



1. Introduction to Chemical Equilibrium
Chemical equilibrium is a fundamental concept in chemistry that describes the state of a reversible chemical reaction where the forward and reverse reaction rates become equal. At this stage, the concentrations of reactants and products remain constant over time, even though the reactions continue to occur.
Many chemical reactions are reversible, meaning that the products formed can react again to produce the original reactants. Such reactions are represented by a double arrow (⇌) in chemical equations.
Example:
N₂ + 3H₂ ⇌ 2NH₃
In this reaction:
- Nitrogen and hydrogen combine to form ammonia (forward reaction)
- Ammonia can decompose back into nitrogen and hydrogen (reverse reaction)
Initially, only the forward reaction occurs. As products accumulate, the reverse reaction begins. Eventually, both reactions occur at equal rates, resulting in chemical equilibrium.
Chemical equilibrium is extremely important in:
- Industrial chemical processes
- Biological systems
- Environmental chemistry
- Pharmaceutical reactions
- Atmospheric chemistry
Understanding equilibrium allows scientists to predict reaction behavior and control chemical processes effectively.
2. Characteristics of Chemical Equilibrium
Chemical equilibrium has several key characteristics.
1. Dynamic Nature
Chemical equilibrium is dynamic, not static. This means reactions continue in both directions even though concentrations remain constant.
Reactant molecules continuously convert to products, and product molecules convert back into reactants.
2. Constant Concentrations
At equilibrium, the concentrations of reactants and products remain constant with time.
However, they are not necessarily equal.
3. Occurs in Closed Systems
Chemical equilibrium is achieved only in a closed system, where no substances enter or leave the reaction mixture.
4. Equal Reaction Rates
The rate of the forward reaction equals the rate of the reverse reaction.
5. Macroscopic Properties Remain Constant
Properties such as pressure, color, and concentration remain constant at equilibrium.
3. Types of Chemical Equilibrium
Chemical equilibrium is classified into two main types.
1. Homogeneous Equilibrium




Homogeneous equilibrium occurs when all reactants and products exist in the same phase.
Examples:
Gas-phase reactions
N₂ + 3H₂ ⇌ 2NH₃
Liquid-phase reactions
CH₃COOH + C₂H₅OH ⇌ CH₃COOC₂H₅ + H₂O
2. Heterogeneous Equilibrium


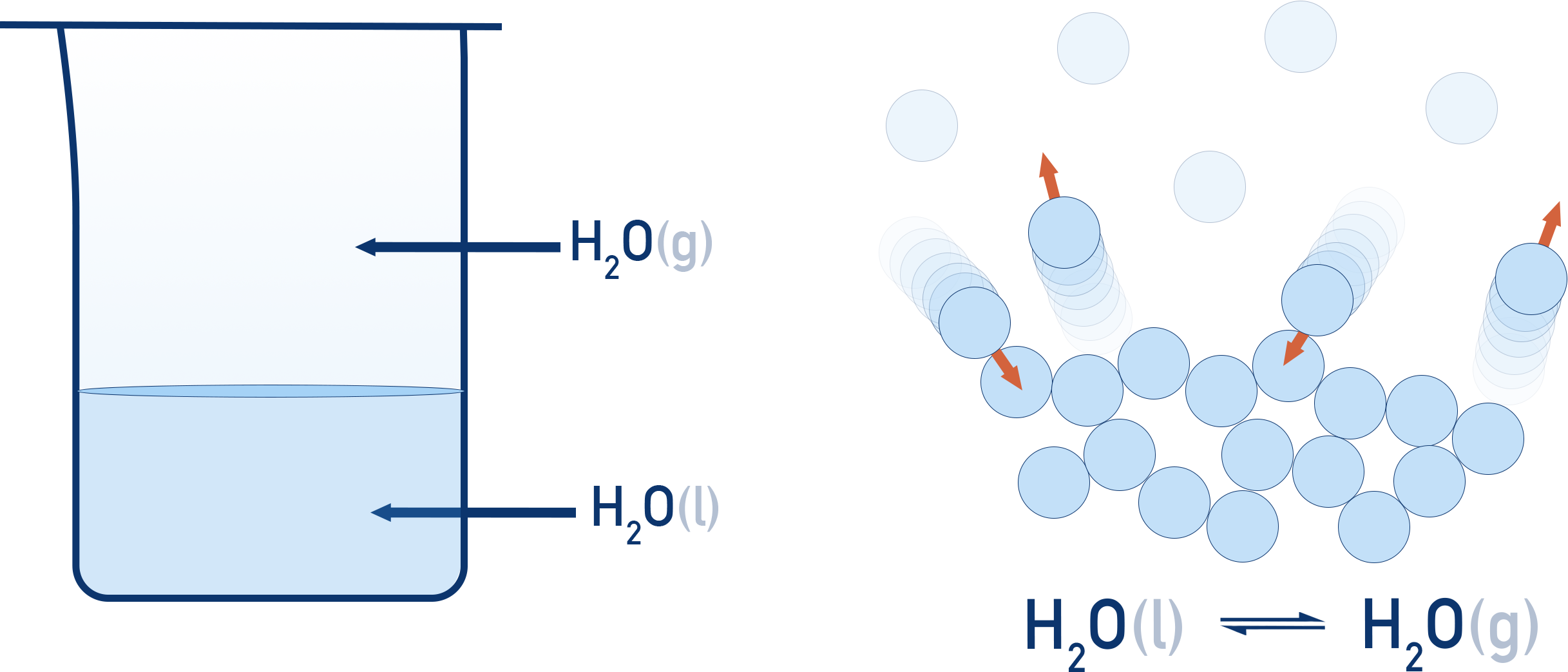
Heterogeneous equilibrium occurs when reactants and products exist in different phases.
Example:
CaCO₃ (s) ⇌ CaO (s) + CO₂ (g)
Here:
- CaCO₃ and CaO are solids
- CO₂ is gas
4. Law of Mass Action
The law of mass action was proposed by Guldberg and Waage.
It states:
The rate of a chemical reaction is proportional to the product of the active masses (concentrations) of reactants.
For a reaction:
aA + bB ⇌ cC + dD
Rate of forward reaction:
Ratef = kf[A]ᵃ[B]ᵇ
Rate of reverse reaction:
Rater = kr[C]ᶜ[D]ᵈ
At equilibrium:
Ratef = Rater
This leads to the equilibrium constant.
5. Equilibrium Constant
The equilibrium constant (K) is a numerical value that describes the ratio of product concentrations to reactant concentrations at equilibrium.
For a reaction:
aA + bB ⇌ cC + dD
Equilibrium constant expression:
[
K = \frac{[C]^c[D]^d}{[A]^a[B]^b}
]
Where:
- [A], [B], [C], [D] are equilibrium concentrations
- a, b, c, d are stoichiometric coefficients
Interpretation of Equilibrium Constant
If:
K > 1 → Products favored
K < 1 → Reactants favored
K = 1 → Comparable amounts
6. Types of Equilibrium Constants
1. Concentration Equilibrium Constant (Kc)
Kc is expressed using molar concentrations.
[
K_c = \frac{[Products]}{[Reactants]}
]
2. Pressure Equilibrium Constant (Kp)
For gaseous reactions, equilibrium can be expressed using partial pressures.
[
K_p = \frac{(P_C)^c (P_D)^d}{(P_A)^a (P_B)^b}
]
Relationship between Kp and Kc
[
K_p = K_c (RT)^{\Delta n}
]
Where:
Δn = difference in moles of gas.
7. Reaction Quotient (Q)
Reaction quotient is similar to equilibrium constant but applies to non-equilibrium conditions.
[
Q = \frac{[Products]}{[Reactants]}
]
Comparison:
Q < K → Reaction moves forward
Q > K → Reaction moves backward
Q = K → System at equilibrium
8. Le Chatelier’s Principle




Le Chatelier’s principle states:
If a system at equilibrium is disturbed, it adjusts itself to counteract the disturbance and restore equilibrium.
Disturbances include:
- Change in concentration
- Change in temperature
- Change in pressure
Effect of Concentration
Adding reactant shifts equilibrium toward products.
Adding product shifts equilibrium toward reactants.
Effect of Pressure
Increasing pressure favors the side with fewer gas molecules.
Example:
N₂ + 3H₂ ⇌ 2NH₃
Increasing pressure favors ammonia formation.
Effect of Temperature
Temperature affects equilibrium depending on reaction type.
Exothermic reaction:
Heat acts as product.
Increasing temperature shifts equilibrium toward reactants.
Endothermic reaction:
Heat acts as reactant.
Increasing temperature shifts equilibrium toward products.
9. Catalysts and Equilibrium
Catalysts do not change the position of equilibrium.
However, they speed up the attainment of equilibrium by accelerating both forward and reverse reactions equally.
10. Equilibrium in Industrial Processes



Chemical equilibrium is critical in industrial chemistry.
Haber Process
N₂ + 3H₂ ⇌ 2NH₃
Conditions:
High pressure
Moderate temperature
Iron catalyst
Contact Process
2SO₂ + O₂ ⇌ 2SO₃
Used to manufacture sulfuric acid.
Methanol Synthesis
CO + 2H₂ ⇌ CH₃OH
Important industrial reaction.
11. Equilibrium in Biological Systems
Chemical equilibrium plays a crucial role in biological processes.
Examples include:
- Oxygen binding to hemoglobin
- Enzyme reactions
- Acid-base balance in blood
Biological equilibrium helps maintain homeostasis in living organisms.
12. Dynamic Nature of Equilibrium
At the molecular level, reactions continue constantly.
Example:
In a closed container with nitrogen dioxide and dinitrogen tetroxide:
2NO₂ ⇌ N₂O₄
Molecules continuously interconvert.
However, concentrations remain constant.
13. Thermodynamics and Equilibrium
Chemical equilibrium is closely related to thermodynamics.
The Gibbs free energy relationship:
ΔG = −RT lnK
Where:
- ΔG = Gibbs free energy change
- R = gas constant
- T = temperature
- K = equilibrium constant
If:
ΔG < 0 → reaction spontaneous
ΔG = 0 → equilibrium
ΔG > 0 → reaction nonspontaneous
14. Factors Affecting Equilibrium
Several factors influence equilibrium conditions.
These include:
- Temperature
- Pressure
- Concentration
- Catalysts
However, only temperature changes the value of equilibrium constant.
15. Applications of Chemical Equilibrium
Chemical equilibrium has numerous practical applications.
Industrial Chemistry
Used to optimize chemical production.
Environmental Chemistry
Helps understand atmospheric reactions.
Biochemistry
Controls metabolic reactions.
Pharmaceutical Chemistry
Important in drug synthesis and stability.
16. Importance of Chemical Equilibrium
Chemical equilibrium helps scientists:
- Predict reaction direction
- Optimize industrial conditions
- Understand biological processes
- Control chemical reactions
- Improve chemical yields
Understanding equilibrium is essential for designing efficient chemical processes and maintaining biological systems.
Conclusion
Chemical equilibrium is a key concept in chemistry that describes the dynamic balance between forward and reverse reactions. At equilibrium, reaction rates become equal, and concentrations remain constant. The equilibrium constant provides a quantitative measure of this balance, while Le Chatelier’s principle explains how systems respond to disturbances. Chemical equilibrium plays an essential role in industrial processes, environmental chemistry, and biological systems. By understanding equilibrium principles, chemists can control reactions, optimize yields, and develop efficient chemical technologies.
