



1. Introduction to Heat Transfer
Heat transfer is a fundamental process in physics and thermodynamics that explains how thermal energy moves from one object or region to another. Whenever there is a temperature difference between two bodies or areas, heat energy flows from the hotter region to the colder region until thermal equilibrium is reached.
Heat transfer plays a vital role in both natural processes and technological systems. It influences weather patterns, ocean currents, climate systems, biological processes, industrial manufacturing, and household appliances. For example, heat transfer occurs when water boils in a pot, when the Sun warms the Earth, when an engine converts heat into mechanical energy, and when a refrigerator removes heat from food to keep it cold.
There are three primary mechanisms through which heat transfer occurs:
- Conduction
- Convection
- Radiation
Each of these processes operates under different physical principles and occurs in different environments. Conduction involves heat transfer through direct contact between particles. Convection involves heat transfer through the movement of fluids such as liquids or gases. Radiation involves the transfer of heat through electromagnetic waves and does not require a medium.
Understanding heat transfer is essential in many areas of science and engineering, including mechanical engineering, environmental science, architecture, electronics, and energy systems.
2. Concept of Heat Transfer
Heat transfer occurs whenever there is a difference in temperature between two systems.
According to thermodynamics:
Heat always flows from a region of higher temperature to a region of lower temperature.
This flow continues until both systems reach the same temperature, a condition known as thermal equilibrium.
The rate and efficiency of heat transfer depend on several factors such as:
- Temperature difference
- Material properties
- Surface area
- Distance between objects
- Nature of the medium
Heat transfer mechanisms explain how energy moves within solids, liquids, gases, and even empty space.
3. Conduction



3.1 Definition of Conduction
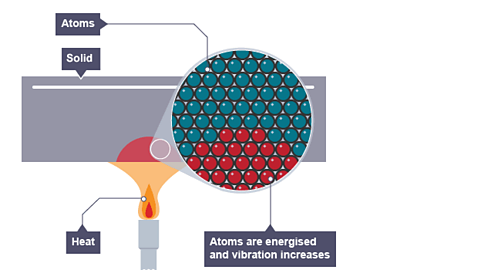
Conduction is the transfer of heat through a material without the movement of the material as a whole.
In conduction, heat energy is transferred through microscopic collisions between atoms, molecules, or electrons.
This process occurs primarily in solids, where particles are closely packed.
3.2 Mechanism of Conduction
When one part of a solid is heated:
- The particles in that region gain kinetic energy.
- These particles vibrate more vigorously.
- They collide with neighboring particles.
- Energy is transferred through the material.
Thus heat gradually spreads from the hot region to the cooler region.
In metals, conduction occurs very efficiently because free electrons carry energy quickly through the material.
3.3 Thermal Conductivity
The ability of a material to conduct heat is called thermal conductivity.
Materials with high thermal conductivity transfer heat rapidly.
Examples of good conductors include:
- Copper
- Aluminum
- Silver
- Iron
Materials with low thermal conductivity are called insulators.
Examples include:
- Wood
- Plastic
- Rubber
- Air
These materials resist the flow of heat and are used in thermal insulation.
3.4 Mathematical Description of Conduction
Heat conduction is described by Fourier’s Law.
Heat transfer rate:
Q/t = kA (ΔT / L)
Where:
Q = heat transferred
t = time
k = thermal conductivity
A = cross-sectional area
ΔT = temperature difference
L = length of material
This equation shows that heat transfer increases with larger temperature difference and larger surface area.
3.5 Examples of Conduction
Common examples include:
- A metal spoon becoming hot in a cup of tea.
- Iron rods heating when placed in a fire.
- Cooking utensils transferring heat from stove to food.
- Heat traveling through walls of buildings.
Conduction is very important in engineering applications such as heat exchangers and thermal insulation.
4. Convection




4.1 Definition of Convection
Convection is the transfer of heat through the movement of fluids such as liquids and gases.
Unlike conduction, convection involves the bulk motion of matter.
When a fluid is heated, its density decreases and it rises. Cooler fluid then moves in to replace it, creating a continuous circulation pattern called convection currents.
4.2 Mechanism of Convection
The process of convection occurs in several steps:
- A fluid near a heat source becomes warmer.
- Warm fluid expands and becomes less dense.
- The warm fluid rises upward.
- Cooler fluid moves downward to replace it.
- This circulation continues, transferring heat through the fluid.
4.3 Types of Convection
Natural Convection
Natural convection occurs due to density differences caused by temperature variations.
Examples include:
- Warm air rising above heaters
- Ocean currents
- Atmospheric circulation
Forced Convection
Forced convection occurs when an external force moves the fluid.
Examples include:
- Fans cooling electronic devices
- Pumps circulating coolant in engines
- Air conditioning systems
Forced convection increases the rate of heat transfer.
4.4 Examples of Convection
Convection occurs in many everyday situations.
Examples include:
- Boiling water in a pot
- Warm air rising from heaters
- Sea breezes and land breezes
- Circulation of air in rooms
Convection is also responsible for large-scale phenomena such as weather systems and ocean currents.
5. Radiation




5.1 Definition of Radiation
Radiation is the transfer of heat through electromagnetic waves.
Unlike conduction and convection, radiation does not require a medium.
Heat can travel through empty space via radiation.
5.2 Mechanism of Radiation
All objects emit electromagnetic radiation due to the thermal motion of their particles.
The amount of radiation depends on:
- Temperature of the object
- Surface properties
- Area of the object
Hotter objects emit more radiation.
5.3 Thermal Radiation
Thermal radiation includes infrared radiation emitted by objects.
Examples include:
- Heat from the Sun reaching Earth
- Heat felt from a fire
- Warmth from a heater
Even human bodies emit infrared radiation.
5.4 Stefan–Boltzmann Law
Radiated energy depends on temperature.
The Stefan–Boltzmann law states:
E = σT⁴
Where:
E = energy emitted
σ = Stefan–Boltzmann constant
T = absolute temperature
This law shows that radiation increases rapidly with temperature.
6. Comparison of Conduction, Convection, and Radiation
These three heat transfer mechanisms differ in several ways.
Conduction occurs mainly in solids through particle collisions.
Convection occurs in liquids and gases through fluid motion.
Radiation occurs through electromagnetic waves and can occur even in vacuum.
Conduction and convection require a medium, while radiation does not.
Each method plays a role in different physical situations.
7. Heat Transfer in Everyday Life
Heat transfer affects many daily activities.
Examples include:
- Cooking food on a stove
- Heating rooms with radiators
- Sunlight warming the Earth
- Cooling systems in vehicles
- Refrigeration systems
Engineers use knowledge of heat transfer to design efficient appliances and energy systems.
8. Heat Transfer in Nature
Heat transfer processes drive many natural phenomena.
Examples include:
- Ocean currents distributing heat around the planet
- Atmospheric circulation causing winds
- Formation of clouds and storms
- Solar radiation heating Earth’s surface
Without heat transfer, life on Earth would not be possible.
9. Applications of Heat Transfer
Understanding heat transfer is essential in many technological fields.
Applications include:
- Power plants generating electricity
- Cooling systems for computers
- Thermal insulation in buildings
- Aerospace engineering
- Solar energy systems
Efficient heat transfer management improves energy efficiency and safety.
Conclusion
Heat transfer is a fundamental concept in physics that describes how thermal energy moves from one place to another due to temperature differences. The three primary mechanisms of heat transfer are conduction, convection, and radiation.
Conduction transfers heat through direct particle interactions in solids. Convection transfers heat through the movement of fluids such as liquids and gases. Radiation transfers heat through electromagnetic waves and can occur even in a vacuum.
These processes are essential for understanding natural phenomena, industrial processes, and everyday activities. From climate systems to cooking, from power generation to space technology, heat transfer plays a critical role in shaping the physical world.
By studying heat transfer, scientists and engineers develop technologies that improve energy efficiency, environmental sustainability, and technological innovation.
Tags:
