Introduction
Matter exists in several physical forms known as states of matter. The most familiar states are solid, liquid, and gas. Solids and liquids are called condensed states of matter because their particles are closely packed together compared with gases.
The study of liquids and solids is an important branch of physical chemistry and materials science because these states are responsible for most of the physical structures we see in the natural world. Rocks, metals, water, plastics, crystals, biological tissues, and many other materials exist as solids or liquids.
Unlike gases, solids and liquids have strong intermolecular interactions that hold their particles close together. However, the arrangement and movement of particles differ between the two states.
- Solids have fixed shape and volume.
- Liquids have fixed volume but no fixed shape.
Understanding the properties, structure, and behavior of liquids and solids helps scientists design materials, understand biological processes, develop new technologies, and explain natural phenomena.
1. The Solid State
Definition of Solids
A solid is a state of matter characterized by a definite shape and definite volume. The particles in a solid are arranged very closely and are held together by strong intermolecular forces.
In solids, particles vibrate around fixed positions but cannot move freely from place to place.
Examples of solids include:
- Metals (iron, copper, aluminum)
- Minerals (quartz, diamond)
- Organic materials (wood, plastic)
- Ice
Characteristics of Solids
1. Definite Shape and Volume
Solids maintain their shape and volume regardless of the container in which they are placed. This happens because the particles are arranged in a stable structure.
For example, a piece of metal or rock retains its shape even when moved.
2. High Density
Solids generally have high density because their particles are packed closely together.
However, some solids such as ice have lower density than their liquid form due to special molecular structures.
3. Very Low Compressibility
Because particles are tightly packed, solids cannot be compressed easily.
Applying pressure to a solid usually results in minimal change in volume.
4. Strong Intermolecular Forces
Particles in solids are held together by strong attractive forces such as:
- Ionic bonds
- Covalent bonds
- Metallic bonds
- Van der Waals forces
These forces maintain the structural stability of solids.
5. Vibrational Motion
Particles in solids are not completely stationary. They vibrate around fixed positions.
The intensity of vibration increases with temperature.
2. Classification of Solids
Solids can be broadly classified into two main types:
- Crystalline solids
- Amorphous solids
Crystalline Solids




Crystalline solids have particles arranged in a regular, repeating pattern known as a crystal lattice.
This orderly arrangement extends throughout the entire structure.
Examples include:
- Salt crystals
- Quartz
- Diamonds
- Metals
Properties of Crystalline Solids
- Definite melting point
- Regular geometric shape
- Anisotropic physical properties
- Long-range order of particles
Types of Crystalline Solids
Crystalline solids can be classified according to the type of bonding between particles.
Ionic Solids
Ionic solids consist of positive and negative ions held together by electrostatic forces.
Examples:
- Sodium chloride
- Potassium bromide
Properties:
- High melting point
- Hard and brittle
- Conduct electricity when molten
Covalent Network Solids
In these solids, atoms are connected through covalent bonds forming a large network.
Examples:
- Diamond
- Silicon carbide
Properties:
- Very high melting point
- Extremely hard
- Poor electrical conductivity
Metallic Solids
Metallic solids consist of metal atoms arranged in a lattice surrounded by a sea of mobile electrons.
Examples:
- Iron
- Copper
- Gold
Properties:
- Good electrical conductivity
- Malleable and ductile
- Shiny appearance
Molecular Solids
These solids are composed of molecules held together by weak intermolecular forces.
Examples:
- Ice
- Dry ice
- Sugar crystals
Properties:
- Low melting points
- Soft structure
Amorphous Solids




Amorphous solids lack long-range order in their particle arrangement.
Their atoms or molecules are arranged randomly.
Examples include:
- Glass
- Rubber
- Plastics
- Wax
Properties of Amorphous Solids
- No definite melting point
- Isotropic properties
- Irregular internal structure
- Gradual softening when heated
These solids behave somewhat like very slow-moving liquids.
3. Crystal Structure and Unit Cells
A unit cell is the smallest repeating structural unit of a crystal lattice.
By repeating the unit cell in three dimensions, the entire crystal structure is formed.
Types of Unit Cells
There are several types of crystal systems including:
- Cubic
- Tetragonal
- Orthorhombic
- Hexagonal
- Monoclinic
- Triclinic
- Rhombohedral
Cubic Crystal System
Common cubic structures include:
- Simple cubic
- Body-centered cubic
- Face-centered cubic
Metals such as copper and aluminum often form cubic crystals.
4. The Liquid State
Definition of Liquids
A liquid is a state of matter with definite volume but no definite shape.
Liquids take the shape of the container in which they are placed.
Examples include:
- Water
- Oil
- Mercury
- Alcohol
Characteristics of Liquids
Definite Volume
Liquids maintain a constant volume because their particles remain close together.
No Fixed Shape
Liquids adapt to the shape of their container.
Moderate Density
Liquids are less dense than solids but much denser than gases.
Ability to Flow
Liquids can flow because their molecules move past one another.
This property is called fluidity.
Low Compressibility
Liquids are only slightly compressible because their particles are still relatively close together.
5. Intermolecular Forces in Liquids



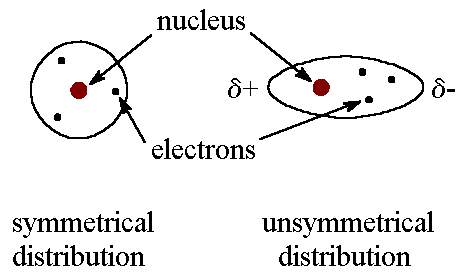
The behavior of liquids is strongly influenced by intermolecular forces.
These are attractive forces between molecules.
Types of Intermolecular Forces
London Dispersion Forces
These are weak forces present in all molecules due to temporary fluctuations in electron distribution.
They are strongest in large molecules.
Dipole–Dipole Forces
These occur between polar molecules with permanent dipole moments.
Example: interactions between hydrogen chloride molecules.
Hydrogen Bonding
Hydrogen bonding is a strong type of dipole interaction.
It occurs when hydrogen is bonded to highly electronegative atoms such as:
- Oxygen
- Nitrogen
- Fluorine
Water exhibits strong hydrogen bonding.
6. Physical Properties of Liquids
Viscosity
Viscosity is the resistance of a liquid to flow.
Examples:
- Honey has high viscosity.
- Water has low viscosity.
Viscosity decreases as temperature increases.
Surface Tension
Surface tension arises from cohesive forces between molecules at the surface of a liquid.
It causes liquids to form droplets.
Water has high surface tension due to hydrogen bonding.
Capillary Action
Capillary action is the ability of liquids to rise in narrow tubes.
This phenomenon is important in plants, where water moves through tiny vessels.
Vapor Pressure
Liquids continuously evaporate, producing vapor above their surface.
The pressure exerted by this vapor is called vapor pressure.
Higher temperature increases vapor pressure.
7. Phase Changes Between Solids and Liquids




Matter can change between solid and liquid states through phase transitions.
Melting
Melting occurs when a solid absorbs heat and becomes a liquid.
Example:
Ice melting into water.
Freezing
Freezing is the opposite of melting.
A liquid loses heat and becomes a solid.
Example:
Water turning into ice.
Crystallization
Crystallization occurs when particles organize into a structured solid during cooling.
8. Comparison Between Solids and Liquids
| Property | Solids | Liquids |
|---|---|---|
| Shape | Fixed | Takes container shape |
| Volume | Fixed | Fixed |
| Particle Motion | Vibrational | Translational and vibrational |
| Density | High | Moderate |
| Compressibility | Very low | Slight |
9. Importance of Solids and Liquids
Solids and liquids play a crucial role in everyday life and scientific applications.
Materials Science
Understanding solid structure helps design:
- Strong metals
- Semiconductors
- Nanomaterials
Biological Systems
Many biological structures depend on solids and liquids:
- Bones (solid)
- Blood (liquid)
Industrial Applications
Liquids and solids are used in:
- Manufacturing
- Chemical reactions
- Energy storage
Environmental Science
Solid and liquid states influence:
- Water cycles
- Geological formations
- Ocean systems
10. Modern Research on Solids and Liquids
Scientists continue studying condensed matter to develop advanced technologies.
Examples include:
- Superconductors
- Liquid crystals
- Nanomaterials
- Smart materials
These materials are used in electronics, medicine, and renewable energy systems.
Conclusion
Solids and liquids represent two important condensed states of matter that play a central role in both nature and technology. In solids, particles are arranged in fixed positions and held together by strong forces, giving solids their rigid shape and structural stability. Solids can exist in crystalline or amorphous forms depending on how their particles are arranged.
Liquids, on the other hand, have particles that are closely packed but able to move past one another. This gives liquids the ability to flow while still maintaining a constant volume. Properties such as viscosity, surface tension, and vapor pressure arise from intermolecular forces within liquids.
Understanding the behavior and structure of solids and liquids helps scientists explain phase changes, design new materials, and develop technologies that rely on condensed matter. From geological formations and biological systems to industrial manufacturing and advanced electronics, the study of solids and liquids continues to be one of the most important areas of physical science.