomes
- Designing chemical syntheses
- Developing pharmaceuticals
- Studying biological processes
- Creating new materials
Mechanistic studies are therefore fundamental to modern organic chemistry.
2. Fundamental Concepts in Reaction Mechanisms
To understand reaction mechanisms, several key concepts must be considered.
Electron Movement
Most organic reactions involve the movement of electrons. Electrons move from regions of high electron density to regions of low electron density.
This movement is represented using curved arrows in reaction mechanisms.
Bond Breaking and Bond Formation
Two main types of bond breaking occur:
Homolytic cleavage
The bond breaks evenly, and each atom receives one electron.
Heterolytic cleavage
One atom receives both electrons from the bond.
Intermediates
Intermediates are unstable species that form during the reaction but do not appear in the final products.
Examples include:
- Carbocations
- Carbanions
- Free radicals
Transition States
A transition state is a high-energy structure that occurs during the transformation from reactants to products.
It represents the point where bonds are partially broken and partially formed.
3. Types of Organic Reaction Mechanisms
Organic reactions can be classified based on how molecules interact and how bonds change.
Major types include:
- Substitution reactions
- Addition reactions
- Elimination reactions
- Rearrangement reactions
Each type follows specific mechanistic pathways.
4. Substitution Reactions

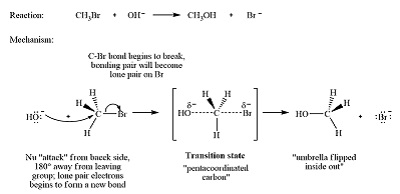

Substitution reactions occur when one atom or group in a molecule is replaced by another atom or group.
Two major mechanisms exist:
SN1 Reaction
SN1 stands for Substitution Nucleophilic Unimolecular.
Characteristics:
- Two-step mechanism
- Formation of carbocation intermediate
- Reaction rate depends on one reactant
Example steps:
- Leaving group departs
- Nucleophile attacks carbocation
SN1 reactions are common in tertiary alkyl halides.
SN2 Reaction
SN2 stands for Substitution Nucleophilic Bimolecular.
Characteristics:
- One-step reaction
- No intermediate
- Reaction occurs through backside attack
The nucleophile attacks the carbon atom while the leaving group leaves simultaneously.
SN2 reactions are common in primary alkyl halides.
5. Addition Reactions
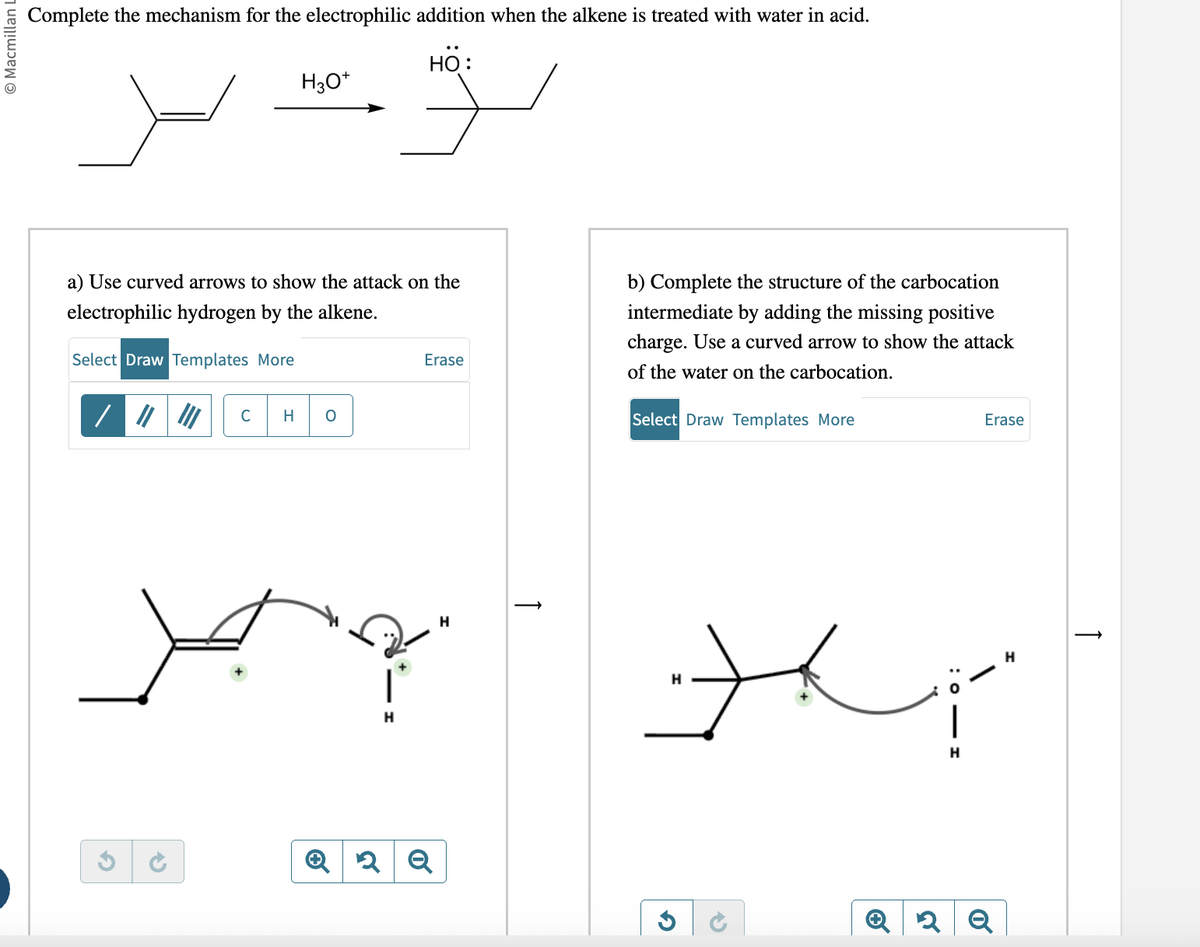

Addition reactions occur when atoms or groups add across a double or triple bond.
Example:
Ethene + HBr → Bromoethane
Steps:
- Electrophile attacks double bond
- Carbocation intermediate forms
- Nucleophile attaches
Addition reactions are common in alkenes and alkynes.
Types include:
- Hydrogenation
- Halogenation
- Hydration
6. Elimination Reactions



Elimination reactions remove atoms from molecules to form double bonds.
Two main mechanisms exist:
E1 Reaction
Two-step mechanism.
- Leaving group leaves
- Proton removed forming double bond
Occurs through carbocation intermediate.
E2 Reaction
One-step mechanism.
Base removes proton while leaving group departs simultaneously.
This produces an alkene.
7. Rearrangement Reactions


Rearrangement reactions involve reorganization of atoms within a molecule.
Example:
Hydride shift.
In this process, a hydrogen atom moves along with its electron pair to form a more stable carbocation.
Rearrangements occur because molecules seek the most stable structure.
8. Important Reactive Intermediates
Several intermediates are commonly encountered in organic mechanisms.
Carbocations
Positively charged carbon atoms.
Stability order:
Tertiary > Secondary > Primary.
Carbanions
Negatively charged carbon atoms.
More stable when electron-withdrawing groups are present.
Free Radicals
Neutral species containing unpaired electrons.
Often formed during homolytic bond cleavage.
9. Reaction Energy Diagrams

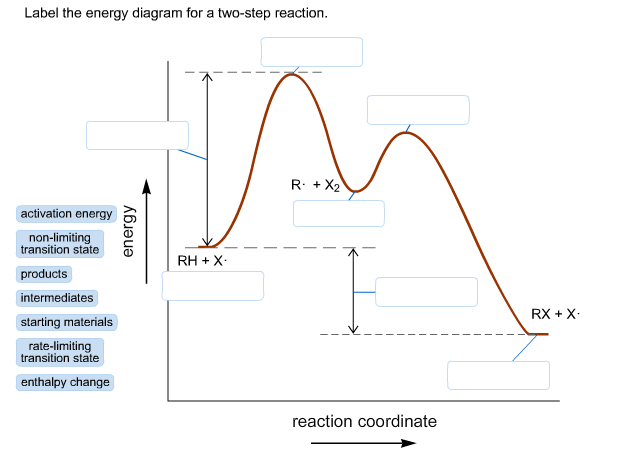


Energy diagrams illustrate how energy changes during reactions.
Important features include:
- Reactants
- Transition states
- Intermediates
- Products
Activation energy determines reaction rate.
Lower activation energy leads to faster reactions.
10. Stereochemistry in Reaction Mechanisms
Reaction mechanisms often involve stereochemical changes.
Examples include:
Inversion of Configuration
Occurs in SN2 reactions.
Racemization
Occurs in SN1 reactions.
Understanding stereochemistry is crucial for pharmaceutical synthesis.
11. Reaction Kinetics and Mechanisms
Reaction mechanisms are closely related to chemical kinetics.
The rate-determining step is the slowest step in the mechanism.
This step controls the overall reaction rate.
Studying kinetics helps identify which mechanism occurs.
12. Organic Catalysis
Catalysts accelerate organic reactions.
Examples include:
- Acid catalysis
- Base catalysis
- Enzyme catalysis
Catalysts lower activation energy and allow reactions to proceed more efficiently.
13. Applications of Reaction Mechanisms
Understanding reaction mechanisms helps chemists:
- Design efficient chemical syntheses
- Develop new pharmaceuticals
- Predict reaction outcomes
- Optimize industrial chemical processes
Mechanistic studies are essential in modern organic chemistry research.
14. Importance of Organic Reaction Mechanisms
Reaction mechanisms provide deep insights into chemical transformations.
They help scientists understand:
- Molecular behavior
- Reaction pathways
- Intermediate species
- Energy changes during reactions
This knowledge allows chemists to control and manipulate chemical reactions.
Conclusion
Organic reaction mechanisms describe the detailed pathways through which organic reactions occur. By studying electron movement, bond breaking, and intermediate formation, chemists can understand how molecules transform during chemical reactions. Major types of mechanisms include substitution, addition, elimination, and rearrangement reactions. These processes involve reactive intermediates such as carbocations, carbanions, and free radicals. Energy diagrams help visualize reaction progress and activation energy barriers. Understanding reaction mechanisms is essential for predicting reaction outcomes, designing chemical syntheses, and advancing pharmaceutical and industrial chemistry.
