

1. Introduction to Le Chatelier’s Principle
Le Chatelier’s Principle is one of the most important concepts in chemical equilibrium. It explains how a chemical system at equilibrium responds to changes in external conditions such as concentration, temperature, and pressure.
The principle was proposed by the French chemist Henri Louis Le Chatelier in 1884. It provides a qualitative method for predicting how a system will react when equilibrium is disturbed.
In simple terms, Le Chatelier’s Principle states:
When a system at equilibrium is subjected to a change in concentration, temperature, pressure, or volume, the system adjusts itself in a way that counteracts the imposed change and establishes a new equilibrium.
This principle helps chemists and engineers understand and control chemical reactions in laboratory experiments and industrial processes.
For example, in the synthesis of ammonia:
N₂ + 3H₂ ⇌ 2NH₃
Changes in pressure or temperature shift the equilibrium position. Understanding these shifts allows industries to optimize ammonia production.
Le Chatelier’s Principle is widely applied in:
- Industrial chemical manufacturing
- Environmental chemistry
- Biological systems
- Atmospheric chemistry
- Pharmaceutical processes
2. Concept of Chemical Equilibrium
Before understanding Le Chatelier’s Principle, it is essential to understand chemical equilibrium.
Chemical equilibrium occurs when:
- Forward reaction rate = Reverse reaction rate
- Concentrations of reactants and products remain constant
Consider the reaction:
A + B ⇌ C + D
At equilibrium:
Rate of forward reaction = Rate of reverse reaction
Although concentrations remain constant, reactions continue at the molecular level. Therefore equilibrium is dynamic, not static.
When a disturbance occurs, the equilibrium position shifts until a new equilibrium state is reached.
3. Statement of Le Chatelier’s Principle
The formal statement of Le Chatelier’s Principle is:
If a system at equilibrium is disturbed by a change in concentration, temperature, pressure, or volume, the system will shift in a direction that reduces the effect of the disturbance and re-establishes equilibrium.
In other words:
The system tries to oppose the change.
This response helps maintain stability in chemical systems.
4. Disturbances That Affect Equilibrium
The equilibrium position can be disturbed by several factors:
- Change in concentration
- Change in pressure
- Change in temperature
- Change in volume
- Addition of inert gas
- Presence of catalyst
Each of these factors affects equilibrium differently.
5. Effect of Concentration Change




Changing the concentration of reactants or products shifts equilibrium.
Increasing Reactant Concentration
If reactant concentration increases, equilibrium shifts toward products.
Example:
N₂ + 3H₂ ⇌ 2NH₃
Adding more hydrogen increases ammonia production.
Increasing Product Concentration
If product concentration increases, equilibrium shifts toward reactants.
This consumes excess product.
Removing Reactants
Removing reactants shifts equilibrium toward reactants.
Removing Products
Removing products shifts equilibrium toward products.
This is often used in industrial chemistry to increase yield.
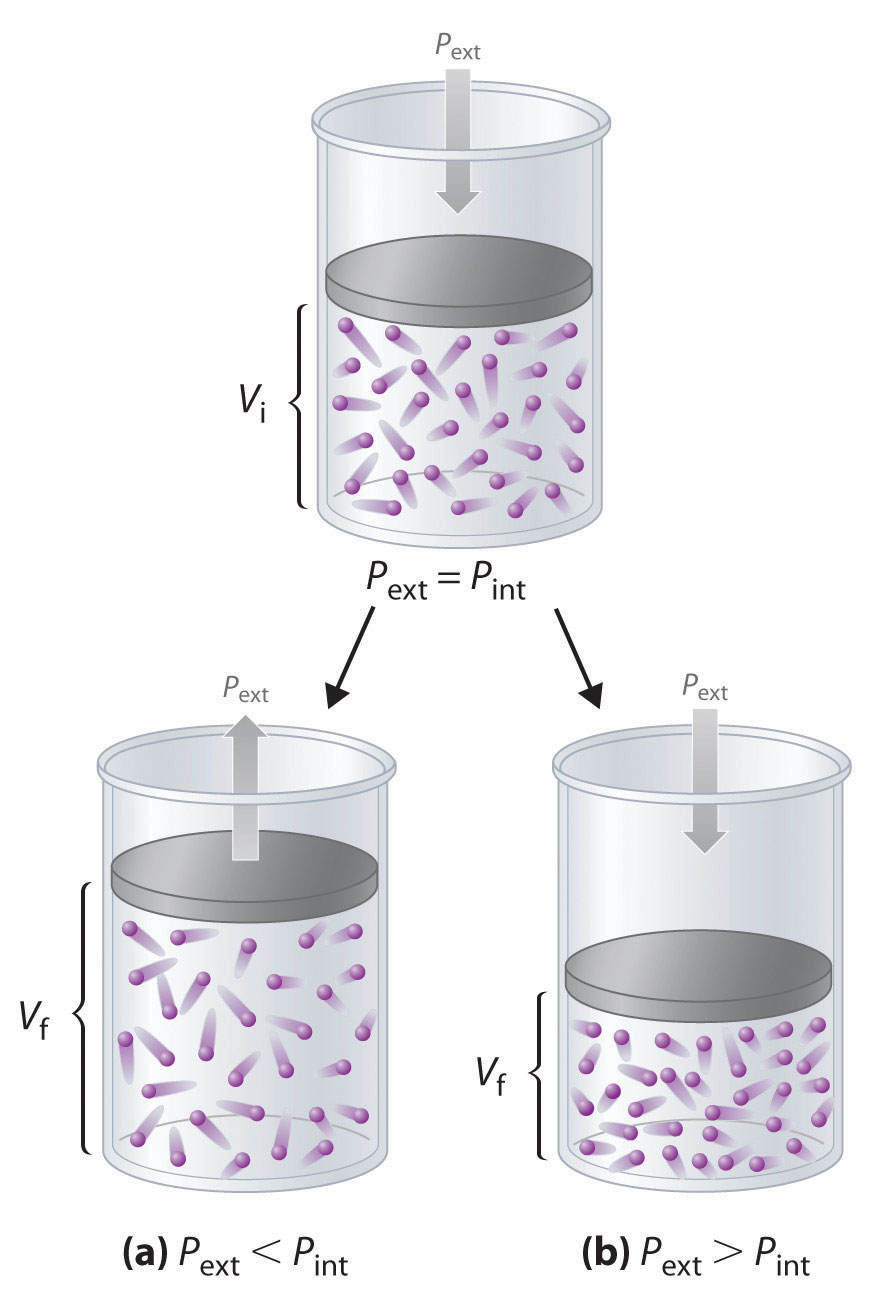
6. Effect of Pressure Change


Pressure changes affect equilibrium in gaseous reactions.
Increasing pressure favors the side with fewer moles of gas.
Decreasing pressure favors the side with more moles of gas.
Example:
N₂ + 3H₂ ⇌ 2NH₃
Gas moles:
Left side = 4 moles
Right side = 2 moles
Increasing pressure shifts equilibrium toward ammonia formation.
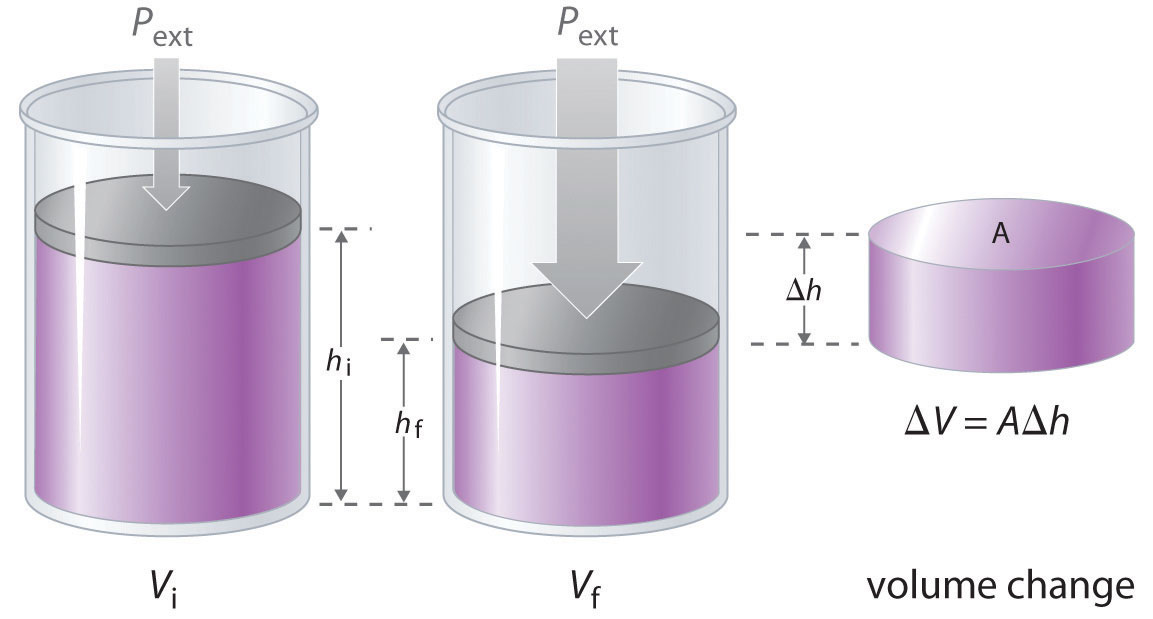
7. Effect of Volume Change
Volume changes are closely related to pressure.
Decreasing volume increases pressure.
Increasing volume decreases pressure.
Therefore:
Reducing volume favors the side with fewer gas molecules.
Increasing volume favors the side with more gas molecules.
8. Effect of Temperature



Temperature changes affect equilibrium differently from other factors.
Temperature changes alter the equilibrium constant.
Exothermic Reactions
Heat acts as a product.
Example:
N₂ + 3H₂ ⇌ 2NH₃ + Heat
Increasing temperature shifts equilibrium toward reactants.
Decreasing temperature shifts equilibrium toward products.
Endothermic Reactions
Heat acts as a reactant.
Increasing temperature shifts equilibrium toward products.
Decreasing temperature shifts equilibrium toward reactants.
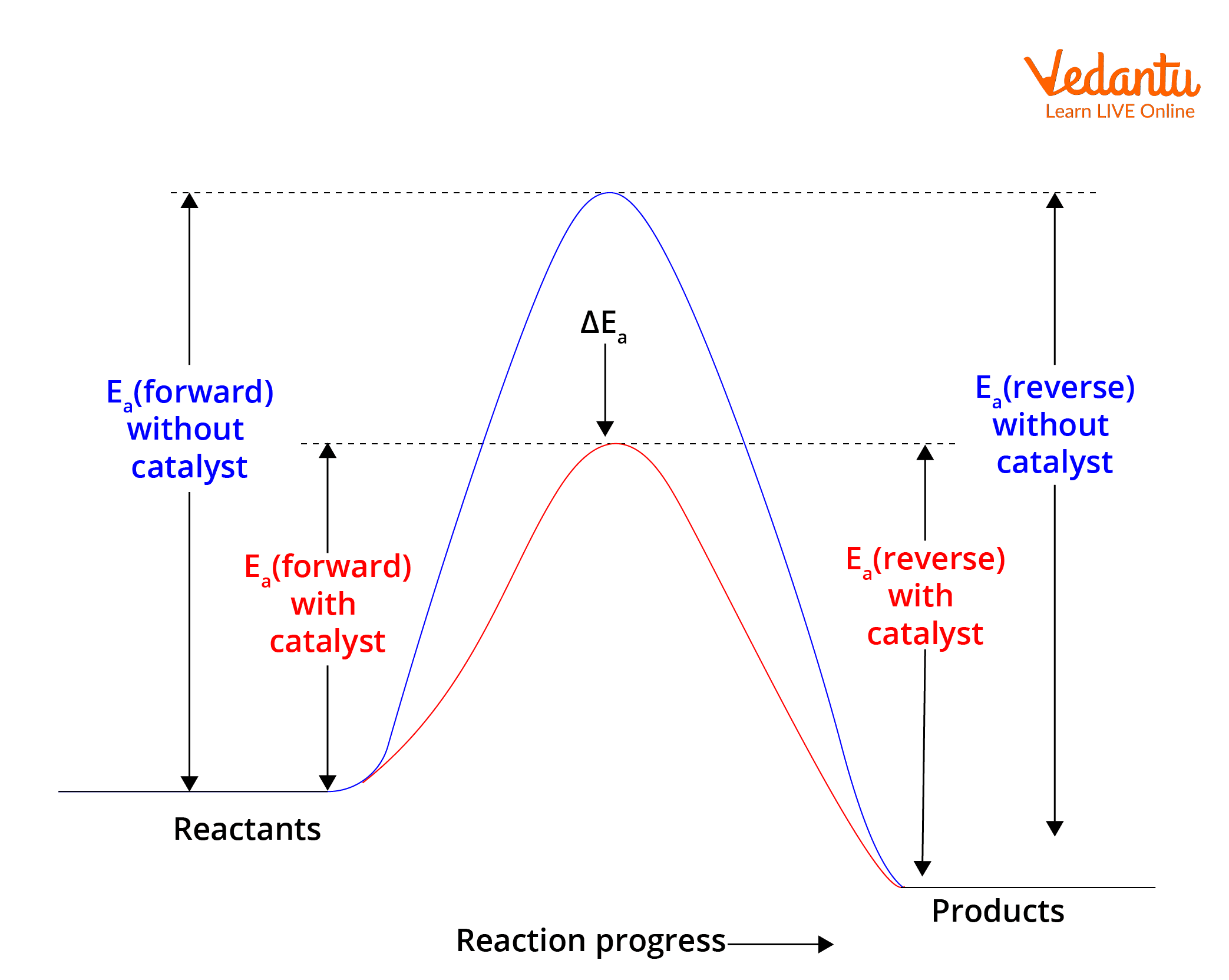
9. Effect of Catalysts
Catalysts do not change equilibrium position.
However, catalysts speed up both forward and reverse reactions.
Therefore:
- Equilibrium is reached faster
- Equilibrium composition remains unchanged
Catalysts lower activation energy but do not affect equilibrium constant.
10. Effect of Inert Gas
Adding inert gas does not affect equilibrium if volume remains constant.
However, if pressure changes due to added gas, equilibrium may shift depending on gas mole changes.
11. Graphical Representation of Equilibrium Shift



Graphs help visualize equilibrium changes.
Typical graphs show:
- Concentration vs time
- Reaction rate vs time
When equilibrium is disturbed:
- Concentrations change
- Reaction rates become unequal
- System adjusts
- New equilibrium is established
12. Industrial Applications of Le Chatelier’s Principle




Le Chatelier’s Principle is extremely important in industrial chemistry.
Industries manipulate conditions to maximize product yield.
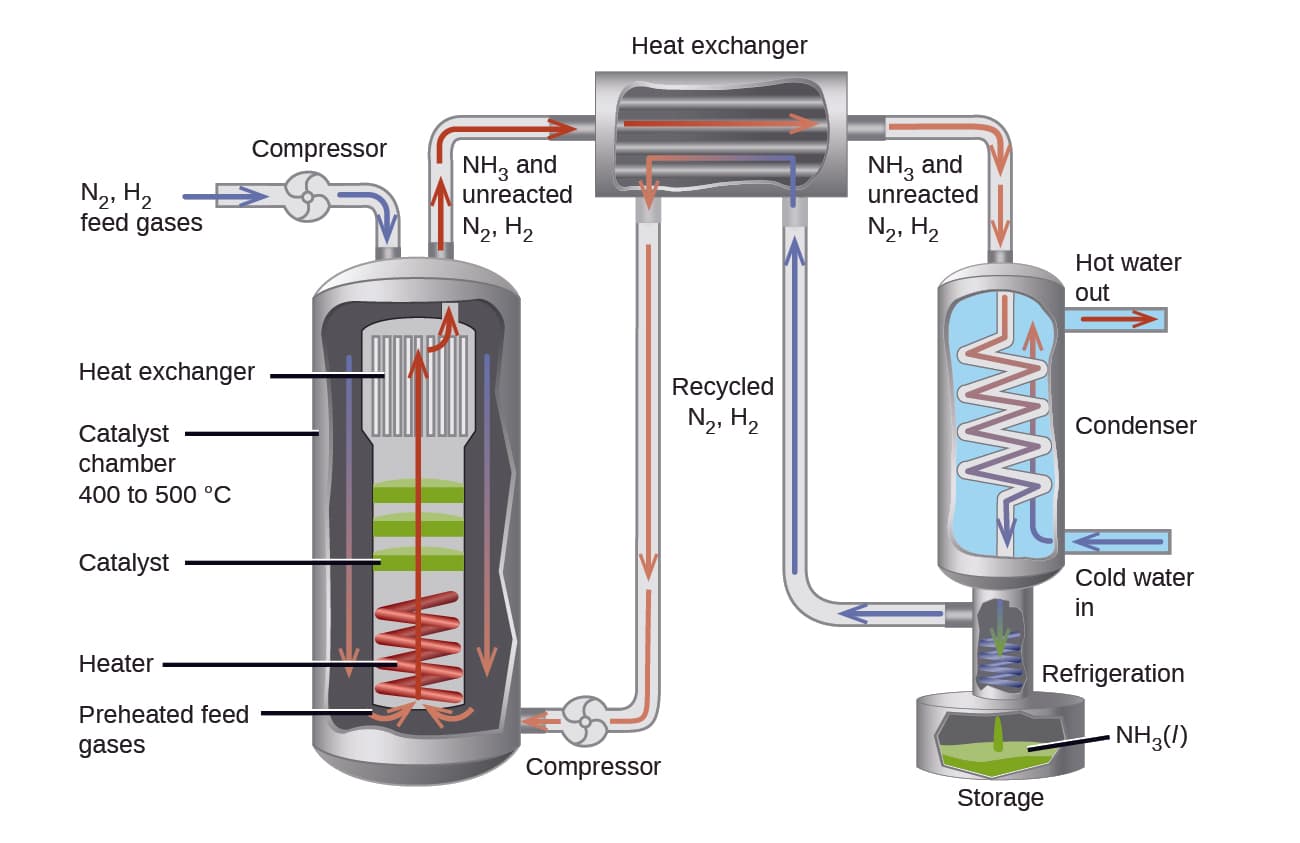
Haber Process
Reaction:
N₂ + 3H₂ ⇌ 2NH₃
Conditions used:
High pressure
Moderate temperature
Iron catalyst
High pressure favors ammonia production.
Contact Process
Reaction:
2SO₂ + O₂ ⇌ 2SO₃
Used to produce sulfuric acid.
Moderate temperature and catalysts optimize equilibrium yield.
Methanol Production
CO + 2H₂ ⇌ CH₃OH
High pressure and catalysts improve production.
13. Biological Applications
Le Chatelier’s Principle also applies to biological systems.
Example:
Oxygen binding with hemoglobin.
Hb + O₂ ⇌ HbO₂
When oxygen concentration increases, more oxyhemoglobin forms.
In tissues where oxygen concentration decreases, oxygen is released.
This equilibrium maintains oxygen supply in the body.
14. Environmental Applications
Le Chatelier’s Principle helps explain environmental chemical processes.
Examples include:
- Carbon dioxide equilibrium in oceans
- Atmospheric ozone formation
- Acid rain formation
Understanding equilibrium shifts helps scientists predict environmental changes.
15. Mathematical Relation with Equilibrium Constant
Le Chatelier’s Principle explains qualitative shifts, while equilibrium constants provide quantitative information.
The relationship between equilibrium and thermodynamics is:
ΔG = −RT lnK
Where:
ΔG = Gibbs free energy change
R = gas constant
T = temperature
K = equilibrium constant
If ΔG = 0, the system is at equilibrium.
16. Importance of Le Chatelier’s Principle
Le Chatelier’s Principle helps chemists:
- Predict reaction behavior
- Control chemical reactions
- Optimize industrial processes
- Understand biological systems
- Study environmental chemistry
Without this principle, designing efficient chemical processes would be extremely difficult.
17. Limitations of Le Chatelier’s Principle
Although useful, the principle has limitations.
It provides qualitative predictions, not quantitative results.
Complex reactions may require detailed mathematical analysis.
Despite these limitations, it remains a fundamental tool in chemistry.
Conclusion
Le Chatelier’s Principle is a cornerstone of chemical equilibrium theory. It explains how equilibrium systems respond to external disturbances such as changes in concentration, pressure, temperature, and volume. By shifting the equilibrium position to counteract these disturbances, chemical systems maintain dynamic balance. This principle is widely applied in industrial chemical production, biological processes, environmental chemistry, and laboratory experiments. Understanding Le Chatelier’s Principle allows chemists to control reaction conditions, improve product yields, and gain deeper insight into chemical systems.
