



1. Introduction to Chromatography
Chromatography is an important analytical technique used to separate, identify, and purify components in a mixture. It is widely used in chemistry, biology, medicine, environmental science, and the pharmaceutical industry. The technique works by passing a mixture dissolved in a fluid (called the mobile phase) through a material that remains fixed (called the stationary phase). The different components of the mixture move at different speeds through the stationary phase, resulting in separation.
The word chromatography originates from the Greek words:
- chroma meaning color
- graphein meaning to write
The technique was first developed in 1903 by the Russian botanist Mikhail Tswett, who used it to separate plant pigments such as chlorophyll and carotenoids.
Chromatography has become one of the most essential techniques in analytical chemistry because it allows scientists to analyze complex mixtures with high precision and sensitivity.
Applications of chromatography include:
- Drug analysis in pharmaceuticals
- Detection of pollutants in environmental samples
- Food quality control
- Forensic investigations
- Biochemical research
- Purification of biological molecules
Chromatography is therefore considered a fundamental tool for chemical analysis and scientific research.
2. Basic Principles of Chromatography
Chromatography is based on the distribution of components between two phases.
These two phases are:
Stationary Phase
The stationary phase is the solid or liquid phase that remains fixed in place.
Examples include:
- Paper
- Silica gel
- Alumina
- Polymer resins
Mobile Phase
The mobile phase is the fluid that moves through the stationary phase.
It may be:
- Liquid
- Gas
The mobile phase carries the mixture through the stationary phase.
Separation Mechanism
The separation occurs because different components interact differently with the stationary phase.
Some compounds:
- Move quickly with the mobile phase
- Interact weakly with the stationary phase
Other compounds:
- Move more slowly
- Interact strongly with the stationary phase
These differences result in separation.
3. Components of a Chromatographic System
A chromatographic system generally includes several key components.
Sample
The mixture of substances to be separated.
Stationary Phase
The material that remains fixed and interacts with the sample components.
Mobile Phase
The solvent or gas that transports the sample through the stationary phase.
Detector
The device that identifies and measures separated compounds.
Column or Support
The structure that holds the stationary phase.
4. Types of Chromatography
Chromatography can be classified in several ways depending on the physical state of the mobile phase or the separation mechanism.
Major types include:
- Paper chromatography
- Thin-layer chromatography (TLC)
- Column chromatography
- Gas chromatography (GC)
- High-performance liquid chromatography (HPLC)
- Ion exchange chromatography
- Gel filtration chromatography
5. Paper Chromatography


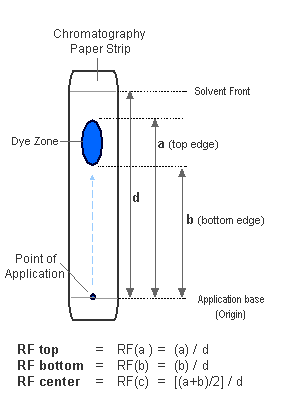

Paper chromatography is one of the simplest forms of chromatography.
In this technique:
- Paper acts as the stationary phase.
- A solvent acts as the mobile phase.
The mixture is placed as a small spot near the bottom of the paper.
As the solvent moves upward through the paper by capillary action, different compounds move at different rates and separate into distinct spots.
Retention Factor (Rf)
Rf value is used to identify compounds.
[
Rf = \frac{\text{Distance traveled by compound}}{\text{Distance traveled by solvent}}
]
Rf values help compare substances in chromatography experiments.
6. Thin-Layer Chromatography (TLC)



Thin-layer chromatography uses a thin layer of adsorbent material such as silica gel or alumina coated on a glass or plastic plate.
Steps include:
- Sample applied near bottom of plate
- Plate placed in solvent chamber
- Solvent rises through plate
- Components separate into spots
TLC is widely used in organic chemistry laboratories for:
- Monitoring reaction progress
- Identifying compounds
- Checking purity
7. Column Chromatography



Column chromatography uses a column packed with stationary phase material.
The sample mixture is introduced at the top of the column.
A solvent passes through the column and carries components downward.
Different compounds move at different speeds, resulting in separation into bands.
The separated components are collected as fractions.
Column chromatography is widely used for purification of organic compounds.
8. Gas Chromatography (GC)

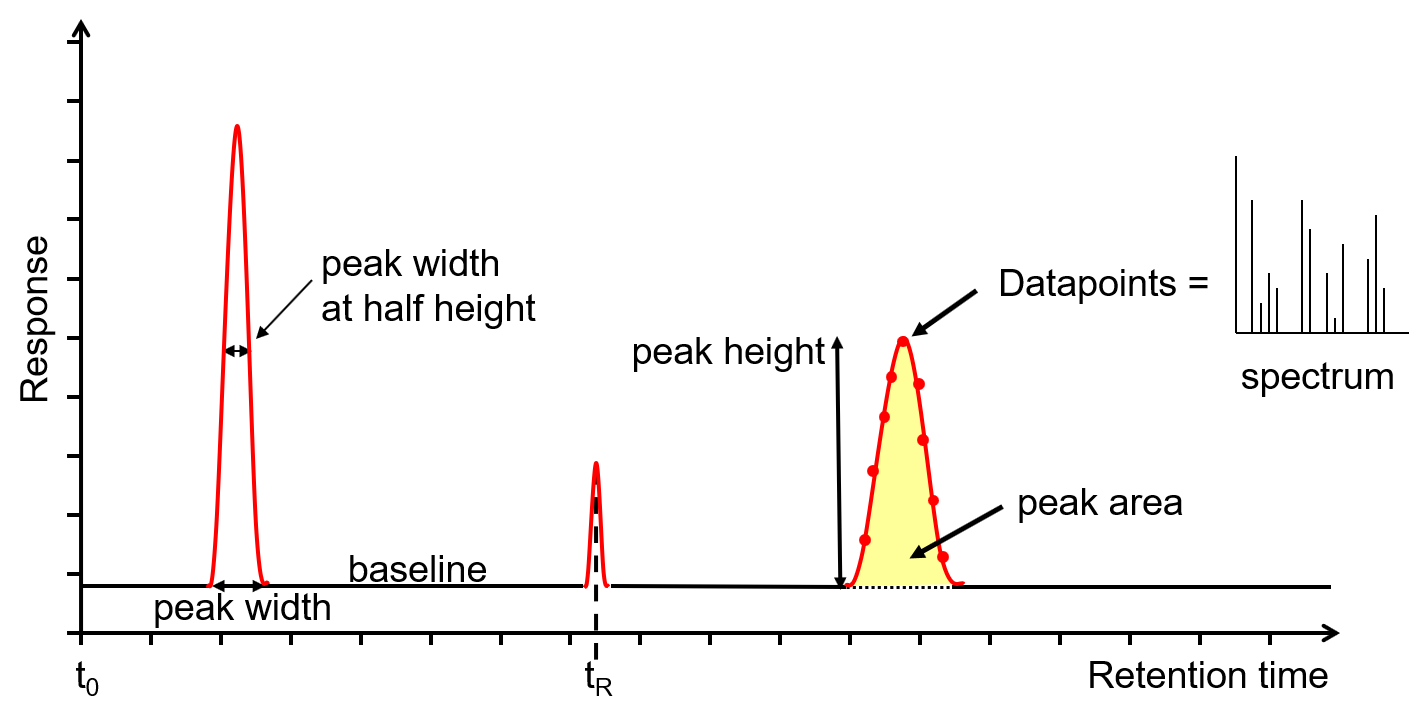


Gas chromatography is used to separate volatile compounds.
In GC:
- Mobile phase = inert carrier gas (helium or nitrogen)
- Stationary phase = liquid or solid coating inside column
Components vaporize and travel through the column.
Different compounds reach the detector at different times.
These times are called retention times.
GC is widely used in:
- Environmental monitoring
- Forensic analysis
- Food analysis
- Petrochemical industries
9. High-Performance Liquid Chromatography (HPLC)
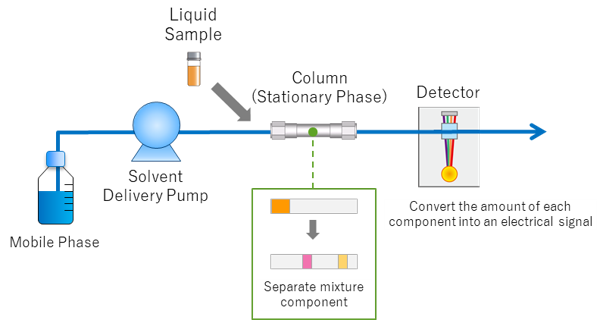



HPLC is an advanced form of liquid chromatography.
It uses high pressure to push solvents through tightly packed columns.
HPLC provides:
- High resolution
- Fast separation
- High sensitivity
It is widely used in:
- Pharmaceutical analysis
- Biochemistry
- Clinical diagnostics
- Food safety testing
10. Ion Exchange Chromatography
Ion exchange chromatography separates molecules based on charge differences.
Stationary phase contains charged groups.
Two types include:
- Cation exchange chromatography
- Anion exchange chromatography
Applications include purification of proteins and nucleic acids.
11. Gel Filtration Chromatography
Gel filtration chromatography separates molecules based on size.
Also called size exclusion chromatography.
Large molecules pass through quickly.
Small molecules enter pores and move more slowly.
This technique is widely used in biochemistry.
12. Applications of Chromatography
Chromatography is used in many fields.
Pharmaceutical Industry
Drug development and quality control.
Environmental Science
Detection of pollutants in air and water.
Food Industry
Analysis of additives and contaminants.
Forensic Science
Detection of drugs and toxins.
Biochemistry
Purification of proteins and DNA.
13. Advantages of Chromatography
Chromatography offers many advantages.
- High sensitivity
- Accurate separation
- Ability to analyze complex mixtures
- Applicable to many types of compounds
Because of these advantages, chromatography is widely used in scientific research.
14. Limitations of Chromatography
Despite its usefulness, chromatography has some limitations.
- Expensive instrumentation for advanced techniques
- Requires skilled operation
- Some methods require careful sample preparation
Nevertheless, it remains one of the most powerful analytical tools.
15. Importance of Chromatography
Chromatography is essential for modern analytical chemistry.
It allows scientists to:
- Separate complex mixtures
- Identify unknown compounds
- Purify chemical substances
- Monitor chemical reactions
The technique continues to evolve with advances in instrumentation and methodology.
Conclusion
Chromatography is a versatile analytical technique used to separate and analyze mixtures based on differences in interactions between components and stationary and mobile phases. Since its discovery by Mikhail Tswett, chromatography has become one of the most important methods in chemistry and related sciences. Various forms of chromatography, including paper chromatography, thin-layer chromatography, gas chromatography, and high-performance liquid chromatography, provide powerful tools for analyzing complex chemical systems. Chromatography has numerous applications in pharmaceuticals, environmental monitoring, food safety, forensic science, and biochemical research. As technology advances, chromatography continues to play a crucial role in scientific discovery and industrial processes.
