
1. Introduction to Specific Heat Capacity
Specific heat capacity is an important concept in thermodynamics and heat transfer that describes how much heat energy is required to change the temperature of a substance. Different materials respond differently when heat is supplied to them. Some substances heat up quickly with a small amount of heat, while others require a large amount of heat to increase their temperature.
Specific heat capacity helps explain these differences. It measures the amount of heat energy required to raise the temperature of a unit mass of a substance by one degree Celsius (or one Kelvin). This property is essential for understanding how materials absorb, store, and release heat energy.
The concept of specific heat capacity plays an important role in many fields including physics, chemistry, engineering, meteorology, and environmental science. It helps scientists understand climate behavior, energy transfer in oceans and atmosphere, heating and cooling processes, and the design of heating systems.
For example, water has a very high specific heat capacity compared to most substances. This means water can absorb large amounts of heat without a large change in temperature. Because of this property, oceans and lakes help regulate Earth’s climate by storing heat energy and releasing it slowly.
Specific heat capacity also influences industrial processes, cooking, refrigeration, and many technological systems where temperature control is important.
2. Definition of Specific Heat Capacity
Specific heat capacity can be defined as:
The amount of heat energy required to raise the temperature of one unit mass of a substance by one degree Celsius or one Kelvin.
Mathematically, it can be expressed as:
Q = mcΔT
Where:
Q = heat energy supplied
m = mass of the substance
c = specific heat capacity
ΔT = change in temperature
The formula shows that the amount of heat required to change temperature depends on three factors:
- Mass of the substance
- Specific heat capacity of the substance
- Temperature change
The higher the specific heat capacity, the more heat energy is needed to increase the temperature.
3. Units of Specific Heat Capacity
The SI unit of specific heat capacity is:
Joule per kilogram per Kelvin (J/kg·K)
This unit indicates the number of joules of heat required to raise the temperature of one kilogram of a substance by one Kelvin.
Other units sometimes used include:
- cal/g°C (calories per gram per degree Celsius)
- kJ/kg·K (kilojoules per kilogram per Kelvin)
In older systems, specific heat capacity was measured in calories. One calorie is the heat required to raise the temperature of 1 gram of water by 1°C.
Conversion relationship:
1 calorie = 4.186 joules
4. Physical Meaning of Specific Heat Capacity
Specific heat capacity reflects how much heat energy a substance can store.
If a substance has:
High specific heat capacity → requires more heat to increase temperature.
Low specific heat capacity → requires less heat to increase temperature.
For example:
Water has a high specific heat capacity. It takes a lot of heat to warm water.
Metals like copper and aluminum have lower specific heat capacities. They heat up more quickly.
This difference explains why metal utensils become hot quickly while water takes longer to heat.
5. Molecular Explanation of Specific Heat Capacity

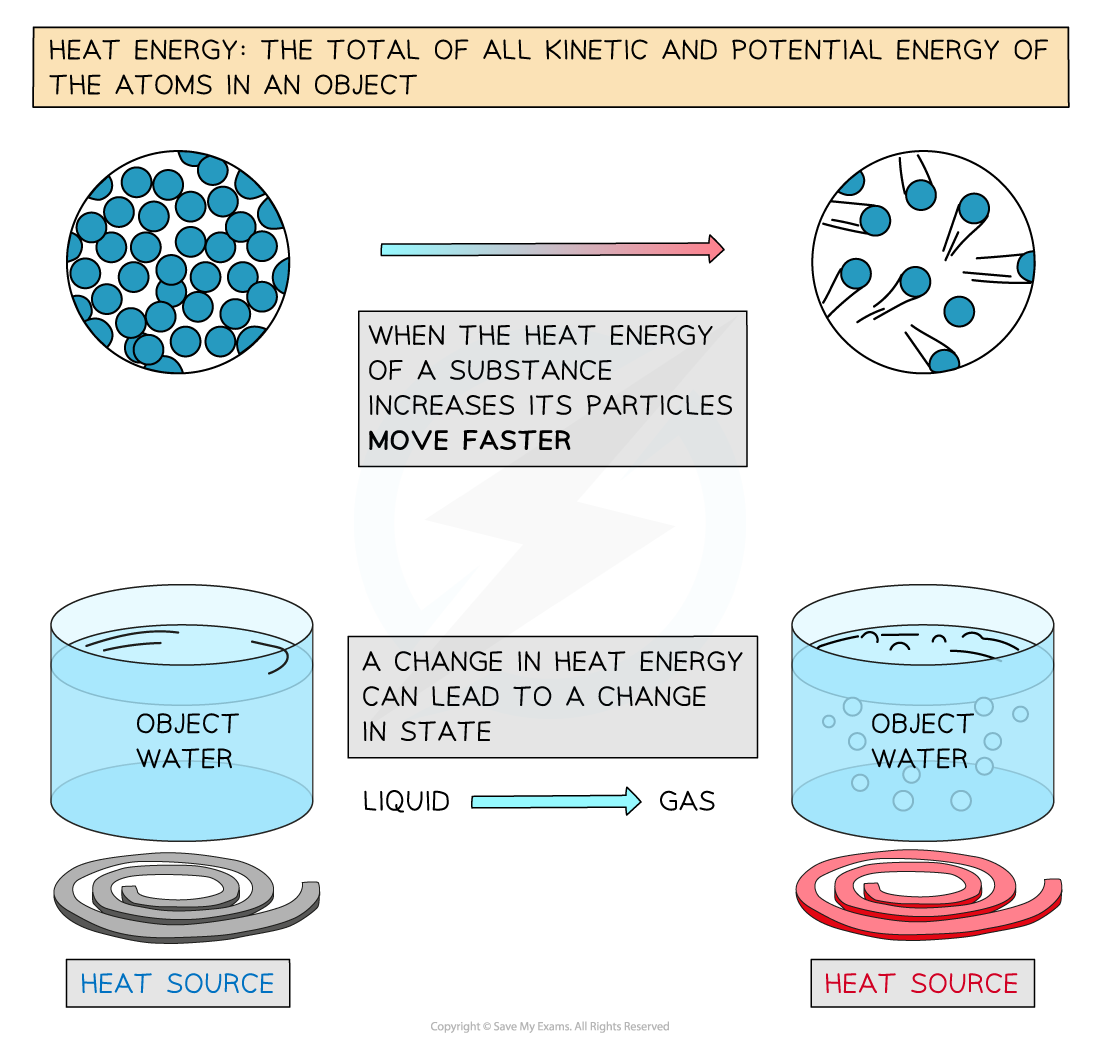
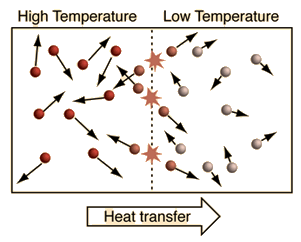
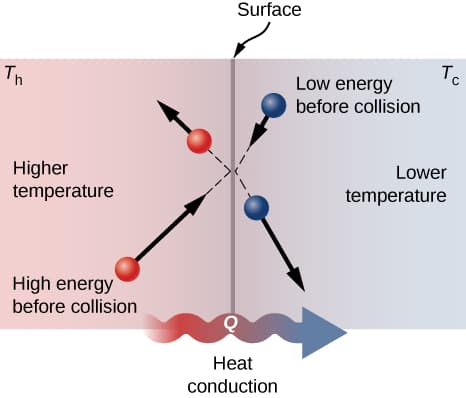
The behavior of specific heat capacity can be explained using the kinetic theory of matter.
When heat energy is supplied to a substance:
- The molecules absorb energy.
- Their kinetic energy increases.
- The molecules move faster or vibrate more strongly.
- The temperature rises.
However, not all heat energy directly increases temperature.
Some energy may go into:
- Rotational motion of molecules
- Vibrational motion of molecules
- Intermolecular forces
Because different substances have different molecular structures, they absorb and distribute heat differently. This results in different specific heat capacities.
Substances with complex molecular structures usually have higher specific heat capacities because energy can be distributed among many molecular motions.
6. Specific Heat Capacity of Common Substances
Different materials have different specific heat capacities.
Some approximate values include:
Water: about 4186 J/kg·K
Aluminum: about 900 J/kg·K
Copper: about 385 J/kg·K
Iron: about 450 J/kg·K
Air: about 1000 J/kg·K
Water’s specific heat capacity is much higher than most materials. This property makes water extremely important for temperature regulation in nature and engineering.
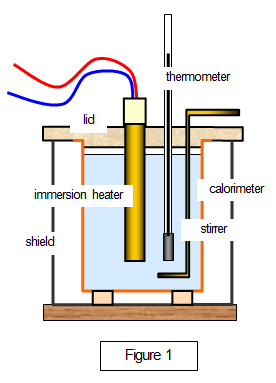
7. Calorimetry and Measurement of Specific Heat Capacity


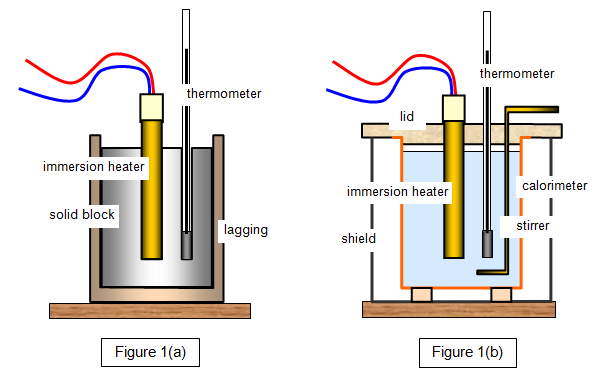

Specific heat capacity can be measured experimentally using a device called a calorimeter.
Calorimetry is the science of measuring heat transfer.
In a typical experiment:
- A known mass of a substance is heated.
- It is placed in water inside a calorimeter.
- Temperature changes are recorded.
- Heat transfer is calculated using formulas.
The heat lost by the hot substance equals the heat gained by the cooler substance.
Using this principle, the specific heat capacity of unknown materials can be determined.
8. Heat Capacity vs Specific Heat Capacity
Although related, heat capacity and specific heat capacity are different concepts.
Heat capacity refers to the total heat required to change the temperature of an entire object by one degree.
Specific heat capacity refers to the heat required per unit mass.
Mathematically:
Heat Capacity = mc
Where:
m = mass
c = specific heat capacity
Thus, heat capacity depends on both the material and its mass.
9. Importance of Specific Heat Capacity in Nature
Specific heat capacity plays a major role in many natural processes.
Climate Regulation
Oceans absorb large amounts of heat because water has a high specific heat capacity.
This helps regulate global temperatures.
Weather Patterns
Large bodies of water warm and cool more slowly than land.
This difference influences weather and climate systems.
Survival of Aquatic Life
Water temperature changes slowly, allowing aquatic organisms to survive even during temperature fluctuations.
10. Applications of Specific Heat Capacity
Specific heat capacity is important in many technological and industrial applications.
Cooking
Water’s high specific heat capacity helps cook food evenly.
Cooling Systems
Car engines use water or coolant with high specific heat capacity to absorb heat.
Thermal Energy Storage
Certain materials store heat energy efficiently due to high specific heat capacity.
Building Design
Materials with appropriate heat capacities help regulate indoor temperatures.
Climate Engineering
Understanding specific heat capacity helps predict climate change effects.
11. Specific Heat Capacity and Phase Changes
Specific heat capacity is related to phase changes of matter.
When substances change phase (such as melting or boiling), heat energy may not change temperature immediately.
Instead, energy is used to overcome intermolecular forces.
This heat is called latent heat.
Although specific heat capacity describes temperature changes, latent heat describes energy required for phase transitions.
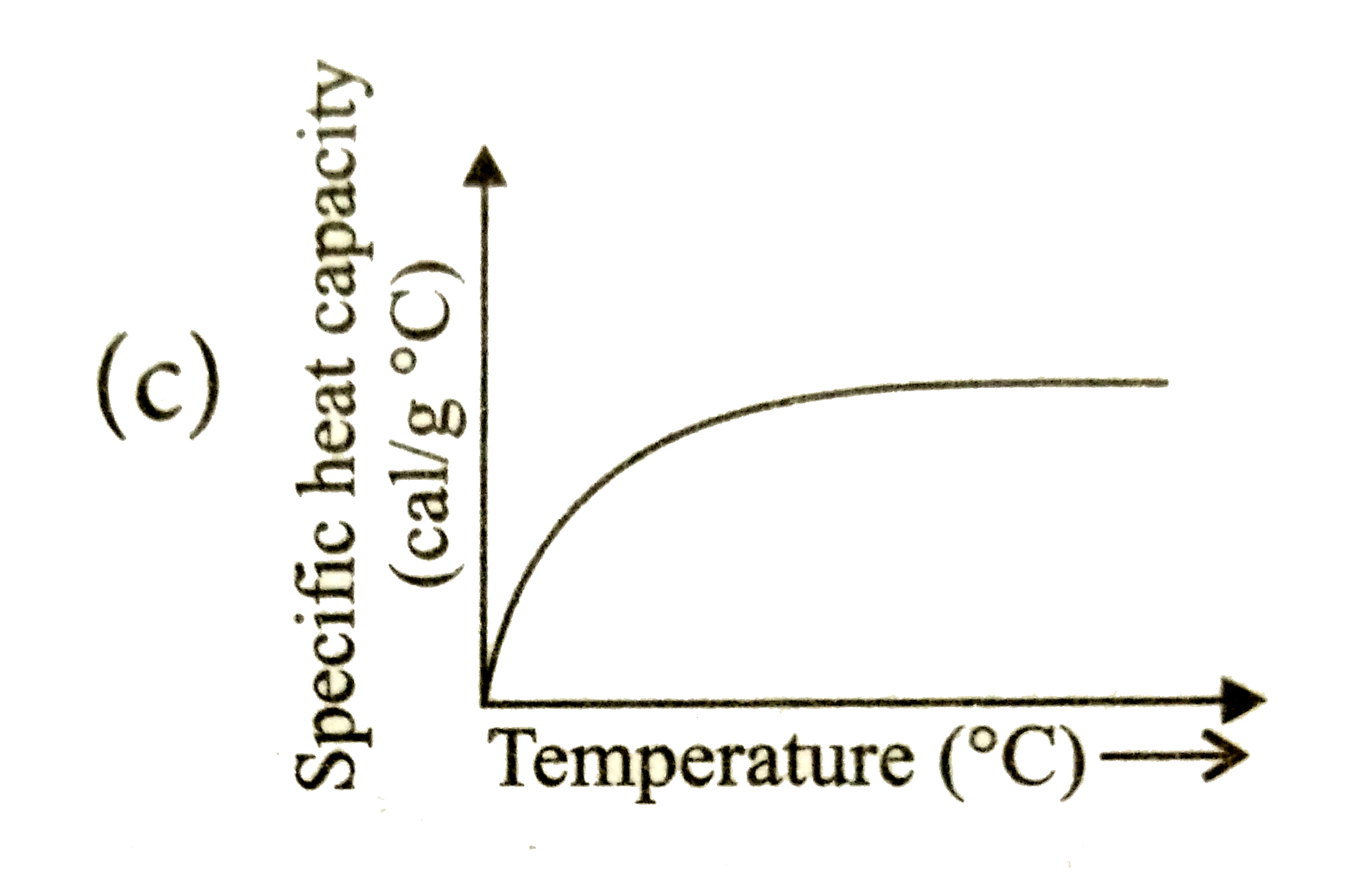
12. Factors Affecting Specific Heat Capacity
Several factors influence the specific heat capacity of a substance.
These include:
- Molecular structure
- Phase of matter
- Temperature
- Pressure
For example, solids, liquids, and gases of the same substance may have different specific heat capacities.
Complex molecules usually have higher heat capacities because they can store energy in multiple ways.
13. Specific Heat Capacity in Engineering
Engineers must consider specific heat capacity when designing machines and systems.
Examples include:
- Heat exchangers
- Power plants
- Air conditioning systems
- Refrigeration systems
- Industrial heating processes
Choosing materials with appropriate heat capacities ensures efficient heat transfer and temperature control.
14. Specific Heat Capacity in Environmental Science
Specific heat capacity is important in environmental studies.
It influences:
- Ocean temperature changes
- Atmospheric heat balance
- Climate systems
- Global warming patterns
Because water stores large amounts of heat, oceans act as thermal reservoirs that stabilize Earth’s climate.
15. Importance of Specific Heat Capacity
Specific heat capacity is one of the most important properties of matter in thermodynamics.
It helps scientists and engineers understand how materials absorb and store energy.
This concept is essential for:
- Energy management
- Climate science
- Industrial heating processes
- Environmental engineering
- Biological temperature regulation
Without knowledge of specific heat capacity, it would be difficult to control temperature in many technological systems.
Conclusion
Specific heat capacity is a fundamental concept in thermodynamics that describes the amount of heat required to raise the temperature of a substance. It varies from one material to another depending on molecular structure and physical properties.
Substances with high specific heat capacity, such as water, can absorb large amounts of heat with only small temperature changes. This property plays a crucial role in regulating Earth’s climate and supporting life.
Specific heat capacity is widely used in scientific research, industrial processes, engineering design, and environmental studies. It helps explain how heat energy is stored, transferred, and utilized in different systems.
Understanding specific heat capacity allows scientists and engineers to design efficient thermal systems, predict environmental changes, and manage energy resources effectively.
Tags: