



1. Introduction to Surface Chemistry
Surface chemistry is the branch of chemistry that studies phenomena occurring at the interface between two phases. A surface or interface is the boundary where two different phases meet. These phases may include combinations such as solid–gas, solid–liquid, liquid–gas, or solid–solid.
Surface chemistry is particularly important because many chemical processes occur at interfaces rather than within the bulk of materials. The molecules present at surfaces often behave differently from those in the interior because they experience different forces and interactions.
The study of surface chemistry helps scientists understand processes such as:
- Adsorption of gases on solid surfaces
- Catalysis in industrial reactions
- Formation of colloids
- Action of detergents and surfactants
- Corrosion and electrochemical reactions
Surface chemistry plays an essential role in many fields including materials science, environmental science, nanotechnology, and industrial chemistry.
Many modern technologies depend on surface phenomena, including fuel cells, catalysts, sensors, and nanomaterials.
2. Concept of Surface and Interface
A surface refers to the boundary between a solid and another phase, such as a gas or liquid.
An interface refers to the boundary between any two phases.
Examples include:
- Solid–gas interface (metal surface exposed to air)
- Solid–liquid interface (solid immersed in liquid)
- Liquid–gas interface (surface of water exposed to air)
- Liquid–liquid interface (oil and water boundary)
Molecules at the interface experience different forces compared to molecules inside the bulk phase. This difference results in various surface phenomena.
3. Surface Energy and Surface Tension
Surface molecules have higher energy because they are not surrounded by molecules on all sides.
This excess energy leads to surface tension, which is a measure of the force required to increase the surface area of a liquid.
Surface tension plays a significant role in processes such as:
- Formation of droplets
- Capillary action
- Wetting of surfaces
Liquids with strong intermolecular forces generally have higher surface tension.
4. Adsorption



Adsorption is one of the most important phenomena studied in surface chemistry.
Definition
Adsorption is the process in which molecules from a gas or liquid accumulate on the surface of a solid or liquid.
The substance that accumulates on the surface is called the adsorbate, while the material providing the surface is called the adsorbent.
Examples include:
- Adsorption of gases on charcoal
- Adsorption of dyes on activated carbon
Adsorption vs Absorption
Adsorption involves accumulation on the surface, whereas absorption involves penetration into the bulk of a material.
Example:
- Water absorbed by sponge (absorption)
- Gas molecules attached to solid surface (adsorption)
5. Types of Adsorption
Adsorption is generally classified into two main types.
Physical Adsorption (Physisorption)
Physical adsorption occurs due to weak intermolecular forces such as van der Waals forces.
Characteristics include:
- Low heat of adsorption
- Reversible process
- Occurs at low temperatures
- No chemical bond formation
Example:
Adsorption of gases on charcoal.
Chemical Adsorption (Chemisorption)
Chemical adsorption involves formation of chemical bonds between adsorbate and adsorbent.
Characteristics include:
- High heat of adsorption
- Irreversible process
- Requires activation energy
- Strong chemical interaction
Example:
Adsorption of hydrogen on metal catalysts.
6. Factors Affecting Adsorption
Several factors influence adsorption.
Nature of Adsorbent
Porous materials with large surface areas show greater adsorption.
Examples include:
- Activated carbon
- Silica gel
Surface Area
Greater surface area increases adsorption capacity.
Temperature
Physical adsorption decreases with increasing temperature.
Chemisorption may increase with temperature initially.
Pressure
Increasing pressure generally increases adsorption of gases.
7. Adsorption Isotherms



Adsorption isotherms describe the relationship between the amount of substance adsorbed and the pressure of gas at constant temperature.
Freundlich Adsorption Isotherm
Empirical equation describing adsorption:
[
x/m = kP^{1/n}
]
Where:
- x = mass of adsorbate
- m = mass of adsorbent
- P = pressure
Langmuir Adsorption Isotherm
Langmuir theory assumes monolayer adsorption on uniform surfaces.
Key assumptions include:
- Fixed number of adsorption sites
- Each site holds one molecule
- No interaction between adsorbed molecules
8. Catalysis and Surface Chemistry


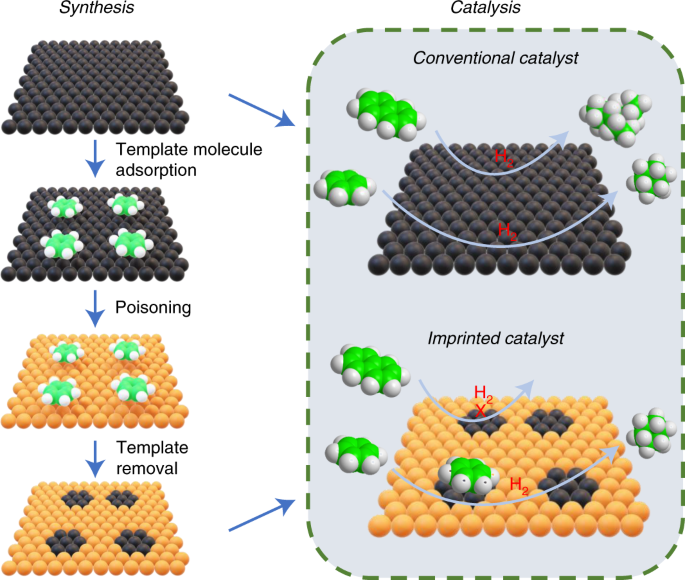

Catalysis often involves surface reactions.
In heterogeneous catalysis:
- Reactants adsorb on catalyst surface
- Reaction occurs on surface
- Products desorb from surface
Examples include:
- Haber process catalyst
- Catalytic converters in automobiles
Surface area of catalysts greatly affects reaction rates.
9. Colloids




Colloids are heterogeneous mixtures where particles of one substance are dispersed in another.
Particle size typically ranges from 1 nm to 1000 nm.
Examples include:
- Milk
- Fog
- Smoke
- Paint
Types of Colloids
Colloids can be classified based on the phases involved.
Examples include:
- Sol (solid in liquid)
- Gel (liquid in solid)
- Emulsion (liquid in liquid)
10. Properties of Colloids
Colloidal systems exhibit several unique properties.
Tyndall Effect
Scattering of light by colloidal particles.
Brownian Motion
Random motion of particles due to collisions with molecules.
Charge on Colloidal Particles
Particles often carry electric charge that stabilizes the colloid.
11. Emulsions


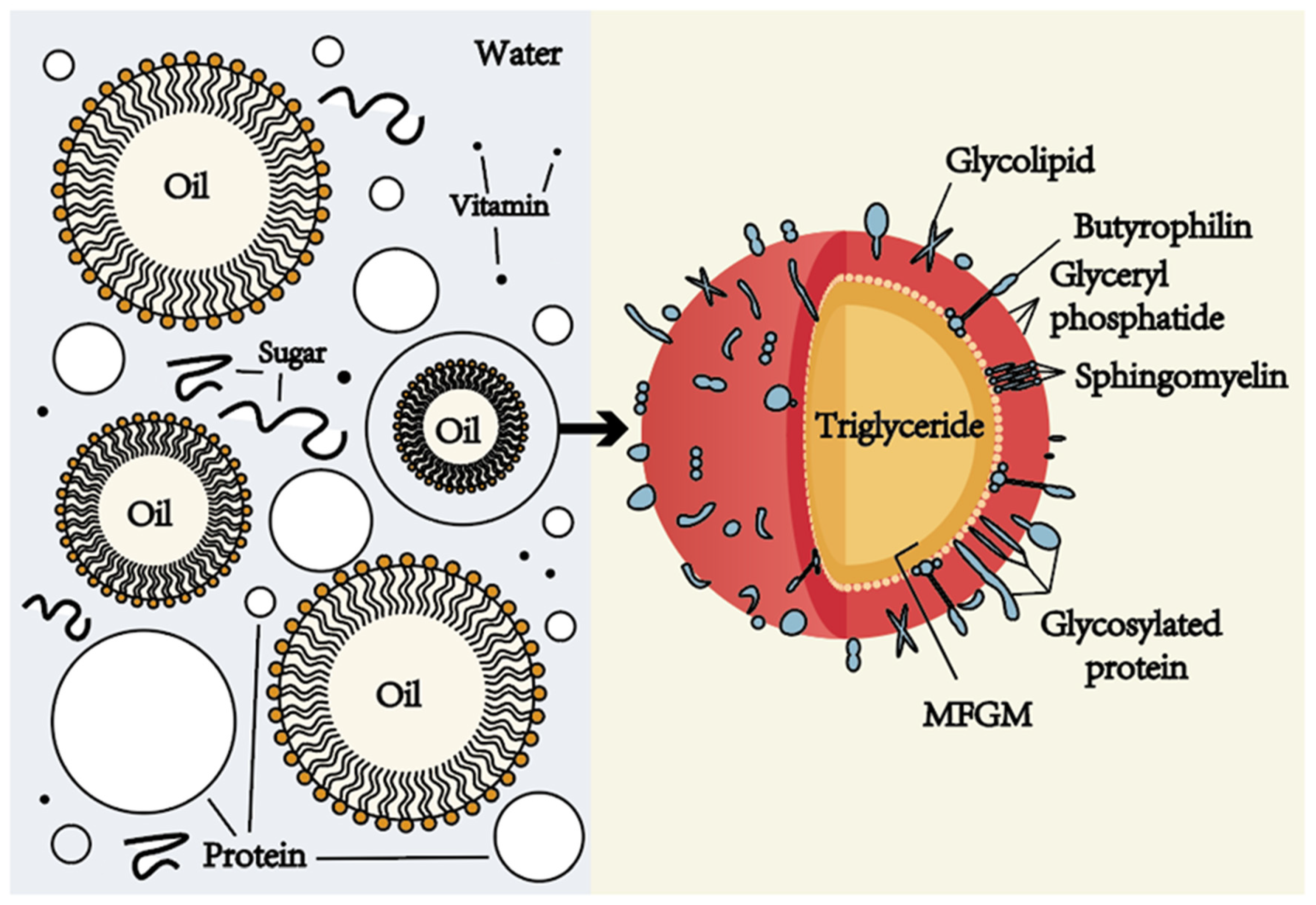

Emulsions are colloidal systems where one liquid is dispersed in another immiscible liquid.
Examples include:
- Milk
- Mayonnaise
- Cosmetic creams
Emulsions are stabilized by substances called emulsifiers.
12. Surfactants and Detergents
Surfactants are compounds that reduce surface tension.
They contain two parts:
- Hydrophilic (water-loving)
- Hydrophobic (water-repelling)
Surfactants form micelles, which trap dirt and grease.
This property makes them useful in:
- Cleaning agents
- Soaps
- Detergents
13. Applications of Surface Chemistry
Surface chemistry has many practical applications.
Industrial Catalysis
Many industrial reactions occur on catalyst surfaces.
Environmental Protection
Adsorption is used in water purification and air pollution control.
Medicine
Drug delivery systems use nanoparticles and colloids.
Food Industry
Colloidal systems are common in foods such as milk and butter.
14. Nanotechnology and Surface Chemistry
Surface chemistry plays a crucial role in nanotechnology.
Nanomaterials have very large surface areas, making surface interactions extremely important.
Applications include:
- Nanocatalysts
- Drug delivery
- Sensors
- Electronic devices
15. Importance of Surface Chemistry
Surface chemistry is essential for understanding many natural and industrial processes.
It helps explain:
- Catalytic reactions
- Adsorption processes
- Stability of colloids
- Behavior of detergents and emulsions
The field continues to play a major role in modern scientific research and technological development.
Conclusion
Surface chemistry studies chemical processes occurring at surfaces and interfaces between different phases. Important phenomena such as adsorption, catalysis, colloid formation, and surface tension arise from interactions at interfaces. Adsorption can occur through physical or chemical interactions, and various models such as Freundlich and Langmuir isotherms describe adsorption behavior. Surface chemistry is fundamental to many applications including industrial catalysis, environmental protection, food science, medicine, and nanotechnology. Understanding surface interactions helps scientists design better catalysts, develop advanced materials, and improve industrial processes.
