


1. Introduction to Periodic Trends
Periodic trends refer to the predictable patterns in the physical and chemical properties of elements across the periodic table. These trends arise because elements are arranged according to their atomic number and electron configuration, leading to recurring patterns in atomic structure and chemical behavior.
The periodic table is not just a list of elements but a powerful tool that allows scientists to predict how elements behave based on their position in the table. When elements are arranged in order of increasing atomic number, certain properties repeat periodically, which is why the table is called the periodic table.
Periodic trends help chemists understand important characteristics of elements, including:
- Atomic size
- Ionization energy
- Electronegativity
- Electron affinity
- Metallic character
- Reactivity
These trends arise due to changes in:
- Nuclear charge
- Electron shielding
- Distance between electrons and the nucleus
- Electron configuration
Understanding periodic trends allows scientists to predict the behavior of elements in chemical reactions, design new materials, and explain many chemical phenomena.
Periodic trends follow two primary directions:
- Across a period (left to right)
- Down a group (top to bottom)
Each direction influences atomic properties differently due to variations in nuclear attraction and electron distribution.
2. Atomic Radius



Atomic radius refers to the size of an atom, usually defined as the distance between the nucleus and the outermost electron shell.
Because the electron cloud does not have a sharp boundary, atomic radius is typically measured as half the distance between the nuclei of two identical atoms bonded together.
Trend Across a Period
As we move from left to right across a period:
- Atomic radius decreases
Reason:
- The number of protons in the nucleus increases.
- Nuclear attraction for electrons becomes stronger.
- Electrons are pulled closer to the nucleus.
Trend Down a Group
As we move down a group:
- Atomic radius increases
Reason:
- Additional electron shells are added.
- Electrons are farther from the nucleus.
- Electron shielding increases.
Thus, atoms become larger down a group.
Example
Lithium has a larger atomic radius than fluorine in the same period because fluorine has more protons pulling electrons inward.
Cesium, located near the bottom of the periodic table, has one of the largest atomic radii among elements.
3. Ionization Energy




Ionization energy is the amount of energy required to remove an electron from an atom in the gaseous state.
It reflects how strongly an atom holds onto its electrons.
First Ionization Energy
The energy required to remove the first electron from an atom.
Example reaction:
Atom → Ion + Electron
Trend Across a Period
Ionization energy increases across a period.
Reason:
- Nuclear charge increases.
- Electrons are held more tightly.
Elements on the right side of the periodic table require more energy to remove electrons.
Trend Down a Group
Ionization energy decreases down a group.
Reason:
- Electrons are farther from the nucleus.
- Electron shielding reduces nuclear attraction.
Thus, electrons are easier to remove.
Example
Helium has extremely high ionization energy because its electrons are very close to the nucleus.
Cesium has very low ionization energy because its outer electron is far from the nucleus.
4. Electronegativity




Electronegativity measures an atom’s ability to attract electrons in a chemical bond.
It is one of the most important properties for understanding chemical bonding.
The most commonly used electronegativity scale was developed by Linus Pauling.
Trend Across a Period
Electronegativity increases across a period.
Reason:
- Increasing nuclear charge
- Smaller atomic radius
- Stronger attraction for bonding electrons
Trend Down a Group
Electronegativity decreases down a group.
Reason:
- Larger atomic size
- Increased electron shielding
Example
Fluorine is the most electronegative element in the periodic table.
Cesium and francium are among the least electronegative.
Electronegativity differences between atoms determine bond types such as:
- Ionic bonds
- Polar covalent bonds
- Nonpolar covalent bonds
5. Electron Affinity




Electron affinity refers to the energy change that occurs when an atom gains an electron.
It indicates how strongly an atom attracts additional electrons.
Trend Across a Period
Electron affinity generally increases across a period.
Reason:
- Higher nuclear charge attracts incoming electrons more strongly.
Halogens have very high electron affinity.
Trend Down a Group
Electron affinity generally decreases down a group.
Reason:
- Larger atomic size reduces attraction for incoming electrons.
However, some irregularities occur due to electron repulsion and subshell stability.
6. Metallic and Nonmetallic Character



Metallic character describes an element’s tendency to lose electrons and form positive ions.
Nonmetallic character describes an element’s tendency to gain electrons.
Metallic Character Trend
Metallic character:
- Increases down a group
- Decreases across a period
Reason:
Atoms with larger size and lower ionization energy lose electrons more easily.
Nonmetallic Character Trend
Nonmetallic character:
- Increases across a period
- Decreases down a group
Elements like fluorine and oxygen are strong nonmetals.
Metals such as sodium and potassium exhibit strong metallic behavior.
7. Reactivity Trends




Reactivity describes how easily an element participates in chemical reactions.
Reactivity of Metals
Metal reactivity increases down a group.
Reason:
- Outer electrons are farther from the nucleus.
- Easier to lose electrons.
Example:
Potassium reacts more vigorously with water than lithium.
Reactivity of Nonmetals
Nonmetal reactivity increases up a group.
Reason:
- Smaller atoms attract electrons more strongly.
Fluorine is the most reactive nonmetal.
8. Shielding Effect

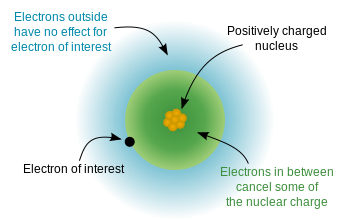


The shielding effect occurs when inner electrons block the attractive force of the nucleus on outer electrons.
As atoms gain more electron shells, inner electrons reduce the effective nuclear attraction experienced by outer electrons.
Shielding explains many periodic trends, including:
- Increasing atomic radius down groups
- Decreasing ionization energy down groups
The concept of effective nuclear charge describes the net positive charge experienced by outer electrons.
9. Importance of Periodic Trends
Periodic trends are essential tools in chemistry because they allow scientists to predict properties of elements without direct experimentation.
They help explain:
- Bond formation
- Chemical reactivity
- Physical properties
- Molecular structure
- Periodic table organization
Periodic trends also help scientists identify unknown elements based on observed properties.
10. Applications in Science and Technology
Periodic trends are widely used in many scientific and industrial fields.
Chemical Engineering
Understanding element properties helps design industrial chemical processes.
Materials Science
Periodic trends guide the development of new alloys, ceramics, and semiconductors.
Medicine
Many pharmaceuticals rely on elements whose properties follow predictable periodic trends.
Environmental Science
Periodic trends help explain the behavior of pollutants and heavy metals.
Nanotechnology
Understanding atomic properties allows scientists to manipulate materials at the nanoscale.
11. Conclusion
Periodic trends represent one of the most powerful concepts in chemistry. They reveal how the properties of elements change systematically across the periodic table due to variations in atomic structure and electron configuration.
Key periodic trends such as atomic radius, ionization energy, electronegativity, and electron affinity allow scientists to predict the behavior of elements in chemical reactions.
These trends arise from fundamental principles such as nuclear charge, electron shielding, and electron shell structure.
By studying periodic trends, chemists gain a deeper understanding of atomic interactions and chemical bonding, enabling the development of new technologies, materials, and scientific discoveries.
