



1. Introduction to Temperature and Heat
Temperature and heat are two closely related concepts in physics that describe the thermal state of matter and the transfer of thermal energy between objects. These concepts are fundamental to the study of thermodynamics, which examines the relationships between heat, energy, work, and temperature.
Although the terms temperature and heat are often used interchangeably in everyday language, they represent different physical quantities. Temperature is a measure of the average kinetic energy of the particles in a substance, while heat refers to the energy that is transferred from one object to another because of a temperature difference.
Understanding temperature and heat is essential for explaining many natural phenomena and technological processes. For example, weather patterns, engine operation, refrigeration systems, cooking, and even biological processes all involve heat transfer and temperature changes.
In physics, heat energy flows from a region of higher temperature to a region of lower temperature until thermal equilibrium is reached. This flow of energy can occur through various mechanisms such as conduction, convection, and radiation.
The study of temperature and heat has played an important role in the development of science and engineering. It has led to the creation of thermometers, heat engines, refrigeration systems, and many other technologies that are vital to modern life.
2. Concept of Temperature
Temperature is a physical quantity that indicates how hot or cold an object is relative to other objects. It provides a measure of the thermal condition of matter and determines the direction of heat flow between objects.
Definition
Temperature can be defined as:
Temperature is the measure of the average kinetic energy of the particles in a substance.
When the particles in a substance move faster, their kinetic energy increases, and the temperature rises. When the particles move slower, the temperature decreases.
Temperature determines the direction of heat flow. Heat always flows from an object with higher temperature to one with lower temperature until both objects reach the same temperature.
Temperature Scales
Several temperature scales are used in science and everyday life.
Celsius Scale
The Celsius scale is widely used around the world. In this scale:
- Water freezes at 0°C
- Water boils at 100°C
This scale is based on the properties of water under standard atmospheric pressure.
Fahrenheit Scale
The Fahrenheit scale is mainly used in the United States.
- Water freezes at 32°F
- Water boils at 212°F
Kelvin Scale
The Kelvin scale is the SI unit of temperature used in scientific measurements.
- Absolute zero: 0 K
- Water freezes: 273 K
- Water boils: 373 K
Kelvin scale is important in thermodynamics because it begins at absolute zero, the point where molecular motion theoretically stops.
3. Concept of Heat
Heat is a form of energy that flows from one body to another due to a temperature difference.
Definition
Heat can be defined as:
Heat is the energy transferred between objects because of a difference in temperature.
Heat is not something contained within an object. Instead, it refers to the energy in transit from one object to another.
Unit of Heat
The SI unit of heat energy is the joule (J).
Historically, heat was also measured in calories.
1 calorie = 4.186 joules
A calorie is defined as the amount of heat required to raise the temperature of 1 gram of water by 1°C.
4. Difference Between Heat and Temperature
Although heat and temperature are related, they represent different physical concepts.
Temperature measures the thermal state of a system, while heat represents energy transfer.
Temperature depends on the average kinetic energy of particles, while heat depends on both temperature and the amount of substance.
For example, a large container of warm water may contain more heat energy than a small cup of boiling water, even though the cup has higher temperature.
Thus, temperature indicates intensity of thermal energy, while heat indicates the total energy transferred.
5. Heat Transfer




Heat can be transferred from one place to another through three main processes.
Conduction
Conduction is the transfer of heat through direct contact between particles.
In conduction:
- Heat flows from hot regions to cold regions.
- Energy transfer occurs through collisions between molecules.
Example:
A metal spoon placed in hot tea becomes hot because heat travels through the metal by conduction.
Metals are good conductors because their electrons transfer energy efficiently.
Convection
Convection occurs in fluids such as liquids and gases.
In convection:
- Heat transfer occurs through the movement of fluid.
- Warmer fluid rises while cooler fluid sinks.
Example:
When water is heated in a pot, hot water rises and cooler water sinks, forming convection currents.
Convection plays an important role in atmospheric circulation and ocean currents.
Radiation
Radiation is the transfer of heat through electromagnetic waves.
Radiation does not require a medium.
Example:
Heat from the Sun reaches Earth through radiation.
All objects emit thermal radiation depending on their temperature.
6. Thermal Expansion

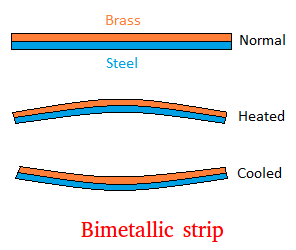

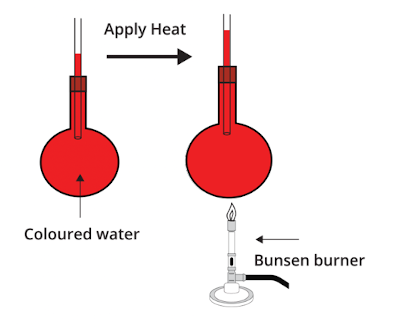
When materials are heated, they usually expand. This phenomenon is called thermal expansion.
As temperature increases, particles move more vigorously and occupy more space.
Types of Thermal Expansion
Linear Expansion
Expansion in one dimension such as length.
Example: expansion of railway tracks in hot weather.
Area Expansion
Expansion in two dimensions.
Example: expansion of metal plates.
Volume Expansion
Expansion in three dimensions.
Example: expansion of liquids and gases when heated.
Thermal expansion is important in engineering design to prevent structural damage.
7. Specific Heat Capacity
Specific heat capacity describes how much heat energy is required to raise the temperature of a substance.
Definition
Specific heat capacity is the amount of heat required to raise the temperature of one unit mass of a substance by one degree Celsius.
Formula:
Q = mcΔT
Where:
Q = heat energy
m = mass
c = specific heat capacity
ΔT = change in temperature
Different substances have different specific heat capacities.
Water has a high specific heat capacity, meaning it requires a large amount of heat to change its temperature.
This property plays an important role in regulating Earth’s climate.
8. Latent Heat
When a substance changes its state, heat energy is absorbed or released without changing temperature.
This heat is called latent heat.
Latent Heat of Fusion
Heat required to change solid into liquid.
Example:
Melting of ice.
Latent Heat of Vaporization
Heat required to change liquid into gas.
Example:
Boiling water turning into steam.
Latent heat explains why temperature remains constant during phase changes.
9. Thermodynamics and Heat
Thermodynamics is the branch of physics that studies heat and energy transformations.
It is governed by several important laws.
First Law of Thermodynamics
Energy cannot be created or destroyed.
It can only change from one form to another.
Example:
Heat energy converted into mechanical work in engines.
Second Law of Thermodynamics
Heat naturally flows from hotter objects to colder objects.
This law introduces the concept of entropy.
Third Law of Thermodynamics
As temperature approaches absolute zero, the entropy of a perfect crystal approaches zero.
These laws govern the behavior of thermal systems.
10. Applications of Heat and Temperature
Temperature and heat have numerous practical applications.
Cooking
Cooking uses heat transfer to change the chemical structure of food.
Refrigeration
Refrigerators remove heat from inside compartments to keep food cold.
Power Plants
Steam engines and turbines use heat to generate electricity.
Climate and Weather
Temperature differences drive atmospheric circulation and weather patterns.
Medical Applications
Thermometers measure body temperature to diagnose illness.
11. Heat in Nature
Heat plays an important role in many natural processes.
Examples include:
- Solar heating of Earth’s atmosphere
- Formation of ocean currents
- Melting of glaciers
- Volcanic heat from Earth’s interior
The Sun is the primary source of heat energy for Earth.
Solar radiation drives climate systems and supports life on the planet.
12. Measurement of Temperature
Temperature is measured using devices called thermometers.
Different types include:
- Mercury thermometers
- Alcohol thermometers
- Digital thermometers
- Infrared thermometers
Thermometers work based on properties that change with temperature, such as expansion of liquids or electrical resistance.
13. Importance of Temperature and Heat
Temperature and heat are fundamental to understanding energy transfer in physical systems.
They help explain:
- Phase changes of matter
- Climate and weather patterns
- Operation of engines
- Biological metabolism
- Industrial processes
Without understanding heat and temperature, many modern technologies would not exist.
Conclusion
Temperature and heat are fundamental concepts in physics that describe the thermal state of matter and the transfer of energy due to temperature differences. Temperature measures the average kinetic energy of particles, while heat refers to energy transferred between objects.
Heat transfer occurs through conduction, convection, and radiation, allowing energy to move between systems. Thermal expansion, specific heat capacity, and latent heat explain how materials respond to temperature changes.
The study of temperature and heat forms the foundation of thermodynamics, which governs energy transformations in nature and technology. These concepts are essential for understanding processes such as climate dynamics, industrial heating, refrigeration, power generation, and biological functions.
By studying heat and temperature, scientists and engineers have developed technologies that improve daily life and deepen our understanding of natural phenomena.
Tags:
