


1. Introduction to Photochemistry
Photochemistry is the branch of chemistry that studies chemical reactions initiated by light or electromagnetic radiation. These reactions occur when molecules absorb photons and undergo electronic transitions that lead to chemical transformations.
Light energy can cause molecules to enter excited states, enabling reactions that would not occur under normal thermal conditions. Photochemical processes are fundamental to many natural phenomena and technological applications.
Photochemistry plays a critical role in several important processes such as:
- Photosynthesis in plants
- Vision in animals
- Atmospheric reactions leading to ozone formation and photochemical smog
- Photodegradation of pollutants
- Photolithography in semiconductor manufacturing
Because light energy can trigger chemical reactions in a highly controlled manner, photochemistry has become an important area of research in fields including materials science, environmental chemistry, medicine, and nanotechnology.
2. Nature of Light and Photons
Photochemical reactions begin when molecules absorb electromagnetic radiation. Light consists of photons, which are packets of energy.
The energy of a photon is given by the equation:
E = hν
Where:
- E = energy of photon
- h = Planck’s constant
- ν = frequency of radiation
Because frequency is related to wavelength, the equation can also be written as:
E = hc/λ
Where:
- c = speed of light
- λ = wavelength
Shorter wavelengths correspond to higher energy photons.
Photochemical reactions usually involve ultraviolet or visible light because these wavelengths have sufficient energy to promote electronic transitions.
3. Absorption of Light




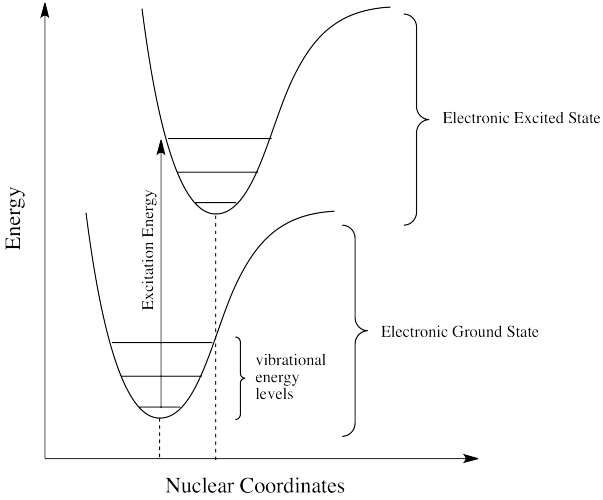
When molecules absorb light, electrons are promoted from lower-energy orbitals to higher-energy orbitals.
The molecule moves from its ground state to an excited state.
Electronic transitions may involve:
- σ → σ* transitions
- π → π* transitions
- n → π* transitions
The excited molecule contains excess energy and is therefore highly reactive.
After excitation, the molecule may undergo several processes including emission of light, chemical reaction, or energy transfer.
4. Laws of Photochemistry
Two fundamental laws govern photochemical reactions.
Grotthuss–Draper Law
This law states that only light that is absorbed by a substance can cause a photochemical reaction.
If light passes through a substance without being absorbed, no photochemical change occurs.
Stark–Einstein Law
Also known as the photochemical equivalence law.
This law states that each photon absorbed by a molecule activates one molecule for reaction.
The relationship between absorbed photons and reacting molecules is fundamental to photochemical processes.
5. Electronic Excited States




When molecules absorb light, they enter excited electronic states.
These states include:
Singlet States
Electrons remain paired with opposite spins.
These states have relatively short lifetimes.
Triplet States
Electrons have parallel spins.
Triplet states are longer-lived and often participate in photochemical reactions.
Jablonski Diagram
The Jablonski diagram illustrates the transitions between energy states.
It includes processes such as:
- Absorption
- Fluorescence
- Phosphorescence
- Intersystem crossing
This diagram helps visualize the pathways involved in photochemical reactions.
6. Photophysical Processes
Photophysical processes involve changes in energy states without chemical reactions.
Examples include:
Fluorescence
Emission of light from excited singlet states.
Occurs rapidly after absorption.
Phosphorescence
Emission from triplet states.
Occurs more slowly than fluorescence.
These processes are widely used in fluorescence spectroscopy and imaging techniques.
7. Photochemical Reactions




Photochemical reactions involve chemical transformations triggered by light.
Examples include:
Photodissociation
Light energy breaks chemical bonds.
Example:
O₂ → 2O
Photoisomerization
Molecules change structure when exposed to light.
Example:
Cis–trans isomerization of alkenes.
Photoreduction and Photooxidation
Light-induced oxidation–reduction reactions occur in many biological and environmental processes.
8. Quantum Yield
Quantum yield measures the efficiency of a photochemical reaction.
It is defined as:
Quantum yield = (Number of molecules reacting) / (Number of photons absorbed)
A quantum yield greater than one indicates chain reactions.
Quantum yield provides important information about reaction mechanisms.
9. Photochemistry in the Atmosphere
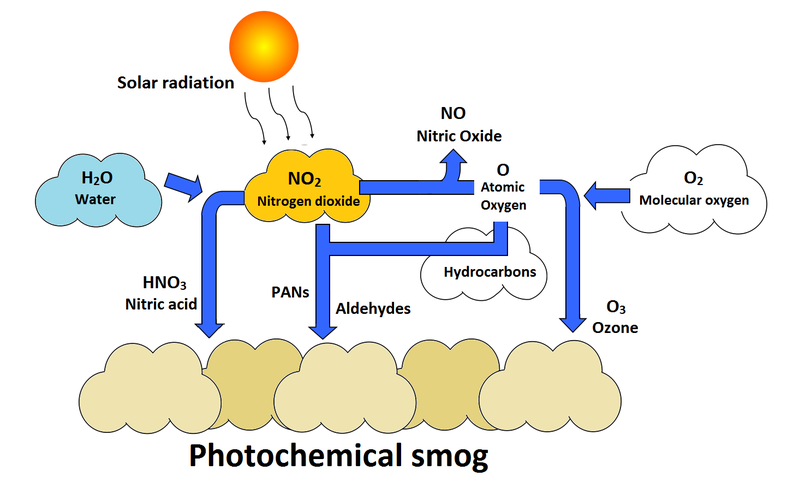



Photochemical reactions occur extensively in Earth’s atmosphere.
Examples include:
Ozone Formation
UV radiation splits oxygen molecules, producing atomic oxygen that forms ozone.
Photochemical Smog
Smog forms when sunlight triggers reactions between nitrogen oxides and hydrocarbons.
This produces harmful pollutants such as ozone and peroxyacyl nitrates.
Photochemistry therefore plays a major role in air pollution and atmospheric chemistry.
10. Photochemistry in Biological Systems




Photochemical reactions are essential for life.
Examples include:
Photosynthesis
Plants absorb sunlight using chlorophyll and convert it into chemical energy.
Vision
The visual process begins when light causes a structural change in retinal molecules in the eye.
These reactions initiate nerve signals that allow vision.
11. Applications of Photochemistry
Photochemistry has numerous technological applications.
Solar Energy
Solar cells convert sunlight into electrical energy.
Photography
Photochemical reactions form images on photographic film.
Photopolymerization
Light is used to initiate polymer formation in coatings and 3D printing.
Medical Applications
Photodynamic therapy uses light to activate drugs that destroy cancer cells.
12. Environmental Applications
Photochemistry helps break down pollutants in the environment.
Sunlight can cause degradation of harmful chemicals in air and water.
Photocatalysts such as titanium dioxide are used in environmental cleanup processes.
13. Importance of Photochemistry
Photochemistry is essential for understanding how light interacts with matter.
It provides insight into:
- Atmospheric chemistry
- Biological processes
- Renewable energy technologies
- Environmental protection
Because sunlight drives many natural processes on Earth, photochemistry is fundamental to life and environmental science.
Conclusion
Photochemistry is the study of chemical reactions that occur when molecules absorb light energy. When photons interact with molecules, electrons are excited to higher energy levels, creating reactive species that can undergo various chemical transformations. Fundamental laws such as the Grotthuss–Draper law and the Stark–Einstein law describe the principles governing photochemical reactions. Processes such as fluorescence, phosphorescence, and photodissociation are important aspects of photochemistry. Photochemical reactions occur in many natural systems including the atmosphere and biological organisms. They are also widely used in technologies such as solar energy conversion, photography, and medical treatments. Photochemistry therefore plays a vital role in both natural phenomena and modern technological applications.
