



1. Introduction to pH and Buffer Solutions
The concepts of pH and buffer solutions are central to acid–base chemistry and play a crucial role in chemistry, biology, medicine, environmental science, and industrial processes. The pH scale provides a quantitative measure of how acidic or basic a solution is, while buffers are systems that help maintain a relatively constant pH when small amounts of acids or bases are added.
Chemical reactions occurring in aqueous solutions often involve hydrogen ions (H⁺) or hydroxide ions (OH⁻). The concentration of these ions determines the acidity or alkalinity of a solution. Maintaining stable pH levels is essential for many natural and industrial processes.
For example:
- The human blood maintains a pH around 7.35–7.45
- Ocean water has a pH around 8.1
- Many enzymes function only within narrow pH ranges
Buffer systems help resist changes in pH and maintain chemical stability.
Understanding pH and buffer solutions is essential in:
- Biological systems
- Medical diagnostics
- Pharmaceutical production
- Environmental monitoring
- Industrial chemistry
- Agricultural science
2. Definition of pH
The pH scale is used to measure the acidity or basicity of a solution.
pH is defined as the negative logarithm of the hydrogen ion concentration.
[
pH = -\log[H^+]
]
Where:
- [H⁺] = concentration of hydrogen ions
The pH scale ranges from 0 to 14.
Classification of solutions:
- pH < 7 → acidic
- pH = 7 → neutral
- pH > 7 → basic (alkaline)
Pure water has a pH of 7, meaning it is neutral.
3. Relationship Between pH and Hydrogen Ion Concentration
Because pH is logarithmic, each unit change represents a tenfold change in hydrogen ion concentration.
Example:
pH = 1 → very strong acid
pH = 2 → ten times weaker than pH 1
pH = 3 → one hundred times weaker than pH 1
Similarly:
pH = 10 is ten times more basic than pH 9.
4. Ionization of Water
Water slightly dissociates into hydrogen ions and hydroxide ions.
[
H_2O \rightleftharpoons H^+ + OH^-
]
At 25°C:
[
[H^+] = [OH^-] = 1 \times 10^{-7}
]
The ionic product of water is:
[
K_w = [H^+][OH^-] = 1 \times 10^{-14}
]
This constant plays a key role in acid–base chemistry.
5. pOH and Relationship with pH
The concentration of hydroxide ions is measured using pOH.
[
pOH = -\log[OH^-]
]
Relationship between pH and pOH:
[
pH + pOH = 14
]
This equation applies at 25°C.
Example:
If pH = 4
pOH = 10
6. Measurement of pH




pH can be measured using several methods.
1. pH Meter
A pH meter measures hydrogen ion activity electronically.
It provides highly accurate results.
2. Indicators
Indicators change color depending on pH.
Examples:
- Litmus
- Phenolphthalein
- Methyl orange
3. Universal Indicator
A mixture of several indicators that shows a full pH range.
4. pH Paper
Special paper impregnated with indicator chemicals.
7. Importance of pH
pH plays a vital role in many chemical and biological processes.
Examples include:
- Enzyme activity
- Soil fertility
- Water quality
- Blood chemistry
- Industrial chemical reactions
Small pH changes can significantly affect chemical reactions and biological systems.
8. Buffer Solutions
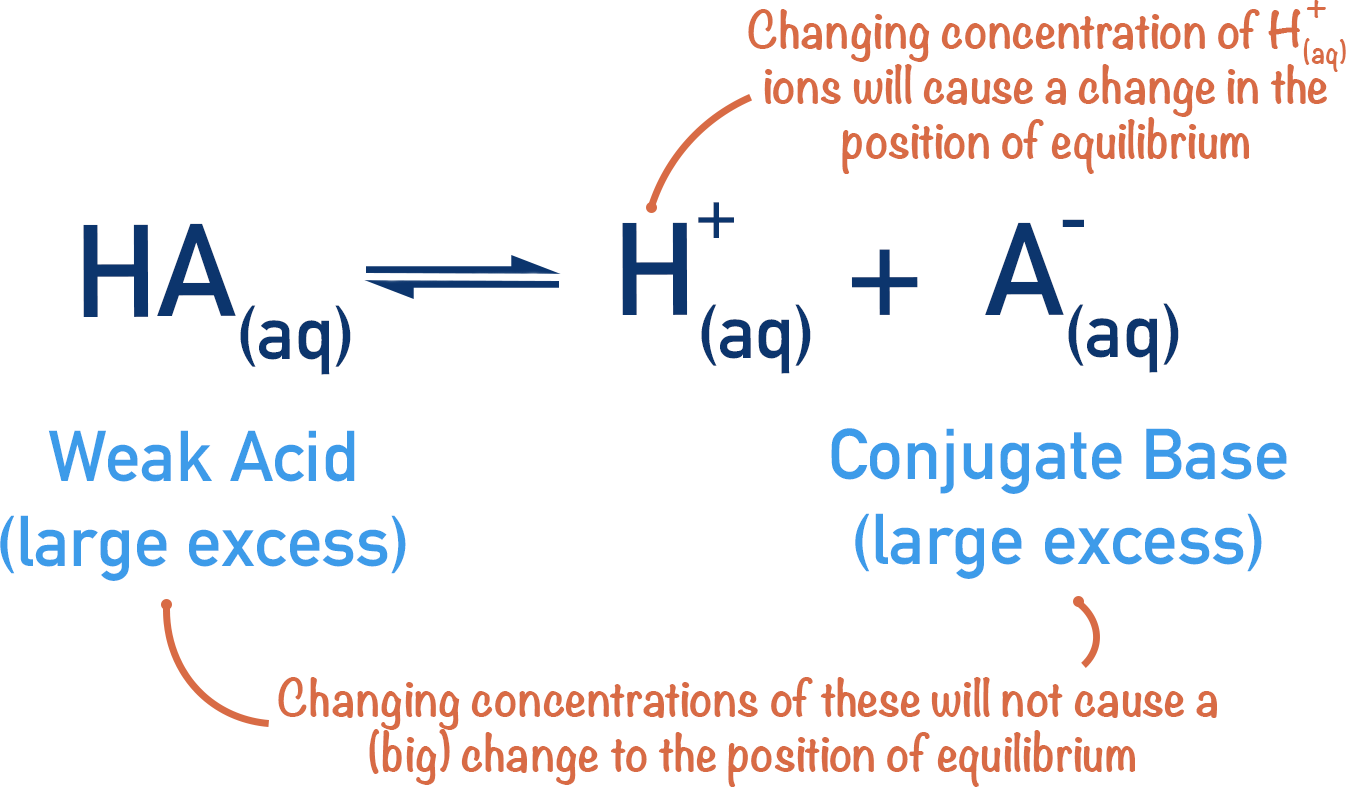


A buffer solution is a solution that resists changes in pH when small amounts of acid or base are added.
Buffers consist of:
- A weak acid and its conjugate base
or - A weak base and its conjugate acid
Buffers maintain pH stability in many chemical and biological systems.
9. Types of Buffer Solutions
Acidic Buffer
Acidic buffers contain:
- Weak acid
- Salt of the acid
Example:
Acetic acid + sodium acetate.
This buffer maintains pH below 7.
Basic Buffer
Basic buffers contain:
- Weak base
- Salt of the base
Example:
Ammonia + ammonium chloride.
This buffer maintains pH above 7.
10. Mechanism of Buffer Action
Buffers resist pH change through reactions involving weak acids and their conjugate bases.
Example acidic buffer:
CH₃COOH ⇌ H⁺ + CH₃COO⁻
When acid is added:
CH₃COO⁻ + H⁺ → CH₃COOH
When base is added:
CH₃COOH + OH⁻ → CH₃COO⁻ + H₂O
Thus the buffer neutralizes added acid or base.
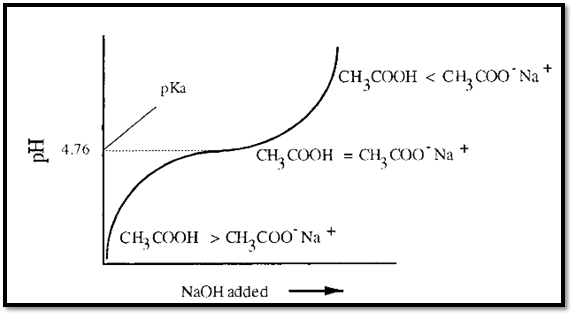
11. Henderson–Hasselbalch Equation
The Henderson–Hasselbalch equation relates buffer pH to acid and conjugate base concentrations.
[
pH = pK_a + \log \frac{[A^-]}{[HA]}
]
Where:
- pKa = acid dissociation constant
- [A⁻] = conjugate base concentration
- [HA] = acid concentration
This equation is widely used in buffer calculations.
12. Buffer Capacity
Buffer capacity refers to the ability of a buffer to resist pH change.
Factors affecting buffer capacity include:
- Concentration of buffer components
- Ratio of acid to conjugate base
- Strength of weak acid/base
Buffers work best when:
pH ≈ pKa
13. Biological Buffers

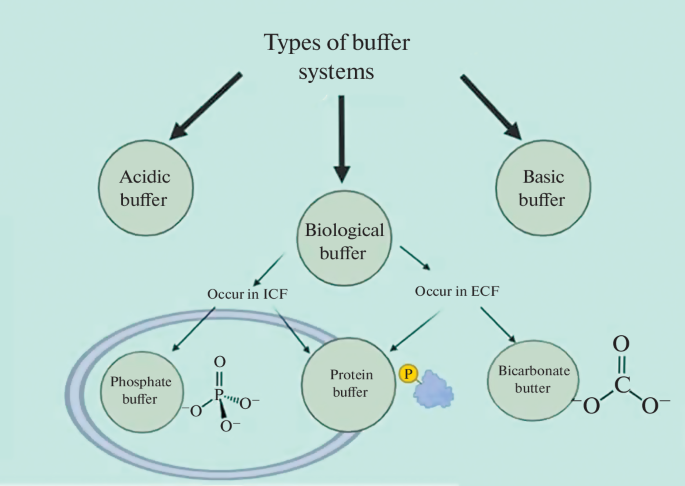
Buffer systems are essential in biological systems.
The human body contains several important buffers.
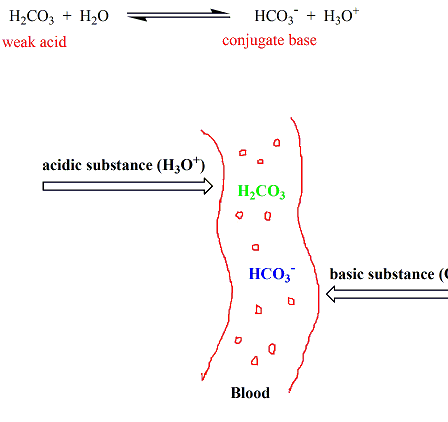
Bicarbonate Buffer System
This is the primary buffer in blood.
CO₂ + H₂O ⇌ H₂CO₃ ⇌ H⁺ + HCO₃⁻
This system maintains blood pH around 7.4.
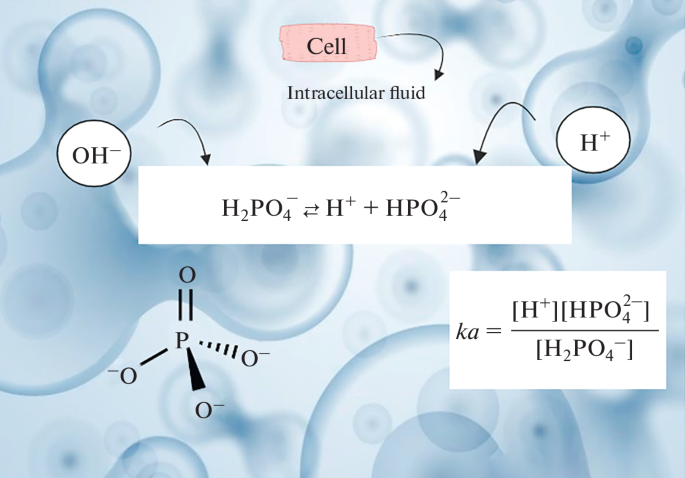
Phosphate Buffer System
Important inside cells.
H₂PO₄⁻ ⇌ H⁺ + HPO₄²⁻
Protein Buffer System
Proteins can accept or donate protons.
Hemoglobin acts as a buffer in blood.
14. Industrial Applications of Buffers
Buffers are used in many industries.
Examples include:
- Pharmaceutical manufacturing
- Food processing
- Chemical synthesis
- Dye production
- Cosmetic manufacturing
Buffers help maintain stable chemical conditions during reactions.
15. Environmental Importance of pH
pH affects many environmental processes.
Examples include:
- Ocean acidity
- Soil fertility
- Water quality
- Acid rain formation
Monitoring pH helps scientists evaluate environmental health.
16. pH in Agriculture
Soil pH determines nutrient availability for plants.
Most crops grow best in slightly acidic soil.
Soil pH affects:
- Fertilizer effectiveness
- Microbial activity
- Plant growth
Farmers often adjust soil pH using lime or sulfur.
17. pH in Medicine
pH plays a crucial role in medicine.
Examples include:
- Blood pH regulation
- Drug stability
- Enzyme activity
- Digestive processes
Abnormal pH levels in blood can cause serious health problems.
18. Importance of Buffers in Chemistry
Buffers are essential for maintaining stable chemical environments.
They allow reactions to occur under controlled pH conditions.
Buffers are critical in:
- Biochemistry
- Analytical chemistry
- Pharmaceutical research
- Industrial chemistry
Conclusion
The concepts of pH and buffer solutions are fundamental to acid–base chemistry and play a crucial role in maintaining chemical balance in many systems. The pH scale provides a quantitative measure of acidity and alkalinity, while buffer solutions help stabilize pH against changes caused by added acids or bases. From biological systems such as blood buffering to industrial chemical processes and environmental monitoring, pH control is essential for maintaining stability and ensuring proper chemical functioning. Understanding these concepts allows scientists to analyze chemical reactions, design stable solutions, and maintain equilibrium in natural and artificial systems.
