



1. Introduction
Reaction rate and rate laws are fundamental concepts in chemical kinetics, the branch of chemistry that studies the speed of chemical reactions and the factors affecting them. Understanding how quickly reactions occur and how reactant concentrations influence reaction speed is essential in chemistry, industry, medicine, environmental science, and biological systems.
In everyday life, chemical reactions occur constantly. Some reactions take place almost instantly, such as explosions or acid–base neutralization, while others occur slowly, such as rusting of iron or decomposition of organic materials. Chemical kinetics helps scientists understand why these differences exist.
Two major concepts form the foundation of kinetic studies:
- Reaction Rate – how fast a reaction occurs
- Rate Laws – mathematical relationships between reaction rate and reactant concentrations
Reaction rates help determine the speed of product formation or reactant consumption, while rate laws describe how reaction rates depend on concentration and reaction conditions.
These concepts allow chemists to design efficient chemical processes, control industrial reactions, optimize pharmaceutical production, and understand biochemical pathways.
2. Reaction Rate
Definition
The reaction rate is defined as the change in concentration of reactants or products per unit time during a chemical reaction.
In simple terms, it tells us how fast a chemical reaction occurs.
For example, consider the reaction:
A → B
As the reaction proceeds:
- Concentration of A decreases
- Concentration of B increases
The rate can therefore be expressed either as the disappearance of reactant or appearance of product.
Mathematical Expression of Reaction Rate
For a reaction:
A → B
Rate can be written as:
[
Rate = -\frac{d[A]}{dt} = \frac{d[B]}{dt}
]
The negative sign is used because reactant concentration decreases with time.
Units of Reaction Rate
Reaction rate depends on concentration change per time, so common units are:
- mol L⁻¹ s⁻¹
- M s⁻¹
- mol dm⁻³ s⁻¹
Where:
- mol = amount of substance
- L = litre
- s = seconds
Example:
If concentration of a reactant decreases from 1.0 mol/L to 0.8 mol/L in 10 seconds:
Rate = (1.0 − 0.8) / 10
Rate = 0.02 mol L⁻¹ s⁻¹
3. Average Rate and Instantaneous Rate
Average Rate
Average rate measures how much concentration changes over a finite time interval.
[
Average\ Rate = \frac{\Delta [Reactant]}{\Delta t}
]
Example:
If concentration changes from 0.8 mol/L to 0.5 mol/L in 5 seconds:
Average Rate = (0.8 − 0.5) / 5
Average Rate = 0.06 mol L⁻¹ s⁻¹
Average rates are useful for general measurements.
Instantaneous Rate
The instantaneous rate measures the reaction rate at a specific moment in time.
It is determined using calculus:
[
Instantaneous\ Rate = \frac{d[A]}{dt}
]
Graphically, it corresponds to the slope of the tangent line on a concentration vs time curve.
4. Rate of Reaction for General Chemical Equations
Consider a reaction:
aA + bB → cC + dD
The rate can be expressed using stoichiometric coefficients.
[
Rate = -\frac{1}{a}\frac{d[A]}{dt}
]
[
Rate = -\frac{1}{b}\frac{d[B]}{dt}
]
[
Rate = \frac{1}{c}\frac{d[C]}{dt}
]
[
Rate = \frac{1}{d}\frac{d[D]}{dt}
]
This ensures the rate is consistent regardless of which species is measured.
5. Methods for Measuring Reaction Rate

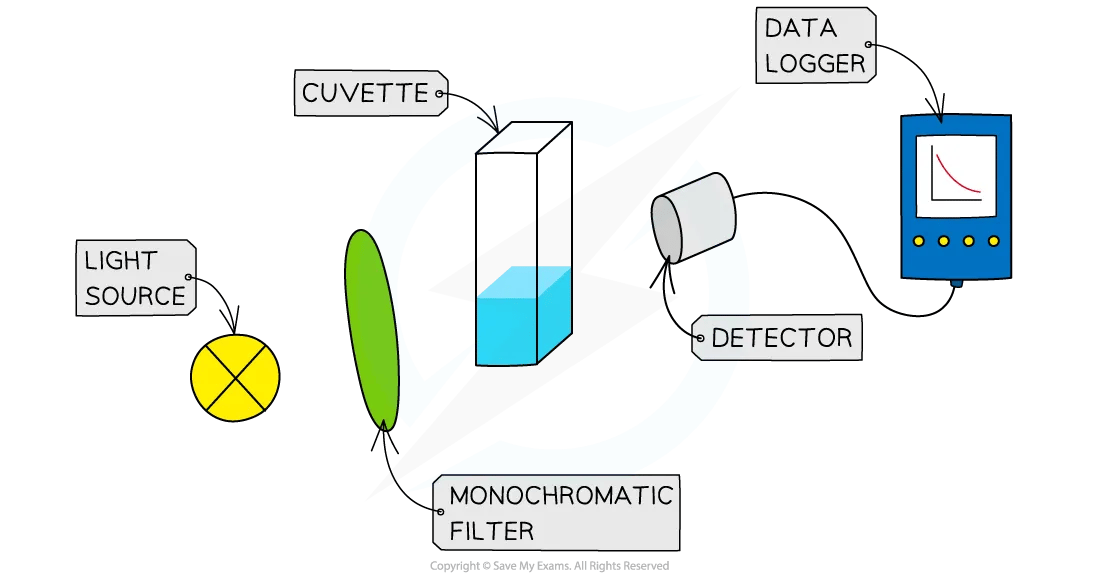

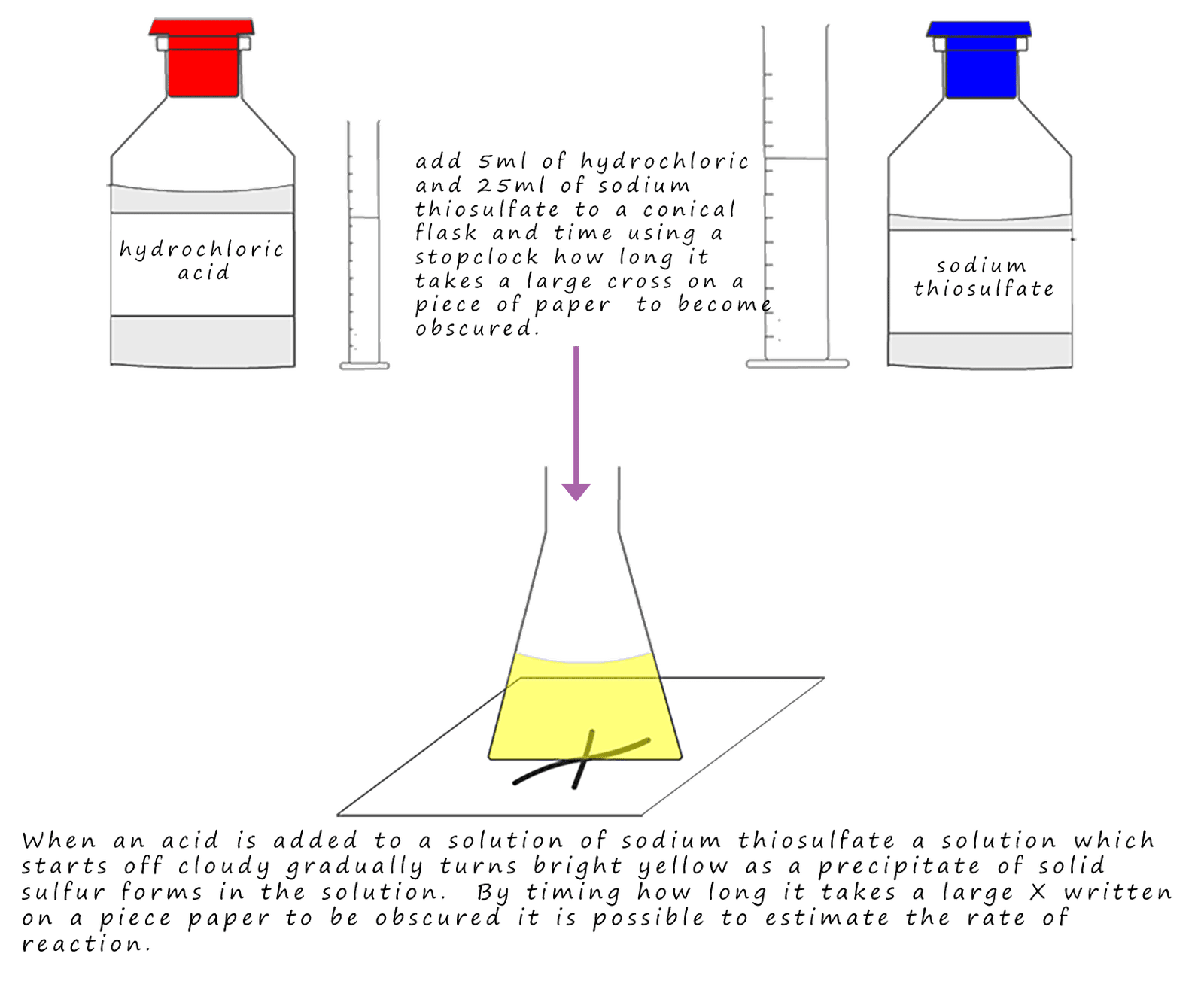
Reaction rates are determined experimentally using various methods.
1. Gas Volume Measurement
If a reaction produces gas, the rate can be measured by monitoring gas volume.
Example:
Zn + HCl → H₂ gas
Hydrogen gas volume increases with time.
2. Pressure Measurement
For gas reactions, pressure changes indicate reaction progress.
Example:
Decomposition reactions.
3. Color Change
Many reactions involve color changes.
Colorimeters measure concentration changes.
Example:
Iodine clock reaction.
4. Precipitation Measurement
Some reactions produce precipitates.
Rate can be measured by observing turbidity.
5. Spectrophotometry
Light absorption measurements track concentration changes.
This technique is widely used in biochemistry.
6. Factors Affecting Reaction Rate
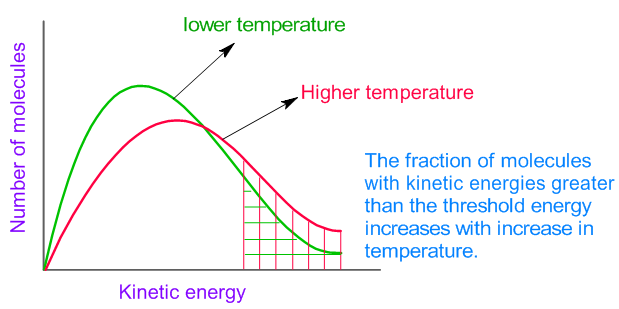


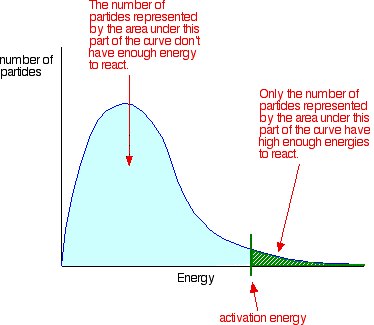
Several factors influence reaction rates.
1. Concentration
Higher concentration increases collision frequency between molecules.
Therefore, reaction rate increases.
Example:
More acid leads to faster reaction with metal.
2. Temperature
Higher temperature increases molecular kinetic energy.
This increases both:
- Collision frequency
- Collision energy
As a result, reactions occur faster.
3. Surface Area
In heterogeneous reactions, larger surface area increases reaction rate.
Example:
Powdered solids react faster than solid blocks.
4. Catalysts
Catalysts increase reaction rate by lowering activation energy.
They provide an alternative reaction pathway.
Example:
Enzymes in biological systems.
5. Pressure
For gaseous reactions, increasing pressure increases reactant concentration.
This increases reaction rate.
6. Nature of Reactants
Ionic reactions in solution occur faster than covalent reactions.
Bond strength also affects reaction rate.
7. Rate Laws
The rate law describes how the rate of a reaction depends on the concentration of reactants.
For a general reaction:
aA + bB → Products
The rate law is:
[
Rate = k[A]^m[B]^n
]
Where:
- k = rate constant
- [A], [B] = concentrations
- m, n = reaction orders
Important Points
- Rate law is determined experimentally
- Reaction order may not equal stoichiometric coefficients
- Rate constant depends on temperature
8. Reaction Order
Reaction order indicates the power of concentration terms in the rate law.
Total order:
[
Order = m + n
]
Types of Reaction Orders
Zero Order
Rate independent of concentration.
Rate = k
First Order
Rate proportional to concentration.
Rate = k[A]
Second Order
Rate proportional to square of concentration.
Rate = k[A]²
Fractional Order
Order may be fractional due to complex mechanisms.
Example:
Rate = k[A]½
9. Integrated Rate Laws
Integrated rate laws describe concentration changes with time.
Zero Order Reaction
[A] = [A]_0 – kt
Characteristics:
- Straight-line graph of concentration vs time
- Rate constant independent of concentration
Half-life:
[
t_{1/2} = \frac{[A]_0}{2k}
]
Examples:
Photochemical reactions.
First Order Reaction
\ln[A] = \ln[A]_0 – kt
Half-life:
[
t_{1/2} = \frac{0.693}{k}
]
Characteristics:
- Exponential decay
- Half-life independent of initial concentration
Examples:
Radioactive decay.
Second Order Reaction
\frac{1}{[A]} = \frac{1}{[A]_0} + kt
Half-life:
[
t_{1/2} = \frac{1}{k[A]_0}
]
Characteristics:
- Linear graph of (1/[A]) vs time.
10. Rate Constant
The rate constant (k) is a proportionality constant in the rate law.
Rate = k[A]ⁿ
Characteristics:
- Depends on temperature
- Independent of concentration
- Indicates reaction speed
Large k → faster reaction.
11. Determination of Rate Law
Rate laws are determined experimentally.
Common methods include:
Method of Initial Rates
Initial reaction rates are measured at different reactant concentrations.
Example table:
| Experiment | [A] | Rate |
|---|---|---|
| 1 | 0.1 | 0.01 |
| 2 | 0.2 | 0.02 |
Doubling concentration doubles rate → first order.
Integrated Rate Law Method
Reaction order determined by plotting:
- [A] vs time
- ln[A] vs time
- 1/[A] vs time
The linear plot indicates reaction order.
12. Molecularity of Reactions
Molecularity refers to the number of molecules involved in an elementary step.
Types:
- Unimolecular
- Bimolecular
- Termolecular
Example:
NO₂ + CO → NO + CO₂
Two molecules → bimolecular reaction.
13. Reaction Mechanisms
A reaction mechanism describes the stepwise process of a chemical reaction.
Example:
Step 1: A + B → Intermediate
Step 2: Intermediate → Product
The slowest step is called the rate-determining step.
Rate law depends on this step.
14. Steady State Approximation
This method assumes the concentration of intermediate species remains constant.
Used to simplify complex rate equations.
15. Catalysis and Rate Laws
Catalysts alter reaction rate but do not appear in the overall reaction.
Catalysts change:
- Reaction mechanism
- Activation energy
- Rate law expression
Examples:
- Enzyme catalysis
- Metal catalysts
16. Applications of Reaction Rate and Rate Laws
Reaction rates and rate laws are essential in many fields.
Industrial Chemistry
Used in:
- Fertilizer production
- Polymer manufacturing
- Petroleum refining
Environmental Chemistry
Helps understand:
- Atmospheric reactions
- Ozone depletion
- Pollution degradation
Biochemistry
Enzyme kinetics describes metabolic reactions.
Medicine
Drug stability and metabolism depend on reaction kinetics.
Food Chemistry
Reaction rates influence:
- Spoilage
- Preservation
- Cooking processes
17. Importance of Studying Reaction Rate
Studying reaction rates helps scientists:
- Control industrial reactions
- Design catalysts
- Understand reaction mechanisms
- Predict chemical behavior
- Improve energy efficiency
Conclusion
Reaction rate and rate laws form the backbone of chemical kinetics. Reaction rate describes how quickly chemical transformations occur, while rate laws mathematically explain how reaction speed depends on reactant concentrations. Through experimental analysis and theoretical models, chemists gain deep insight into molecular processes governing reactions. These principles are essential in scientific research, industrial chemistry, environmental protection, and biological systems, making them fundamental topics in modern chemistry.
