



1. Introduction to Spectroscopy
Spectroscopy is a branch of science that studies the interaction between electromagnetic radiation and matter. It is one of the most important analytical techniques in chemistry, physics, astronomy, and biology. Spectroscopy allows scientists to investigate the structure, composition, and properties of atoms and molecules by analyzing how they absorb, emit, or scatter light.
The term spectroscopy originates from the Latin word spectrum, meaning image or apparition, and the Greek word skopein, meaning to observe or examine.
Spectroscopic techniques are widely used in scientific research because they provide detailed information about:
- Atomic and molecular structure
- Chemical composition of substances
- Electronic transitions
- Molecular vibrations and rotations
- Chemical bonding and interactions
Spectroscopy has numerous applications in fields such as:
- Analytical chemistry
- Medical diagnostics
- Environmental monitoring
- Pharmaceutical research
- Astronomy and astrophysics
- Materials science
Through spectroscopy, scientists can analyze substances at extremely small concentrations and gain insight into the microscopic world of atoms and molecules.
2. Electromagnetic Radiation
Spectroscopy is based on electromagnetic radiation, which consists of waves that travel through space at the speed of light.
Electromagnetic radiation has both wave-like and particle-like properties. It can be described by several important parameters:
Wavelength (λ)
Wavelength is the distance between two consecutive wave peaks.
Frequency (ν)
Frequency is the number of wave cycles passing a point per second.
Energy (E)
Energy is related to frequency through the equation:
E = hν
Where:
- E = energy
- h = Planck’s constant
- ν = frequency
The relationship between wavelength and frequency is:
c = λν
Where:
- c = speed of light
3. Electromagnetic Spectrum




The electromagnetic spectrum includes all types of electromagnetic radiation arranged according to wavelength or frequency.
Major regions of the spectrum include:
- Radio waves
- Microwaves
- Infrared radiation
- Visible light
- Ultraviolet radiation
- X-rays
- Gamma rays
Different spectroscopic techniques use different regions of the spectrum.
4. Principles of Spectroscopy
Spectroscopy is based on the principle that atoms and molecules can absorb or emit electromagnetic radiation when they change energy states.
When a molecule absorbs energy:
- Electrons move to higher energy levels.
When energy is released:
- Electrons return to lower energy levels.
This process produces spectral lines that are characteristic of specific elements or compounds.
Each element has a unique spectrum, making spectroscopy a powerful method for identifying substances.
5. Types of Spectroscopy
Spectroscopy can be classified into several types depending on how radiation interacts with matter.
Major categories include:
- Absorption spectroscopy
- Emission spectroscopy
- Fluorescence spectroscopy
- Scattering spectroscopy
6. Absorption Spectroscopy



In absorption spectroscopy, molecules absorb specific wavelengths of electromagnetic radiation.
The absorbed energy causes transitions between energy levels.
Common types include:
UV–Visible Spectroscopy
Studies electronic transitions in molecules.
Used to analyze organic and inorganic compounds.
Infrared Spectroscopy
Studies molecular vibrations.
Useful for identifying functional groups in organic compounds.
7. Emission Spectroscopy



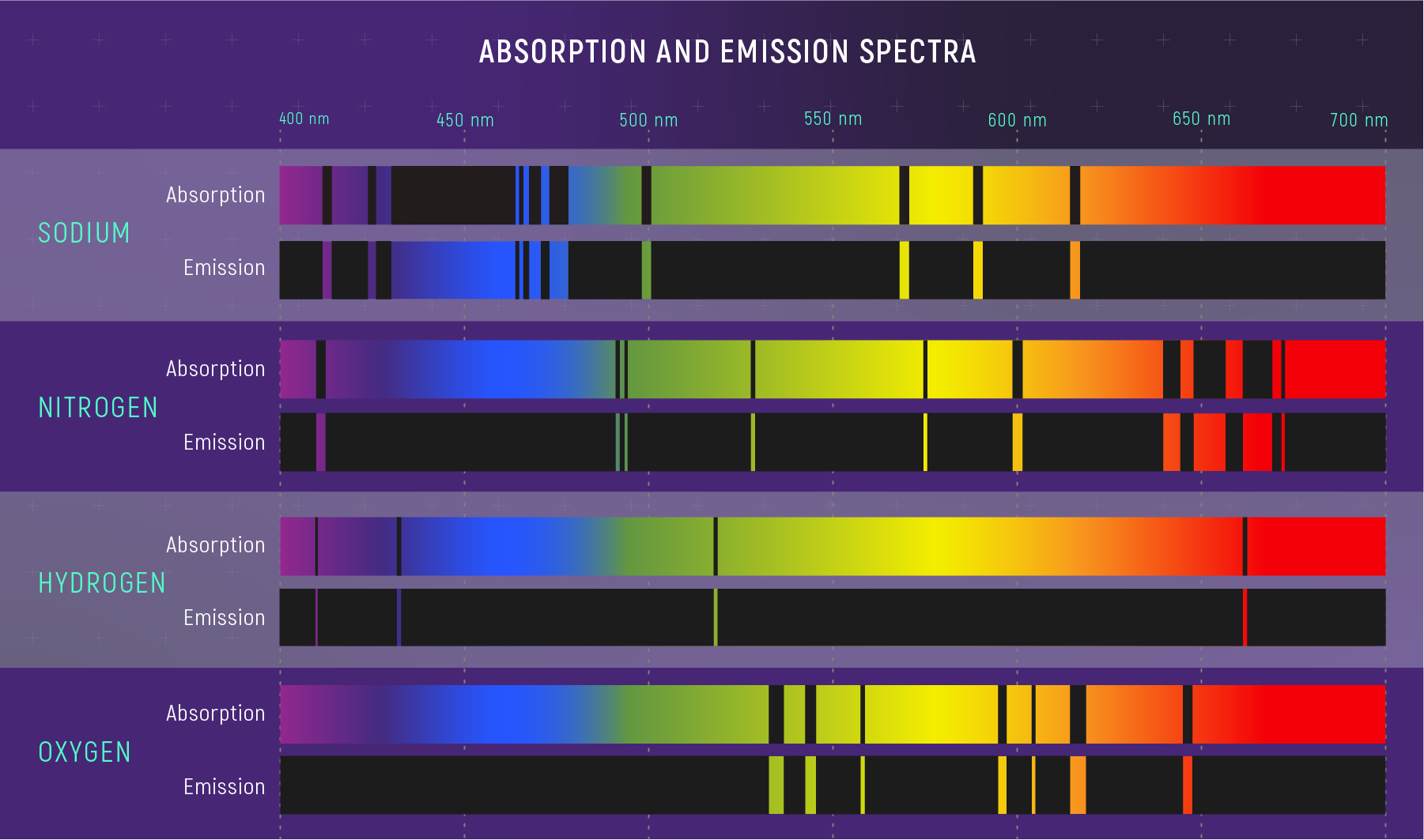
Emission spectroscopy occurs when atoms or molecules emit radiation after being excited.
Example:
Flame tests in chemistry.
Each element emits characteristic wavelengths of light.
Applications include:
- Element identification
- Metal analysis
- Astronomical studies
8. Infrared Spectroscopy
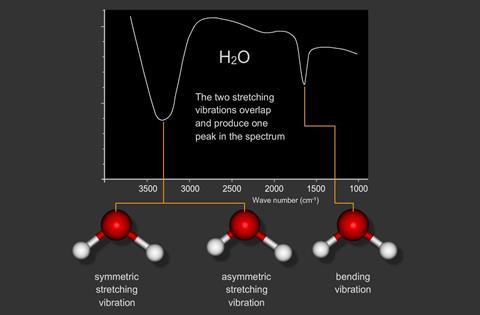



Infrared spectroscopy studies how molecules absorb infrared radiation.
IR radiation causes vibrational transitions in molecules.
Common vibrational motions include:
- Stretching
- Bending
IR spectroscopy is widely used to identify functional groups in organic molecules.
For example:
O–H stretch indicates alcohols.
C=O stretch indicates carbonyl compounds.
9. Nuclear Magnetic Resonance (NMR) Spectroscopy
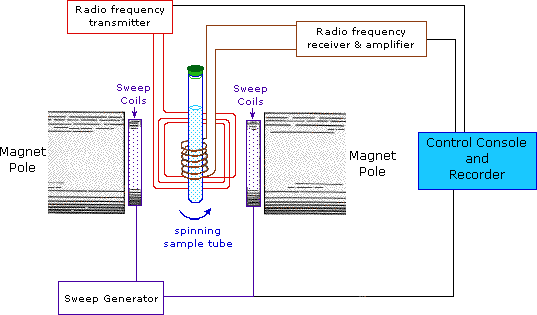

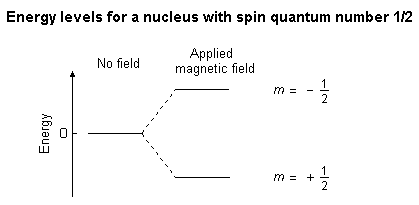

NMR spectroscopy studies the interaction between atomic nuclei and magnetic fields.
Nuclei such as hydrogen and carbon behave like tiny magnets.
When placed in a magnetic field, they absorb radiofrequency radiation.
NMR provides detailed information about:
- Molecular structure
- Chemical environment
- Connectivity of atoms
NMR is one of the most powerful tools in organic chemistry.
10. Mass Spectrometry
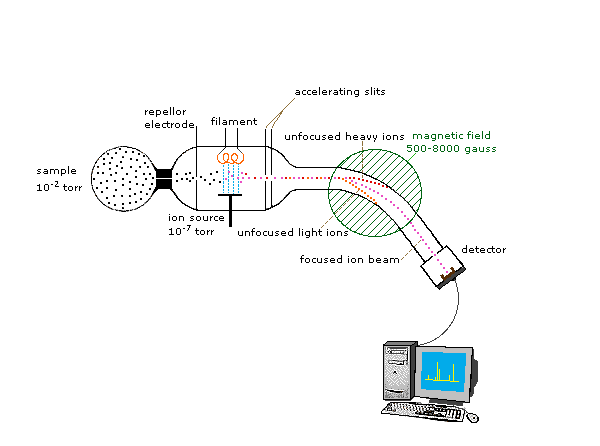

Mass spectrometry measures the mass-to-charge ratio of ions.
The process involves:
- Ionization of molecules
- Separation of ions
- Detection of ion masses
Mass spectrometry provides information about:
- Molecular weight
- Molecular structure
- Fragmentation patterns
It is widely used in chemical analysis and drug development.
11. UV–Visible Spectroscopy



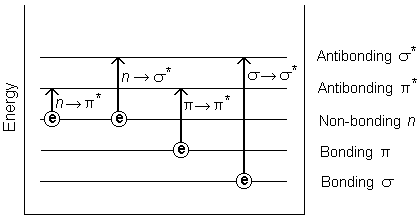
UV–Visible spectroscopy studies absorption of ultraviolet and visible light.
It involves electronic transitions in molecules.
The intensity of absorption follows Beer–Lambert law:
A = εcl
Where:
- A = absorbance
- ε = molar absorptivity
- c = concentration
- l = path length
UV–Vis spectroscopy is commonly used for concentration analysis.
12. Raman Spectroscopy
Raman spectroscopy studies scattering of light by molecules.
When light interacts with molecules, most photons scatter without energy change.
However, a small fraction undergo energy shifts called Raman scattering.
This technique provides information about molecular vibrations.
13. Applications of Spectroscopy
Spectroscopy has numerous applications across different scientific fields.
Analytical Chemistry
Identification and quantification of chemical substances.
Medicine
Medical imaging and diagnostics.
Environmental Science
Detection of pollutants and contaminants.
Astronomy
Determining composition of stars and planets.
Pharmaceutical Industry
Drug development and quality control.
14. Importance of Spectroscopy
Spectroscopy is one of the most powerful analytical tools in science.
It allows scientists to:
- Determine molecular structures
- Identify chemical compounds
- Study reaction mechanisms
- Analyze materials
Because of its versatility and precision, spectroscopy is essential for modern scientific research.
Conclusion
Spectroscopy is the study of how electromagnetic radiation interacts with matter. By analyzing absorption, emission, and scattering of light, scientists can obtain valuable information about the structure, composition, and behavior of atoms and molecules. Different spectroscopic techniques, such as UV–Visible spectroscopy, infrared spectroscopy, NMR spectroscopy, and mass spectrometry, are used to investigate different aspects of molecular structure. Spectroscopy plays a crucial role in chemistry, physics, biology, medicine, environmental science, and astronomy. Its ability to provide detailed molecular information makes it one of the most important analytical techniques in modern science.
