



1. Introduction to Intermolecular Forces
Intermolecular forces are the attractive forces that occur between molecules. These forces are weaker than chemical bonds such as ionic or covalent bonds, but they play a crucial role in determining the physical properties of substances. Properties such as boiling point, melting point, viscosity, solubility, and surface tension are largely influenced by intermolecular interactions.
While chemical bonds hold atoms together within molecules, intermolecular forces act between separate molecules. These forces determine how molecules interact with one another in liquids, solids, and gases.
Intermolecular forces are essential in many chemical and biological processes. They influence the structure of biological macromolecules, determine how substances dissolve in solvents, and control the behavior of materials at the molecular level.
Examples of phenomena influenced by intermolecular forces include:
- Water forming droplets due to surface tension
- Ice floating on water because of hydrogen bonding
- The condensation of gases into liquids
- The folding of proteins in biological systems
Understanding intermolecular forces helps chemists predict and explain the behavior of substances in different states of matter.
2. Types of Intermolecular Forces



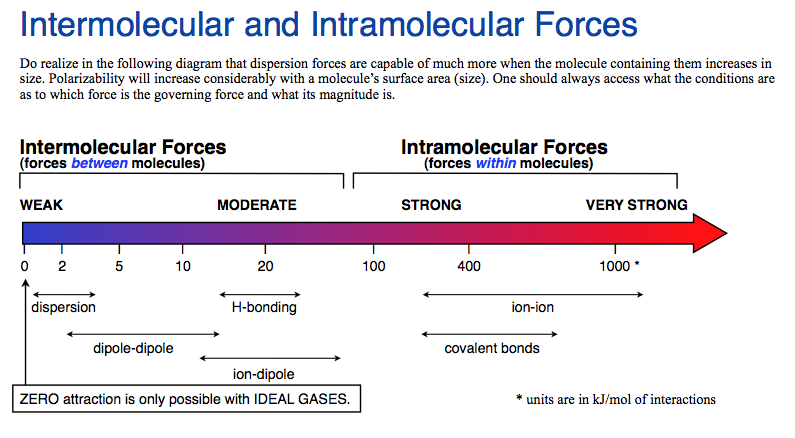
There are several types of intermolecular forces. The most common include:
- London dispersion forces
- Dipole–dipole interactions
- Hydrogen bonding
- Ion–dipole interactions
These forces vary in strength and arise from different types of molecular interactions.
3. London Dispersion Forces


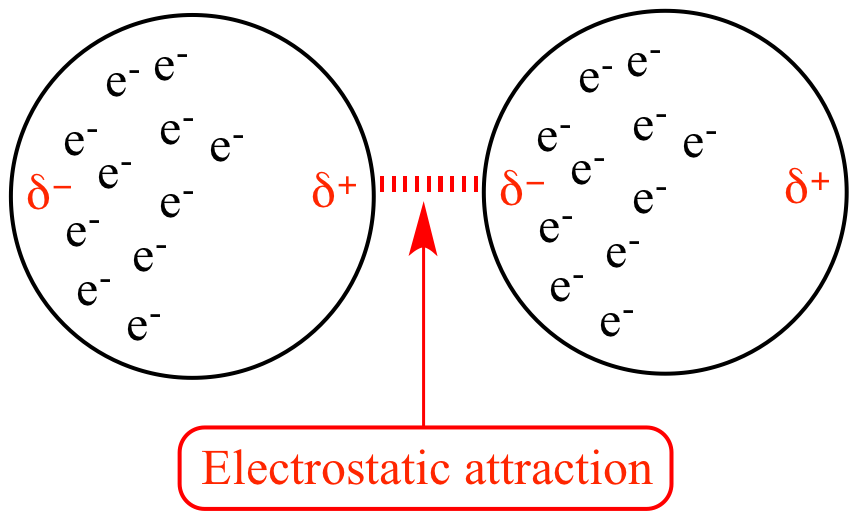
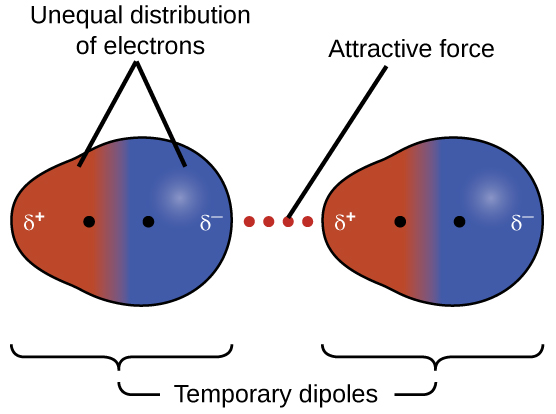
London dispersion forces are the weakest type of intermolecular force. They arise from temporary fluctuations in the electron distribution of atoms or molecules.
At any moment, electrons may be unevenly distributed around an atom or molecule. This uneven distribution creates a temporary dipole, where one part of the molecule becomes slightly negative and another part becomes slightly positive.
This temporary dipole can induce a dipole in a neighboring molecule, resulting in an attractive force between them.
London dispersion forces occur in all atoms and molecules, regardless of whether they are polar or nonpolar.
These forces become stronger under certain conditions:
- Larger molecules with more electrons
- Greater molecular surface area
- Increased polarizability of electron clouds
Examples of substances dominated by dispersion forces include noble gases and nonpolar molecules such as methane.
4. Dipole–Dipole Interactions



Dipole–dipole interactions occur between polar molecules. In these molecules, electrons are shared unequally, resulting in partial positive and negative charges.
The positive end of one molecule is attracted to the negative end of another molecule. This attraction creates dipole–dipole forces.
These interactions are stronger than London dispersion forces but weaker than hydrogen bonding.
Examples of polar molecules that exhibit dipole–dipole interactions include:
- Hydrogen chloride (HCl)
- Sulfur dioxide (SO₂)
- Acetone
The strength of dipole–dipole interactions depends on the magnitude of the molecular dipole moment.
5. Hydrogen Bonding




Hydrogen bonding is a special type of dipole–dipole interaction that occurs when hydrogen is bonded to highly electronegative atoms such as:
- Oxygen
- Nitrogen
- Fluorine
When hydrogen is bonded to these atoms, it becomes strongly positive because the electronegative atom pulls electrons toward itself.
The positively charged hydrogen atom can then attract the lone pair electrons of nearby electronegative atoms.
Hydrogen bonds are significantly stronger than other intermolecular forces.
Importance of Hydrogen Bonding
Hydrogen bonding is responsible for many important properties of substances.
Properties of Water
Water has unusually high boiling and melting points because of strong hydrogen bonding between molecules.
Hydrogen bonding also causes water to have:
- High surface tension
- High heat capacity
- Expansion upon freezing
Biological Systems
Hydrogen bonds stabilize biological molecules such as:
- DNA double helix
- Protein structures
- Enzyme-substrate interactions
These interactions are essential for life processes.
6. Ion–Dipole Interactions

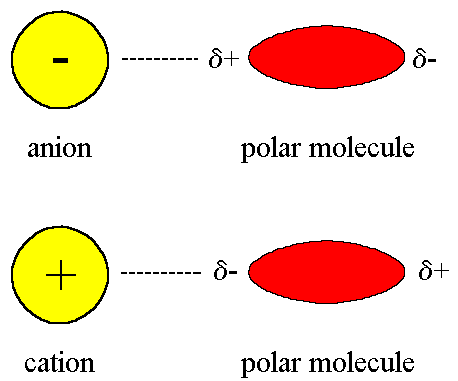

Ion–dipole interactions occur between ions and polar molecules.
This type of force is particularly important when ionic compounds dissolve in polar solvents such as water.
For example, when sodium chloride dissolves in water:
- Sodium ions attract the negative oxygen end of water molecules.
- Chloride ions attract the positive hydrogen end of water molecules.
These interactions stabilize ions in solution and allow the compound to dissolve.
Ion–dipole forces are stronger than most intermolecular forces.
7. Relative Strength of Intermolecular Forces
The strength of intermolecular forces generally follows this order:
Ion–dipole interactions (strongest)
Hydrogen bonding
Dipole–dipole interactions
London dispersion forces (weakest)
However, large molecules with many electrons can have strong dispersion forces that rival other intermolecular interactions.
The strength of intermolecular forces affects many physical properties of substances.
8. Intermolecular Forces and Physical Properties




Intermolecular forces strongly influence physical properties.
Boiling Point
Stronger intermolecular forces require more energy to separate molecules.
Therefore, substances with stronger forces have higher boiling points.
Melting Point
Substances with strong intermolecular attractions require more energy to transition from solid to liquid.
Viscosity
Viscosity is a measure of a liquid’s resistance to flow.
Liquids with strong intermolecular forces tend to have higher viscosity.
Surface Tension
Surface tension arises from intermolecular forces acting on molecules at the surface of a liquid.
Water has high surface tension due to hydrogen bonding.
9. Intermolecular Forces in Biology
Intermolecular forces are essential for biological systems.
They help maintain the structure of many biological molecules.
Examples include:
- Hydrogen bonding in DNA base pairing
- Protein folding through hydrogen bonding and dipole interactions
- Lipid membrane formation through dispersion forces
Without these forces, biological molecules would not maintain their functional shapes.
10. Applications in Chemistry and Technology
Understanding intermolecular forces is important in many scientific fields.
Drug Design
Drug molecules must interact with biological targets through intermolecular forces.
Materials Science
Intermolecular interactions determine properties of polymers and plastics.
Environmental Chemistry
Pollutant behavior depends on intermolecular interactions with water and air.
Nanotechnology
Weak intermolecular forces help assemble nanoscale structures.
11. Comparison with Chemical Bonds
It is important to distinguish intermolecular forces from chemical bonds.
Chemical bonds:
- Hold atoms together within molecules
- Strong interactions
- Include ionic, covalent, and metallic bonds
Intermolecular forces:
- Act between molecules
- Much weaker than chemical bonds
- Influence physical properties rather than chemical composition
12. Conclusion
Intermolecular forces are essential interactions that occur between molecules. Although weaker than chemical bonds, they play a crucial role in determining the physical and chemical behavior of substances.
Different types of intermolecular forces—including London dispersion forces, dipole–dipole interactions, hydrogen bonding, and ion–dipole interactions—arise from various molecular interactions and charge distributions.
These forces influence many properties such as boiling point, melting point, viscosity, and solubility. They are also critical for biological systems, materials science, and many technological applications.
By understanding intermolecular forces, chemists can explain how molecules interact, how substances change state, and how complex molecular systems function.
















