



Introduction
In chemistry, a solution is a homogeneous mixture composed of two or more substances that are uniformly distributed at the molecular or ionic level. Solutions are fundamental to both natural processes and industrial applications. Many chemical reactions occur in solutions, making them essential in biological systems, environmental science, pharmaceuticals, food chemistry, and manufacturing.
The study of solutions and concentration helps scientists understand how substances interact, dissolve, and react with one another. It also provides methods to quantify how much solute is present in a given amount of solvent or solution.
A typical solution consists of:
- Solute – the substance that is dissolved
- Solvent – the substance that dissolves the solute
For example, when salt dissolves in water:
- Salt is the solute
- Water is the solvent
Solutions can exist in all three states of matter: solid, liquid, or gas. The concept of concentration is used to describe the relative amounts of solute and solvent in a solution.
Understanding solutions and concentration is crucial in fields such as medicine, environmental monitoring, food production, chemical analysis, and industrial processes.
1. Definition of Solutions
A solution is defined as a homogeneous mixture in which one or more substances (solutes) are dissolved in another substance (solvent).
Key characteristics of solutions include:
- Uniform composition throughout the mixture
- Solute particles are extremely small
- Solute particles do not settle over time
- Components cannot be separated by simple filtration
Solutions are stable mixtures because the solute particles are dispersed at the molecular or ionic level.
2. Components of a Solution
Solvent
The solvent is the component present in the largest amount. It determines the physical state of the solution.
Examples include:
- Water in saltwater
- Alcohol in alcoholic beverages
- Nitrogen in air
Water is the most common solvent and is often referred to as the universal solvent because it can dissolve many substances.
Solute
The solute is the substance that dissolves in the solvent. Solutes may be solids, liquids, or gases.
Examples include:
- Sugar in tea
- Carbon dioxide in soda
- Salt in seawater
The amount of solute relative to solvent determines the concentration of the solution.
3. Types of Solutions


Solutions can be classified based on the physical state of the solvent.
Gaseous Solutions
In gaseous solutions, both solute and solvent are gases.
Example:
- Air (mixture of nitrogen, oxygen, carbon dioxide)
Gases mix completely due to the high mobility of their molecules.
Liquid Solutions
Liquid solutions are the most common type.
Examples include:
- Salt in water
- Sugar in water
- Alcohol in water
In these solutions, a solid, liquid, or gas may dissolve in a liquid solvent.
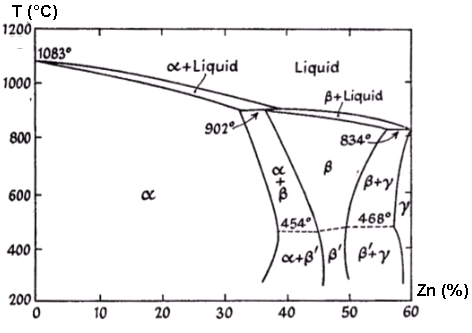
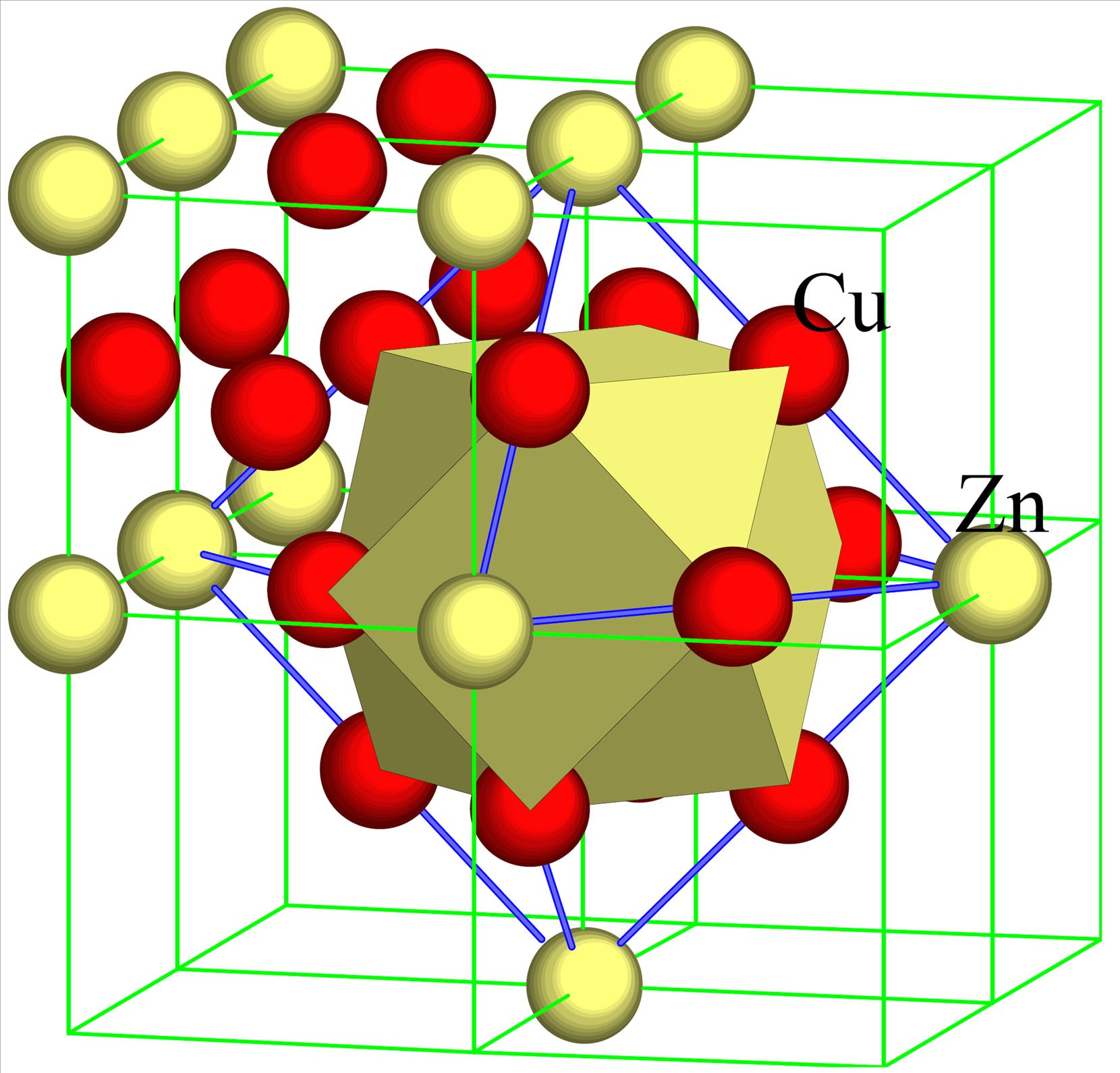
Solid Solutions
Solid solutions occur when one solid dissolves in another solid.
Examples include:
- Alloys such as brass (copper and zinc)
- Steel (iron and carbon)
These solutions play an important role in metallurgy and materials science.
4. Factors Affecting Solubility


Solubility refers to the maximum amount of solute that can dissolve in a given quantity of solvent at a specific temperature and pressure.
Several factors influence solubility.
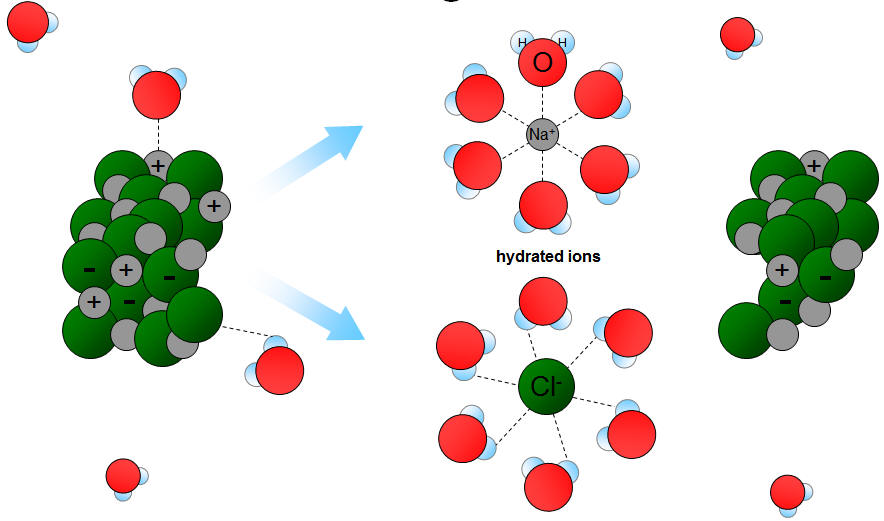
Nature of Solute and Solvent
The principle “like dissolves like” states that substances with similar polarity dissolve in each other.
Examples:
- Polar substances dissolve in polar solvents
- Nonpolar substances dissolve in nonpolar solvents
Water dissolves salt because both are polar.
Temperature
Temperature has different effects depending on the type of solute.
For solids in liquids:
- Solubility usually increases with temperature.
For gases in liquids:
- Solubility decreases as temperature increases.
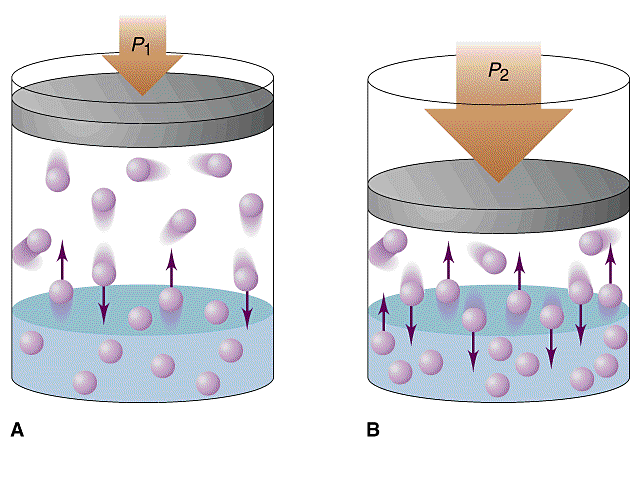
Pressure
Pressure mainly affects gases.
Increasing pressure increases the solubility of gases in liquids.
This principle explains why carbon dioxide stays dissolved in sealed soft drink bottles.
5. Concentration of Solutions
The concentration of a solution describes how much solute is present relative to the solvent or total solution.
Concentration can be expressed in several ways depending on the application.
6. Methods of Expressing Concentration
Mass Percentage
Mass percentage expresses the mass of solute relative to the mass of the entire solution.
Formula:
Mass % = (mass of solute / mass of solution) × 100
This method is commonly used in food and industrial chemistry.
Example:
A solution containing 10 g salt in 100 g solution has a mass percentage of 10%.
Volume Percentage
Volume percentage is used for liquid solutions.
Formula:
Volume % = (volume of solute / volume of solution) × 100
Example:
Alcohol concentration in beverages is expressed as volume percentage.
Parts per Million (ppm)
Parts per million is used for extremely dilute solutions.
Example:
Pollutant concentration in water.
1 ppm means one part of solute per one million parts of solution.
Molarity
M = \frac{n}{V}
Molarity is one of the most common concentration units in chemistry.
It represents the number of moles of solute per liter of solution.
Where:
M = molarity
n = number of moles of solute
V = volume of solution in liters
Molality
Molality expresses concentration as moles of solute per kilogram of solvent.
Formula:
Molality = moles of solute / mass of solvent (kg)
Molality is independent of temperature.
Mole Fraction
Mole fraction represents the ratio of moles of a component to the total moles in the solution.
It is often used in thermodynamics.
7. Dilution of Solutions
Dilution occurs when additional solvent is added to reduce the concentration of a solution.
The relationship between initial and final concentrations is:
M_1V_1 = M_2V_2
Where:
M₁ = initial concentration
V₁ = initial volume
M₂ = final concentration
V₂ = final volume
This equation is widely used in laboratory preparations.
8. Saturated and Unsaturated Solutions
Unsaturated Solutions
An unsaturated solution contains less solute than the maximum amount that can dissolve.
More solute can still dissolve in the solvent.
Saturated Solutions
A saturated solution contains the maximum amount of dissolved solute at a given temperature.
Any additional solute will remain undissolved.
Supersaturated Solutions
Supersaturated solutions contain more solute than the normal saturation limit.
They are unstable and can crystallize easily.
9. Colligative Properties of Solutions



Colligative properties depend on the number of solute particles rather than their chemical identity.
Vapor Pressure Lowering
When a nonvolatile solute is added to a solvent, the vapor pressure of the solution decreases.
This occurs because solute particles reduce the number of solvent molecules escaping into vapor.
Boiling Point Elevation
Adding solute raises the boiling point of the solvent.
Example:
Saltwater boils at a higher temperature than pure water.
Freezing Point Depression
Dissolving solute lowers the freezing point of a solvent.
Example:
Salt is used to melt ice on roads.
Osmotic Pressure
Osmosis is the movement of solvent through a semipermeable membrane from low solute concentration to high solute concentration.
Osmotic pressure is important in biological systems such as cells and blood circulation.
10. Applications of Solutions
Solutions are essential in many scientific and technological fields.
Biological Systems
Many biological processes occur in aqueous solutions.
Examples include:
- Blood plasma
- Cellular fluids
- Nutrient transport
Medicine and Pharmaceuticals
Drug formulations often require precise solution concentrations.
Examples include:
- Saline solutions
- Intravenous fluids
- Syrups
Environmental Science
Solutions are important in studying:
- Water pollution
- Ocean chemistry
- Acid rain
Industrial Chemistry
Solutions are widely used in:
- Chemical manufacturing
- Electroplating
- Food processing
11. Importance of Studying Solutions and Concentration
The study of solutions and concentration is fundamental in chemistry because most chemical reactions occur in solution form. By understanding how substances dissolve and how concentration affects reactions, scientists can control chemical processes with high precision.
Concentration measurements allow chemists to prepare accurate mixtures for laboratory experiments, industrial production, medical treatments, and environmental monitoring. Without accurate concentration control, reactions may proceed incorrectly or produce unsafe conditions.
Solutions also provide insight into intermolecular interactions, thermodynamics, and molecular behavior. This knowledge helps scientists design new materials, improve pharmaceuticals, and understand biological systems.
Conclusion
Solutions are homogeneous mixtures formed when one or more solutes dissolve in a solvent. They are fundamental to chemistry, biology, medicine, and industry because many natural and technological processes occur in solution form.
The behavior of solutions depends on the interactions between solute and solvent molecules, as well as factors such as temperature, pressure, and polarity. Solubility determines how much solute can dissolve, while concentration describes how much solute is present in a given amount of solution.
Various methods are used to express concentration, including mass percentage, molarity, molality, mole fraction, and parts per million. These measurements allow scientists to control chemical reactions and prepare accurate solutions for scientific and industrial applications.
Solutions also exhibit unique properties known as colligative properties, which depend on the number of dissolved particles. These properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
Understanding solutions and concentration is essential for modern science and technology. From biological systems and environmental processes to industrial manufacturing and medical treatments, the principles governing solutions continue to play a vital role in our understanding of the chemical world.
