


Introduction
Thermodynamic potentials are important functions used in thermodynamics to describe the energy of a system under different physical conditions. These potentials provide powerful tools for analyzing equilibrium, energy transformations, and spontaneous processes in thermodynamic systems.
In thermodynamics, systems can exchange energy in several forms, such as heat, work, and matter. Thermodynamic potentials help determine how energy is stored and how systems evolve toward equilibrium. They are particularly useful because they incorporate variables such as temperature, pressure, entropy, and volume into a single mathematical function.
The main thermodynamic potentials include:
- Internal Energy (U)
- Enthalpy (H)
- Helmholtz Free Energy (F)
- Gibbs Free Energy (G)
Each potential is useful under different experimental conditions. For example, the Gibbs free energy is widely used in chemistry and materials science to predict whether a reaction will occur spontaneously.
Thermodynamic potentials are derived using Legendre transformations, which allow energy functions to be expressed in terms of different sets of natural variables.
These potentials play a central role in many areas of physics, chemistry, and engineering, including:
- Chemical thermodynamics
- Phase transitions
- Statistical mechanics
- Material science
Understanding thermodynamic potentials allows scientists to analyze energy transformations and predict equilibrium states of systems.
Internal Energy



Internal energy is the total energy contained within a thermodynamic system.
It includes:
- Kinetic energy of molecules
- Potential energy of molecular interactions
Internal energy is usually represented by the symbol U.
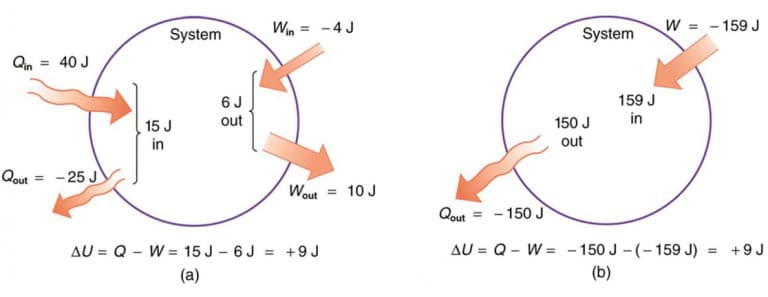
The change in internal energy is described by the first law of thermodynamics:
[
dU = \delta Q – \delta W
]
Where:
- (Q) = heat added to the system
- (W) = work done by the system
Internal energy depends on variables such as entropy and volume:
[
U = U(S, V)
]
Internal energy is the fundamental thermodynamic potential from which other potentials are derived.
Enthalpy


Enthalpy is another thermodynamic potential that accounts for energy associated with pressure and volume.
It is defined as:
[
H = U + PV
]
Where:
- (H) = enthalpy
- (U) = internal energy
- (P) = pressure
- (V) = volume
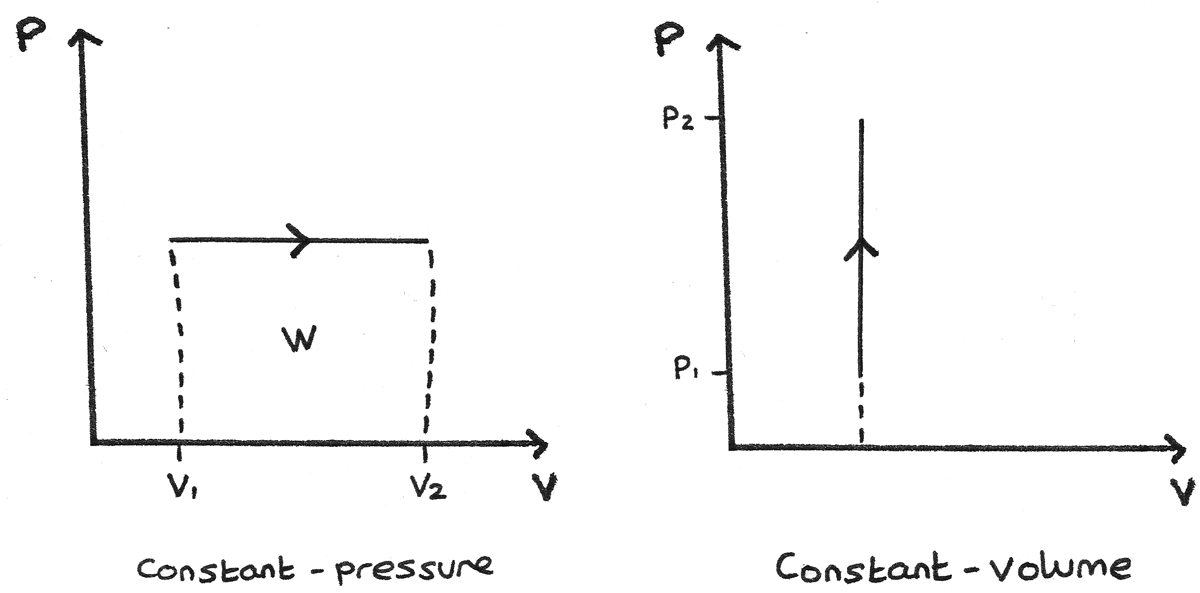
Enthalpy is particularly useful in processes occurring at constant pressure, which are common in many chemical and physical systems.
The change in enthalpy represents the heat absorbed or released at constant pressure.
Examples include:
- Chemical reactions
- Phase changes
- Combustion processes
Helmholtz Free Energy


The Helmholtz free energy is defined as:
[
F = U – TS
]
Where:
- (F) = Helmholtz free energy
- (T) = temperature
- (S) = entropy
Helmholtz free energy represents the amount of useful work obtainable from a system at constant temperature and volume.
It is widely used in:
- Statistical mechanics
- Solid-state physics
- Thermodynamic modeling of materials
At equilibrium, Helmholtz free energy reaches a minimum.
Gibbs Free Energy



The Gibbs free energy is one of the most important thermodynamic potentials.
It is defined as:
[
G = U + PV – TS
]
Or equivalently:
[
G = H – TS
]
Where:
- (G) = Gibbs free energy
Gibbs free energy is especially useful for systems at constant temperature and pressure, which are common in laboratory and natural environments.
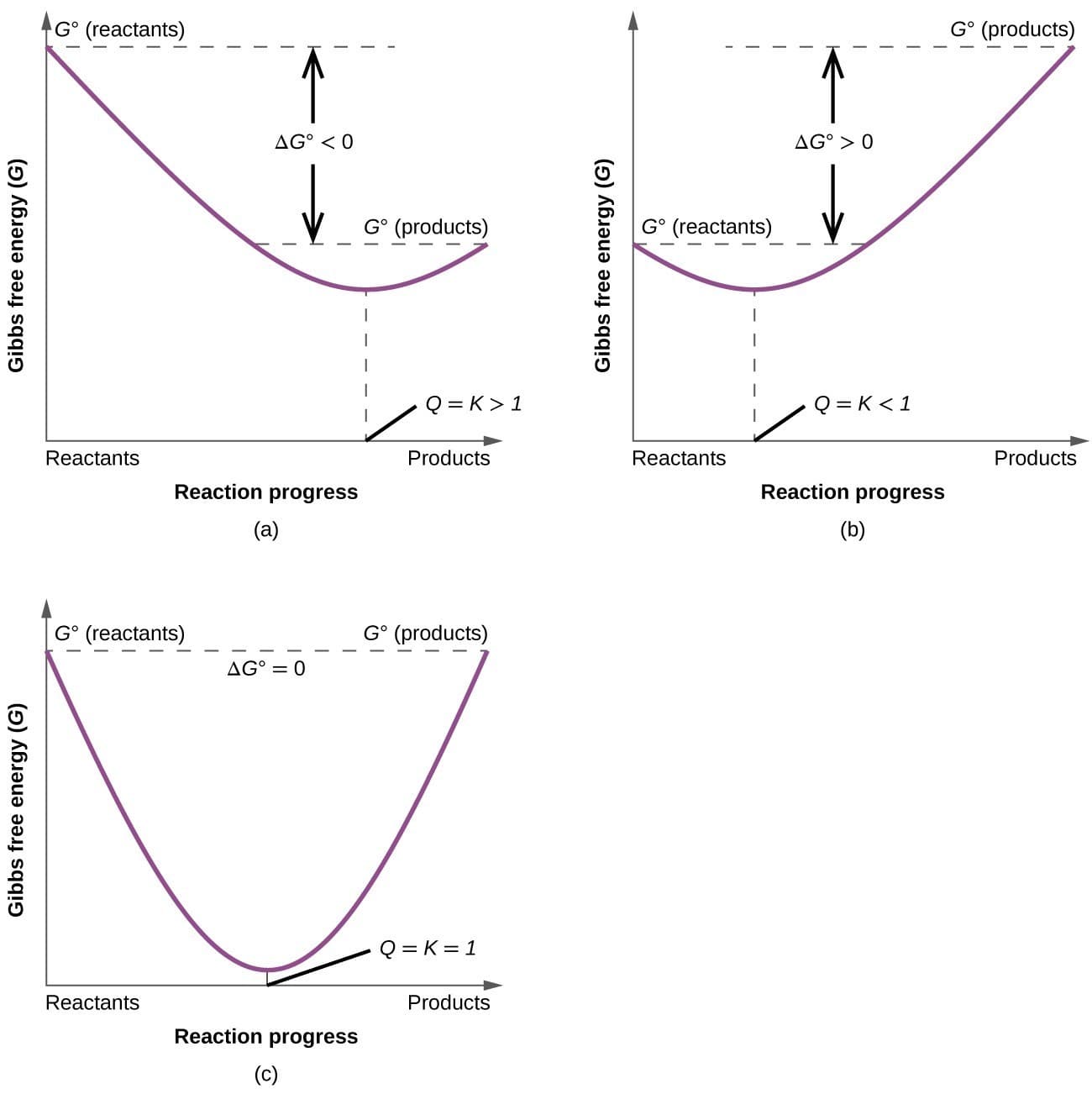
A process occurs spontaneously when:
[
\Delta G < 0
]
Examples include:
- Chemical reactions
- Phase transitions
- Biological processes
Natural Variables of Thermodynamic Potentials




Each thermodynamic potential has its own natural variables.
| Potential | Natural Variables |
|---|---|
| Internal Energy (U) | Entropy, Volume |
| Enthalpy (H) | Entropy, Pressure |
| Helmholtz Free Energy (F) | Temperature, Volume |
| Gibbs Free Energy (G) | Temperature, Pressure |
These variables determine the conditions under which the potential is most useful.
Legendre Transformations



Thermodynamic potentials are related through Legendre transformations.
Legendre transformations allow energy functions to be rewritten in terms of new variables.
For example:
- Internal energy uses entropy as a variable.
- Helmholtz free energy replaces entropy with temperature.
These transformations simplify the analysis of thermodynamic systems under different conditions.
Thermodynamic Potentials and Equilibrium




Thermodynamic potentials help determine equilibrium conditions.
For example:
- A system at constant temperature and volume minimizes Helmholtz free energy.
- A system at constant temperature and pressure minimizes Gibbs free energy.
These principles help predict:
- Chemical equilibrium
- Phase stability
- Material behavior
Applications of Thermodynamic Potentials




Thermodynamic potentials are used in many scientific fields.
Chemistry
Predict reaction spontaneity and equilibrium.
Materials Science
Analyze phase transitions and material stability.
Statistical Mechanics
Relate microscopic states to macroscopic properties.
Engineering
Design energy systems and thermodynamic processes.
Importance in Modern Physics
Thermodynamic potentials provide a powerful framework for analyzing energy transformations and equilibrium.
They connect thermodynamics with other fields such as:
- Statistical mechanics
- Quantum physics
- Chemical thermodynamics
These potentials also help simplify complex thermodynamic calculations.
Conclusion
Thermodynamic potentials are fundamental energy functions used to analyze thermodynamic systems. The main potentials—internal energy, enthalpy, Helmholtz free energy, and Gibbs free energy—describe how energy is stored and transformed under different conditions.
Each potential is useful in specific situations depending on the variables held constant, such as temperature, pressure, or volume. By analyzing these potentials, scientists can determine equilibrium conditions, predict spontaneous processes, and study phase transitions.
Thermodynamic potentials form the foundation of many modern scientific fields, making them essential tools for understanding energy transformations in physical and chemical systems.
