



1. Introduction to Computational Chemistry
Computational chemistry is a branch of chemistry that uses computer simulations, mathematical models, and theoretical chemistry methods to study the structure, properties, and reactions of molecules and materials. Instead of relying solely on laboratory experiments, scientists use computational techniques to predict chemical behavior and analyze molecular systems at the atomic level.
Computational chemistry combines principles from:
- Quantum mechanics
- Classical mechanics
- Mathematics
- Computer science
- Physical chemistry
The development of powerful computers and advanced algorithms has made computational chemistry an essential tool in modern scientific research.
Computational methods allow chemists to:
- Predict molecular structures
- Study reaction mechanisms
- Simulate chemical reactions
- Design new materials and drugs
- Analyze molecular properties
These techniques are widely used in industries such as pharmaceuticals, materials science, nanotechnology, and environmental chemistry.
2. Importance of Computational Chemistry
Computational chemistry plays an important role in modern scientific research.
Some advantages include:
Predicting Molecular Behavior
Computational models can predict properties of molecules before they are synthesized in the laboratory.
Reducing Experimental Costs
Simulations can reduce the need for expensive laboratory experiments.
Understanding Complex Systems
Many chemical processes occur at extremely small scales that are difficult to observe experimentally.
Computational chemistry allows scientists to visualize molecular interactions and understand chemical mechanisms in detail.
3. Foundations of Computational Chemistry
Computational chemistry is based on two fundamental theoretical approaches.
Quantum Mechanics
Quantum mechanics describes the behavior of electrons and atoms at the atomic scale.
It is used to calculate:
- Electronic structures
- Molecular energies
- Chemical bonding
The fundamental equation in quantum chemistry is the Schrödinger equation, which describes how the quantum state of a system changes with time.
Solving this equation provides information about molecular properties.
Classical Mechanics
Classical mechanics is used to describe motion of atoms and molecules using Newton’s laws of motion.
It is particularly useful for studying large molecular systems such as proteins or polymers.
4. Quantum Chemistry Methods

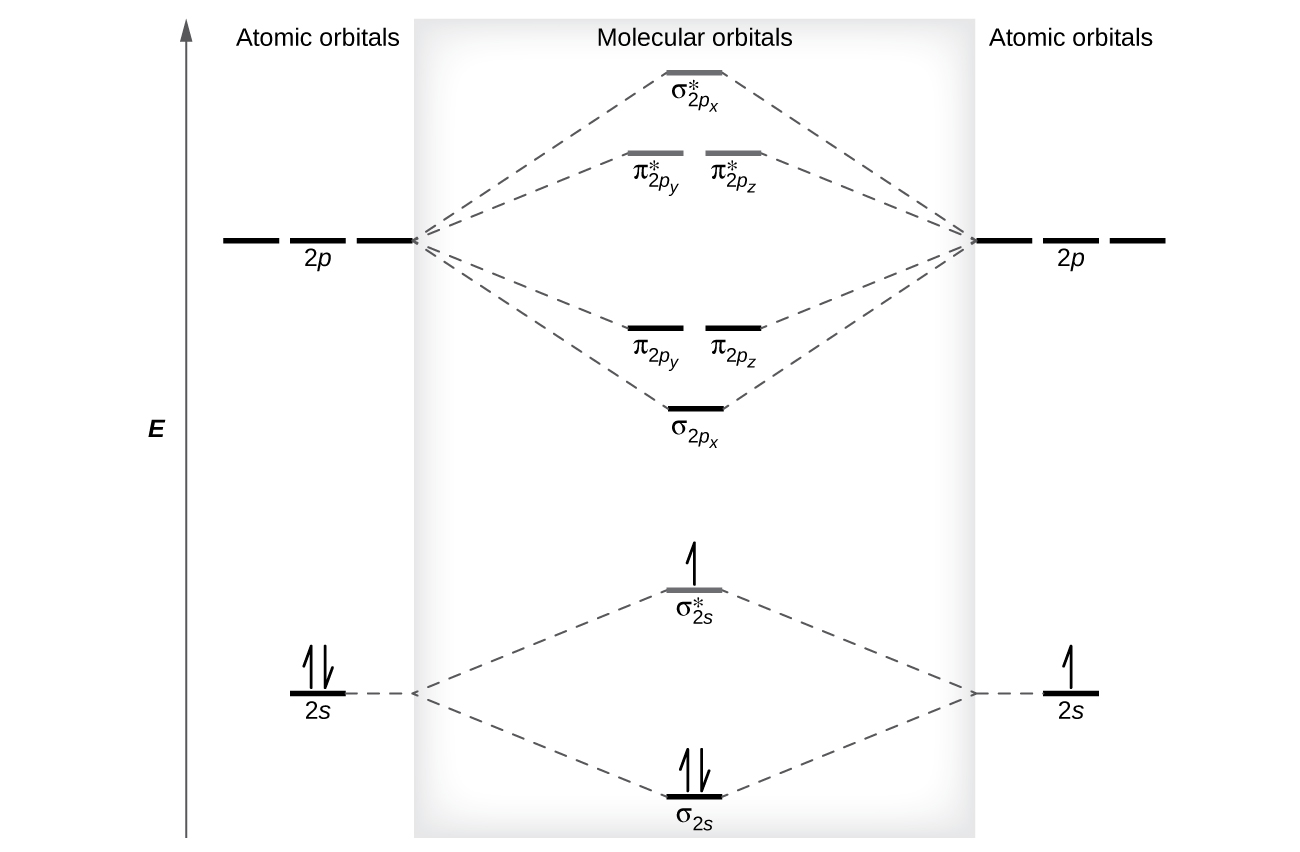

Quantum chemistry methods calculate electronic structures of molecules.
These methods include:
Ab Initio Methods
Ab initio methods use fundamental physical principles without empirical parameters.
Examples include:
- Hartree–Fock method
- Post-Hartree–Fock methods
These calculations are accurate but computationally expensive.
Density Functional Theory (DFT)
DFT is one of the most widely used computational chemistry methods.
Instead of calculating individual electron interactions, it focuses on electron density.
DFT provides a good balance between accuracy and computational efficiency.
Semi-Empirical Methods
These methods simplify quantum calculations using experimental parameters.
They are faster but less accurate than ab initio methods.
5. Molecular Mechanics


Molecular mechanics treats molecules as systems of atoms connected by springs.
Atoms are represented as spheres, and bonds behave like springs that stretch or bend.
This method uses force fields to calculate molecular energies.
Common force fields include:
- AMBER
- CHARMM
- OPLS
Molecular mechanics is particularly useful for studying large biological molecules.
6. Molecular Dynamics Simulations
Molecular dynamics (MD) simulations study how molecules move over time.
These simulations use Newton’s laws of motion to predict atomic movements.
Applications include:
- Protein folding
- Enzyme dynamics
- Membrane interactions
- Drug binding studies
Molecular dynamics simulations allow scientists to observe molecular behavior over time.
7. Monte Carlo Simulations
Monte Carlo simulations use random sampling techniques to model molecular systems.
They are useful for studying:
- Thermodynamic properties
- Phase transitions
- Complex molecular systems
Monte Carlo methods are widely used in statistical mechanics and materials science.
8. Molecular Modeling




Molecular modeling involves constructing and manipulating molecular structures using computer software.
It allows scientists to visualize molecules in three dimensions and analyze their interactions.
Common molecular modeling techniques include:
Molecular Docking
Predicts how molecules bind to biological targets such as proteins.
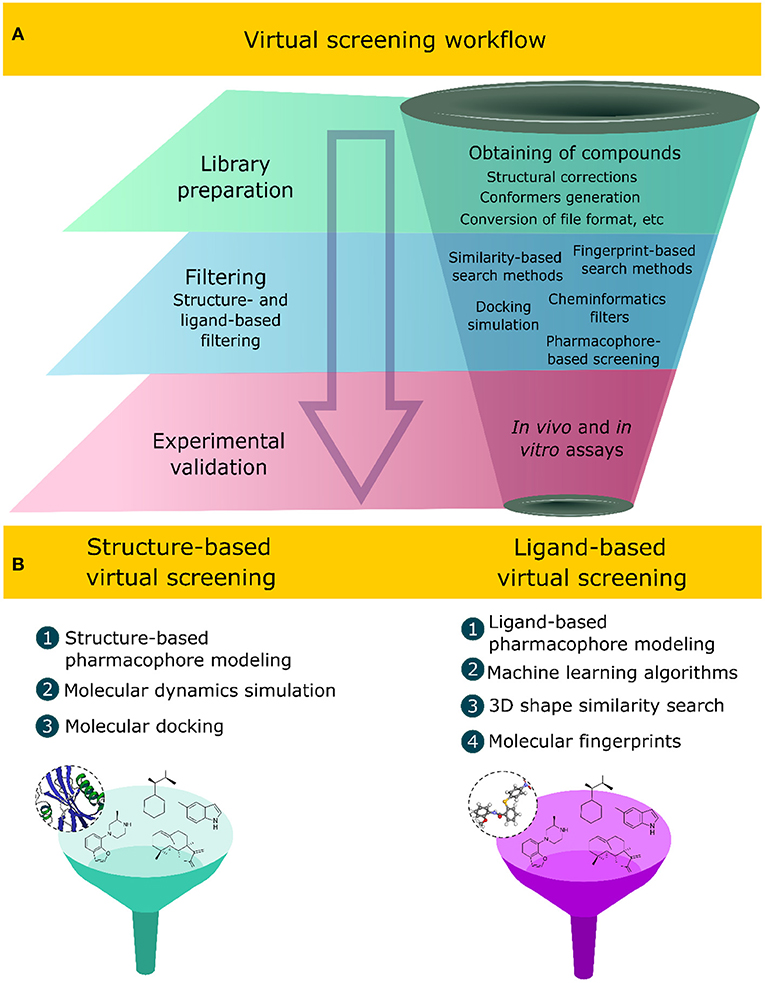
Virtual Screening
Computationally tests thousands of compounds to identify potential drug candidates.
Structure Optimization
Determines the most stable molecular structure.
9. Computational Chemistry Software
Various software programs are used in computational chemistry.
Examples include:
- Gaussian
- ORCA
- GAMESS
- NWChem
- VASP
These programs perform complex quantum chemical calculations and molecular simulations.
Visualization tools such as:
- PyMOL
- VMD
- Avogadro
help scientists analyze molecular structures.
10. Applications of Computational Chemistry
Computational chemistry has applications across many scientific fields.
Drug Discovery



Computational chemistry helps design new medicines.
Applications include:
- Predicting drug-target interactions
- Optimizing drug molecules
- Studying enzyme mechanisms
Computer-aided drug design significantly accelerates pharmaceutical research.
Materials Science
Computational methods help design advanced materials such as:
- Semiconductors
- Nanomaterials
- Catalysts
Simulations help predict material properties before synthesis.
Environmental Chemistry
Computational chemistry helps study:
- Atmospheric chemical reactions
- Pollutant degradation
- Climate modeling
These simulations support environmental protection efforts.
Nanotechnology
Computational models help design nanomaterials with specific properties.
Examples include:
- Carbon nanotubes
- Graphene structures
- Nanoparticle catalysts
11. Advantages of Computational Chemistry
Computational chemistry offers many benefits.
- Reduces experimental costs
- Allows study of dangerous reactions safely
- Provides detailed atomic-level information
- Accelerates scientific research
It complements experimental chemistry rather than replacing it.
12. Limitations of Computational Chemistry
Despite its advantages, computational chemistry has limitations.
Examples include:
- High computational cost
- Approximation errors
- Limitations in modeling extremely large systems
Advances in computing power and algorithms continue to address these challenges.
13. Role of Supercomputers and Artificial Intelligence
Modern computational chemistry often uses powerful supercomputers.
Supercomputers can perform millions of calculations simultaneously.
Artificial intelligence and machine learning are increasingly used to:
- Predict molecular properties
- Accelerate drug discovery
- Optimize chemical reactions
These technologies are transforming computational chemistry research.
14. Future of Computational Chemistry
The future of computational chemistry includes exciting developments.
Examples include:
- Quantum computing for chemical simulations
- AI-driven molecular design
- Large-scale biological simulations
- Digital chemical laboratories
These innovations will greatly expand our ability to understand and manipulate molecular systems.
15. Importance of Computational Chemistry
Computational chemistry has become an essential tool in modern science.
It helps scientists understand chemical systems that are difficult or impossible to study experimentally.
By combining theoretical models with computer simulations, researchers can explore chemical phenomena at unprecedented levels of detail.
Conclusion
Computational chemistry is a powerful field that applies computer simulations and theoretical methods to study chemical systems. Using quantum mechanics, molecular mechanics, and advanced algorithms, scientists can predict molecular structures, reaction mechanisms, and material properties. Computational chemistry complements experimental research by providing insights into molecular behavior at the atomic level. It plays a crucial role in drug discovery, materials science, nanotechnology, and environmental research. As computational power continues to grow and artificial intelligence becomes more integrated into scientific research, computational chemistry will continue to expand its impact on science and technology.
