


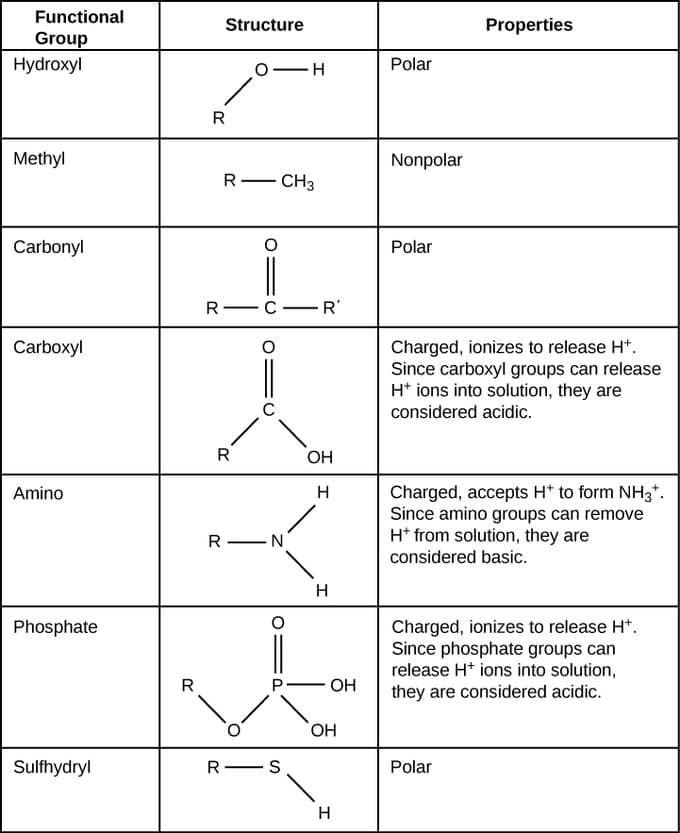
1. Introduction to Functional Groups
Functional groups are specific groups of atoms within molecules that determine the chemical properties, reactivity, and behavior of organic compounds. They are the key structural features that define different classes of organic molecules. In organic chemistry, compounds with similar functional groups tend to exhibit similar chemical reactions and physical properties.
A functional group typically consists of a specific arrangement of atoms attached to a carbon skeleton. The carbon framework may vary in size and structure, but the presence of a particular functional group gives the compound its characteristic properties.
For example:
- The –OH group defines alcohols.
- The –COOH group defines carboxylic acids.
- The –CHO group defines aldehydes.
Functional groups are extremely important because they allow chemists to categorize millions of organic compounds into manageable families.
Understanding functional groups is essential for:
- Predicting chemical reactions
- Identifying organic compounds
- Designing pharmaceuticals
- Synthesizing new materials
- Studying biochemical processes
Functional groups serve as the foundation of organic chemistry and are widely used in chemistry, biology, medicine, and materials science.
2. Role of Functional Groups in Organic Molecules
Functional groups determine several important properties of organic molecules.
Chemical Reactivity
The functional group is usually the most reactive part of the molecule. It participates directly in chemical reactions.
Physical Properties
Functional groups influence:
- Boiling point
- Solubility
- Polarity
- Melting point
Molecular Interactions
Functional groups affect how molecules interact through hydrogen bonding and other intermolecular forces.
Biological Activity
Many biological molecules depend on specific functional groups for their activity.
For example:
- Amino groups in amino acids
- Phosphate groups in DNA
3. Classification of Functional Groups
Functional groups can be classified into several major categories:
- Hydrocarbon functional groups
- Oxygen-containing functional groups
- Nitrogen-containing functional groups
- Sulfur-containing functional groups
- Halogen-containing functional groups
Each group has characteristic properties and reactions.
4. Hydrocarbon Functional Groups




Hydrocarbon functional groups consist of carbon and hydrogen atoms only.
Alkanes
Alkanes contain only single carbon–carbon bonds.
Example:
Methane (CH₄)
Properties:
- Low reactivity
- Nonpolar molecules
Alkenes
Alkenes contain carbon–carbon double bonds.
Example:
Ethene (C₂H₄)
Reactivity:
- Undergo addition reactions.
Alkynes
Alkynes contain carbon–carbon triple bonds.
Example:
Ethyne (C₂H₂)
They are more reactive due to triple bonds.
5. Oxygen-Containing Functional Groups




Oxygen-containing functional groups are very common in organic chemistry.
Alcohols (–OH)
Alcohols contain the hydroxyl group.
Example:
Ethanol (C₂H₅OH)
Properties:
- Polar molecules
- Hydrogen bonding
- Higher boiling points
Applications:
- Solvents
- Fuels
- Antiseptics
Ethers (–O–)
Ethers contain an oxygen atom between two carbon atoms.
Example:
Diethyl ether.
Used as solvents and anesthetics.
Aldehydes (–CHO)
Aldehydes contain a carbonyl group bonded to hydrogen.
Example:
Formaldehyde.
Used in preservatives and plastics.
Ketones (–CO–)
Ketones contain a carbonyl group bonded to two carbon atoms.
Example:
Acetone.
Widely used as a solvent.
Carboxylic Acids (–COOH)
Carboxylic acids contain the carboxyl group.
Example:
Acetic acid.
Properties:
- Acidic nature
- Hydrogen bonding
Used in food preservation and chemical synthesis.
Esters (–COOR)
Esters form from reactions between alcohols and carboxylic acids.
Example:
Ethyl acetate.
Many esters have pleasant fruity smells.
Used in:
- Flavors
- Perfumes
- Solvents
6. Nitrogen-Containing Functional Groups



Nitrogen-containing functional groups play important roles in biological chemistry.
Amines (–NH₂)
Amines are derivatives of ammonia.
Example:
Methylamine.
Used in pharmaceuticals and dyes.
Amides (–CONH₂)
Amides contain a carbonyl group attached to nitrogen.
Example:
Peptide bonds in proteins.
Amides are important in biological molecules.
Nitriles (–CN)
Nitriles contain a carbon–nitrogen triple bond.
Example:
Acetonitrile.
Used as solvents and chemical intermediates.
7. Sulfur-Containing Functional Groups



Sulfur-containing groups appear in many biological molecules.
Thiols (–SH)
Also called mercaptans.
Example:
Cysteine amino acid.
Important in protein structure.
Sulfides
Contain sulfur between two carbon atoms.
Example:
Dimethyl sulfide.
8. Halogen-Containing Functional Groups



Halogen-containing compounds include chlorine, bromine, iodine, or fluorine atoms attached to carbon.
Example:
Chloroform (CHCl₃)
Applications:
- Solvents
- Refrigerants
- Pharmaceuticals
9. Importance of Functional Groups in Organic Reactions
Functional groups determine how molecules react chemically.
Examples:
- Alcohol oxidation
- Ester hydrolysis
- Amide formation
- Substitution reactions
Functional groups allow chemists to predict reaction mechanisms and outcomes.
10. Functional Groups in Biological Molecules
Functional groups are essential components of biomolecules.
Examples include:
Carbohydrates
Contain hydroxyl and carbonyl groups.
Proteins
Contain amino and carboxyl groups.
DNA
Contains phosphate groups and nitrogen bases.
Lipids
Contain ester functional groups.
Functional groups determine the biological function of these molecules.
11. Industrial Applications of Functional Groups
Functional groups are used extensively in chemical industries.
Examples include:
- Pharmaceutical drug synthesis
- Polymer production
- Agricultural chemicals
- Dye manufacturing
- Cosmetic ingredients
Many industrial reactions are designed specifically to modify functional groups.
12. Importance of Functional Groups in Organic Chemistry
Functional groups allow chemists to:
- Classify organic compounds
- Predict chemical reactions
- Understand molecular structure
- Design synthetic pathways
They are the fundamental building blocks for studying organic chemistry.
Conclusion
Functional groups are specific groups of atoms within organic molecules that determine their chemical properties and reactivity. They allow chemists to classify and understand the behavior of millions of organic compounds. Major functional groups include hydrocarbons, oxygen-containing groups, nitrogen-containing groups, sulfur-containing groups, and halogen-containing groups. These groups influence molecular structure, chemical reactions, physical properties, and biological activity. Functional groups play a crucial role in industrial chemistry, pharmaceuticals, biochemistry, and materials science, making them one of the most fundamental concepts in organic chemistry.
