Introduction
In thermodynamics and chemistry, enthalpy is a fundamental concept used to describe the heat energy of a system. It is especially important in studying chemical reactions and physical changes, where energy is either released or absorbed.
When substances undergo chemical reactions or phase transitions, energy changes occur in the form of heat. These heat changes help scientists understand whether a reaction will release energy to the surroundings or absorb energy from the environment.
Enthalpy provides a convenient way to measure these energy changes when reactions occur at constant pressure, which is the condition under which most chemical reactions take place in open systems such as laboratories and natural environments.
The study of enthalpy and heat changes helps scientists analyze reaction energetics, predict reaction behavior, design industrial processes, and understand biological systems. It is also essential for fields such as materials science, environmental chemistry, chemical engineering, and biochemistry.
1. Concept of Enthalpy
Definition of Enthalpy
Enthalpy is defined as the total heat content of a system.
Mathematically, enthalpy is expressed as:
H = U + PV
Where:
H = Enthalpy
U = Internal energy of the system
P = Pressure
V = Volume
This equation indicates that enthalpy includes both the internal energy of the system and the energy associated with pressure and volume.
Enthalpy Change
During chemical reactions or physical processes, the enthalpy of the system may change.
The enthalpy change is represented as:
\Delta H = H_{products} – H_{reactants}
Where:
ΔH = Change in enthalpy
Hproducts = Enthalpy of products
Hreactants = Enthalpy of reactants
A positive ΔH indicates heat absorption, while a negative ΔH indicates heat release.
2. Heat and Energy Transfer
Heat is the transfer of thermal energy from one system to another due to a temperature difference.
In thermodynamics, heat flow occurs between a system and its surroundings.
Heat can move in different directions depending on the temperature difference between the system and its surroundings.
Heat Flow
Heat flows from higher temperature regions to lower temperature regions until thermal equilibrium is reached.
For example:
- A hot object cools down in a cold room.
- Ice melts when placed in warm water.
Units of Heat
Heat energy can be measured using different units:
- Joule (J)
- Kilojoule (kJ)
- Calorie (cal)
Conversion relationship:
1 calorie = 4.184 joules
3. Exothermic and Endothermic Reactions




Chemical reactions are classified according to their heat changes.
Exothermic Reactions
Exothermic reactions release heat to the surroundings.
Characteristics:
- ΔH is negative
- Temperature of surroundings increases
Examples:
- Combustion of fuels
- Respiration
- Formation of ice from water
In energy diagrams, products have lower energy than reactants.
Endothermic Reactions
Endothermic reactions absorb heat from the surroundings.
Characteristics:
- ΔH is positive
- Temperature of surroundings decreases
Examples:
- Photosynthesis
- Melting of ice
- Evaporation of water
In energy diagrams, products have higher energy than reactants.
4. Enthalpy Changes in Chemical Reactions
Several types of enthalpy changes occur in chemical processes.
Enthalpy of Reaction
The enthalpy of reaction is the heat change when a chemical reaction occurs under constant pressure.
It represents the difference in energy between reactants and products.
Enthalpy of Formation
The enthalpy of formation refers to the heat change when one mole of a compound forms from its elements in their standard states.
Example:
Formation of water from hydrogen and oxygen.
Enthalpy of Combustion




The enthalpy of combustion is the heat released when one mole of a substance completely burns in oxygen.
Examples include:
- Burning methane
- Burning gasoline
- Burning coal
Combustion reactions are usually highly exothermic.
Enthalpy of Neutralization
This refers to the heat change when an acid reacts with a base to form water and salt.
Example:
Hydrochloric acid reacting with sodium hydroxide.
Enthalpy of Solution
Enthalpy of solution is the heat change when a solute dissolves in a solvent.
It may be either exothermic or endothermic.
5. Enthalpy Changes in Physical Processes
Enthalpy changes are not limited to chemical reactions. They also occur during physical changes.
Enthalpy of Fusion



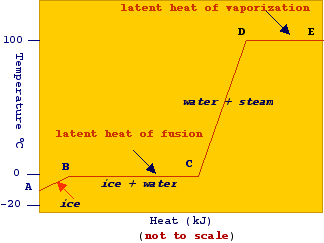
The enthalpy of fusion is the heat required to convert a solid into a liquid at its melting point.
Example:
Ice melting into water.
Enthalpy of Vaporization
The enthalpy of vaporization is the heat required to convert a liquid into vapor at its boiling point.
Example:
Water boiling into steam.
Enthalpy of Sublimation
Sublimation occurs when a solid changes directly into gas.
Example:
Dry ice turning into carbon dioxide gas.
6. Hess’s Law
Hess’s Law states that the total enthalpy change of a reaction is independent of the pathway taken.
In other words, the overall enthalpy change depends only on the initial and final states.
This law allows scientists to calculate enthalpy changes for reactions that are difficult to measure directly.
7. Bond Enthalpy
Bond enthalpy refers to the energy required to break a chemical bond.
Breaking bonds requires energy, while forming bonds releases energy.
The overall enthalpy change of a reaction depends on the balance between these two processes.
8. Calorimetry




Calorimetry is the experimental technique used to measure heat changes during chemical reactions.
The instrument used is called a calorimeter.
Coffee Cup Calorimeter
Used to measure heat changes in reactions occurring in solution.
Typically used in laboratory experiments.
Bomb Calorimeter
Used to measure heat released during combustion reactions.
The reaction occurs in a sealed metal container surrounded by water.
9. Thermochemical Equations
Thermochemical equations represent chemical reactions along with their enthalpy changes.
Example:
Hydrogen + Oxygen → Water + Heat
These equations help scientists quantify energy changes in chemical reactions.
10. Applications of Enthalpy
Enthalpy changes are important in many fields.
Industrial Chemistry
Industries use enthalpy data to design energy-efficient processes.
Examples include:
- Fuel production
- Fertilizer manufacturing
- Petrochemical processing
Energy Production
Combustion reactions release large amounts of heat energy used in power plants.
Environmental Science
Enthalpy changes influence atmospheric processes and climate systems.
Biological Systems
Living organisms rely on energy released from biochemical reactions.
For example:
Cellular respiration releases heat and energy needed for life processes.
11. Importance of Enthalpy in Chemistry
Enthalpy provides insight into how energy changes occur during chemical reactions and physical transformations. By studying enthalpy, chemists can predict reaction behavior, determine whether a reaction releases or absorbs energy, and design chemical processes that maximize efficiency.
Understanding enthalpy changes also allows scientists to study reaction mechanisms, measure bond strengths, and analyze energy flow within complex systems. These insights are essential for developing new fuels, improving industrial processes, and understanding biological energy systems.
Conclusion
Enthalpy and heat changes are central concepts in thermodynamics and chemistry. Enthalpy represents the heat content of a system and allows scientists to measure energy changes during chemical reactions and physical transformations.
Reactions may be classified as exothermic or endothermic depending on whether heat is released or absorbed. Enthalpy changes also occur during processes such as melting, vaporization, combustion, and dissolution.
Principles such as Hess’s Law and bond enthalpy provide tools for calculating energy changes in complex reactions. Experimental techniques like calorimetry allow scientists to measure these heat changes accurately.
The study of enthalpy is essential for understanding energy transformations in nature and technology. From chemical manufacturing and power generation to biological metabolism and environmental processes, enthalpy plays a crucial role in explaining how energy flows through physical and chemical systems.