



Introduction
Ionized gases, commonly known as plasma, represent a unique state of matter in which atoms are partially or completely ionized. In this state, electrons are separated from atoms, producing a mixture of free electrons, ions, and neutral particles. Because of the presence of charged particles, ionized gases exhibit properties that differ significantly from ordinary gases.
Plasma is often referred to as the fourth state of matter, in addition to solids, liquids, and gases. When sufficient energy is supplied to a gas—through heating, electrical discharge, or radiation—the atoms gain enough energy for electrons to escape from their atomic orbitals. This process results in ionization and leads to the formation of plasma.
Ionized gases are extremely common in the universe. In fact, most of the visible matter in the universe exists in the plasma state. Stars, including the Sun, consist primarily of plasma. Plasma also occurs naturally in lightning, auroras, and the ionosphere of Earth’s atmosphere.
The study of ionized gases forms the basis of plasma physics, a field that combines elements of electromagnetism, thermodynamics, and quantum mechanics. Important theoretical contributions were made by scientists such as Irving Langmuir, who studied ionized gases in electrical discharges and helped establish the foundations of plasma physics.
Ionized gases have numerous applications in science and technology, including fusion energy research, plasma televisions, semiconductor manufacturing, and space propulsion systems.
Nature of Ionized Gases
In ordinary gases, atoms are neutral and consist of electrons bound to a positively charged nucleus.
However, when sufficient energy is supplied to a gas:
- Electrons gain kinetic energy.
- Some electrons escape from atoms.
- Positive ions are formed.
This process is known as ionization.
The resulting mixture contains:
- Free electrons
- Positive ions
- Neutral atoms
- Electromagnetic radiation
Because of the presence of charged particles, ionized gases interact strongly with electric and magnetic fields.
Ionization Process
Ionization occurs when atoms lose electrons.
The ionization energy is the minimum energy required to remove an electron from an atom.
Ionization can occur through several mechanisms.
Thermal Ionization
Occurs when gases are heated to very high temperatures.
Electrical Ionization
Occurs when strong electric fields accelerate electrons.
Photoionization
Occurs when atoms absorb high-energy photons.
Impact Ionization
Occurs when energetic particles collide with atoms.
These processes create plasma in various natural and laboratory environments.
Plasma as the Fourth State of Matter




Plasma is often described as the fourth state of matter.
The progression of states occurs as energy increases.
Solid → Liquid → Gas → Plasma
In plasma:
- Atoms are ionized.
- Charged particles move freely.
- Strong electromagnetic interactions occur.
Unlike neutral gases, plasmas conduct electricity and respond strongly to magnetic fields.
Properties of Ionized Gases
Ionized gases exhibit several unique properties.
Electrical Conductivity
Plasma is an excellent conductor of electricity.
This is due to the presence of free electrons and ions that move easily under electric fields.
Collective Behavior
Charged particles in plasma interact through electromagnetic forces.
These interactions cause collective motion of particles.
Plasma Oscillations
Electrons in plasma can oscillate collectively around ions.
These oscillations produce waves known as plasma oscillations.
Magnetic Field Interaction
Plasma responds strongly to magnetic fields.
Magnetic fields can confine or guide plasma motion.
This property is essential in fusion reactors.
Debye Shielding
In plasma, electric fields are screened over short distances.
This phenomenon is called Debye shielding.
It occurs because electrons rearrange themselves to neutralize electric fields.
Plasma Frequency
The natural oscillation frequency of electrons in plasma is called the plasma frequency.
It is given by:
[
\omega_p = \sqrt{\frac{n e^2}{\varepsilon_0 m_e}}
]
where
- (n) = electron density
- (e) = electron charge
- (m_e) = electron mass
Plasma frequency determines how plasma interacts with electromagnetic waves.
Types of Plasma
Ionized gases can be classified into different types.
Thermal Plasma



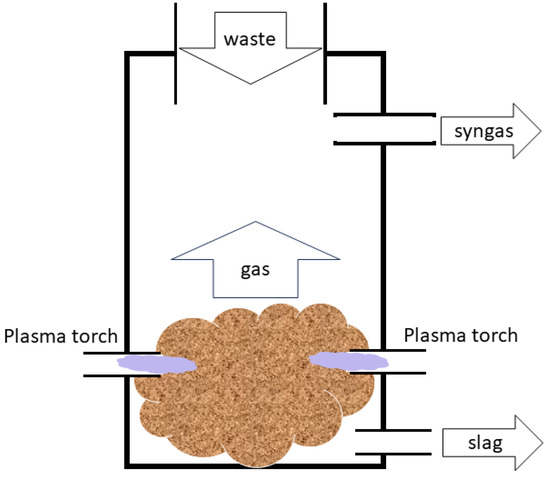
In thermal plasma, all particles have approximately the same temperature.
These plasmas occur at extremely high temperatures.
Examples include:
- Plasma torches
- Lightning
- Stellar interiors
Non-Thermal Plasma




In non-thermal plasma:
- Electrons have very high temperatures
- Ions and neutral atoms remain relatively cool.
These plasmas are used in many technological applications such as surface treatment and sterilization.
Natural Occurrence of Ionized Gases
Plasma occurs naturally in many environments.
Stars
Stars are composed mainly of plasma.
High temperatures in stellar interiors cause continuous ionization of atoms.
Lightning
Lightning produces ionized channels in the atmosphere.
These channels conduct large electric currents.
Aurora
Auroras occur when charged particles from the Sun interact with Earth’s atmosphere.
These interactions create glowing ionized gases.
Ionosphere
The ionosphere is a region of Earth’s upper atmosphere containing ionized particles.
This region plays an important role in radio communication.
Plasma in Space
Most of the universe exists in the plasma state.
Examples include:
- Solar wind
- Interstellar plasma
- Planetary magnetospheres
These plasmas interact with magnetic fields and electromagnetic radiation.
Plasma Confinement



Controlling plasma is challenging because of its high temperature and dynamic behavior.
Scientists use magnetic confinement to control plasma in fusion experiments.
Devices such as tokamaks use strong magnetic fields to confine plasma inside toroidal chambers.
Magnetic confinement prevents plasma from touching reactor walls.
Applications of Ionized Gases
Ionized gases have many practical applications.
Fusion Energy
Plasma is the key medium for nuclear fusion reactions.
Fusion reactors attempt to produce energy by fusing hydrogen isotopes.
Plasma Displays
Plasma televisions use ionized gases to produce light in display panels.
Semiconductor Manufacturing
Plasma processes are used in microelectronics fabrication.
These processes include plasma etching and deposition.
Plasma Propulsion
Ionized gases are used in spacecraft propulsion systems.
Ion thrusters accelerate plasma to generate thrust.
Medical Applications
Cold plasma is used for sterilization and wound treatment.
It can kill bacteria without damaging human tissue.
Industrial Applications
Plasma technologies are used for:
- Welding
- Cutting metals
- Surface coating
- Waste treatment
Plasma Physics Research
Plasma physics is an active research field.
Scientists study:
- Plasma waves
- Plasma instabilities
- Magnetohydrodynamics
- Plasma turbulence
Understanding these phenomena is essential for developing fusion energy and space technologies.
Challenges in Plasma Control
Plasma is difficult to control due to its high temperature and complex behavior.
Major challenges include:
- Plasma instabilities
- Energy losses
- Material limitations
Overcoming these challenges is necessary for practical fusion reactors.
Future of Plasma Technology
Plasma science is expected to play a major role in future technologies.
Possible developments include:
- Fusion power plants
- Advanced propulsion systems
- Plasma medicine
- Environmental plasma technologies
Research in plasma physics continues to expand our understanding of ionized gases and their applications.
Conclusion
Ionized gases, or plasma, represent a unique state of matter consisting of free electrons, ions, and neutral particles. Unlike ordinary gases, plasmas exhibit strong electrical conductivity, collective behavior, and interactions with electromagnetic fields.
Plasma occurs naturally in stars, lightning, auroras, and the ionosphere, making it the most abundant state of matter in the universe. Advances in plasma physics have led to numerous technological applications, including fusion energy research, semiconductor manufacturing, plasma displays, and space propulsion systems.
As scientific research continues, ionized gases are expected to play an increasingly important role in energy production, advanced materials, medicine, and space exploration.
