
1. Introduction to Thermal Expansion
Thermal expansion is an important physical phenomenon that occurs when materials change their size, shape, or volume due to changes in temperature. When a substance is heated, its particles gain energy and move more vigorously. As a result, the distance between the particles increases, causing the material to expand. Conversely, when a substance is cooled, the particles lose energy, move less vigorously, and come closer together, causing the material to contract.
Thermal expansion occurs in all states of matter—solids, liquids, and gases—but the degree of expansion varies depending on the material and its physical properties. In solids, expansion is generally small because the particles are tightly packed. In liquids, expansion is more noticeable, while gases show the largest expansion because their particles are far apart and move freely.
Understanding thermal expansion is essential in many areas of science and engineering. It explains why bridges have expansion joints, why railway tracks have small gaps between rails, why thermometers work, and why certain materials are used in temperature-sensitive devices.
Thermal expansion is also closely related to thermodynamics, which studies the relationship between heat, temperature, and energy. When heat is supplied to a material, part of the energy increases the kinetic energy of particles, leading to expansion.
Because of its widespread impact in engineering structures, industrial processes, and natural phenomena, thermal expansion is an important topic in physics.
2. Basic Concept of Thermal Expansion
Thermal expansion occurs because atoms and molecules in a material vibrate more strongly when heated. These vibrations cause the average distance between particles to increase.
In most materials, heating leads to expansion and cooling leads to contraction.
This phenomenon occurs because of the following process:
- When temperature increases, particles gain kinetic energy.
- Increased kinetic energy causes particles to vibrate faster.
- Vibrations push particles slightly farther apart.
- The overall size of the material increases.
Although individual particles move only slightly, the combined effect across millions of particles produces noticeable expansion in the material.
Thermal expansion is usually proportional to the change in temperature. Larger temperature changes typically produce larger expansion.
3. Types of Thermal Expansion
Thermal expansion can occur in different dimensions depending on the nature of the material and the situation. There are three main types of thermal expansion.
Linear Expansion
Linear expansion refers to the change in length of a material due to temperature change.
It occurs in objects such as rods, wires, and beams.
Formula:
ΔL = αL₀ΔT
Where:
ΔL = change in length
L₀ = original length
α = coefficient of linear expansion
ΔT = temperature change
This equation shows that expansion depends on the material, initial length, and temperature change.
Area Expansion
Area expansion occurs when a two-dimensional surface expands due to temperature increase.
Examples include metal plates and sheets.
Formula:
ΔA = βA₀ΔT
Where:
ΔA = change in area
A₀ = original area
β = coefficient of area expansion
ΔT = temperature change
Area expansion is approximately twice the linear expansion for isotropic materials.
Volume Expansion
Volume expansion occurs when the entire volume of a material increases due to heating.
This type of expansion occurs in solids, liquids, and gases.
Formula:
ΔV = γV₀ΔT
Where:
ΔV = change in volume
V₀ = original volume
γ = coefficient of volume expansion
ΔT = temperature change
Volume expansion is approximately three times the linear expansion for many materials.
4. Coefficient of Thermal Expansion


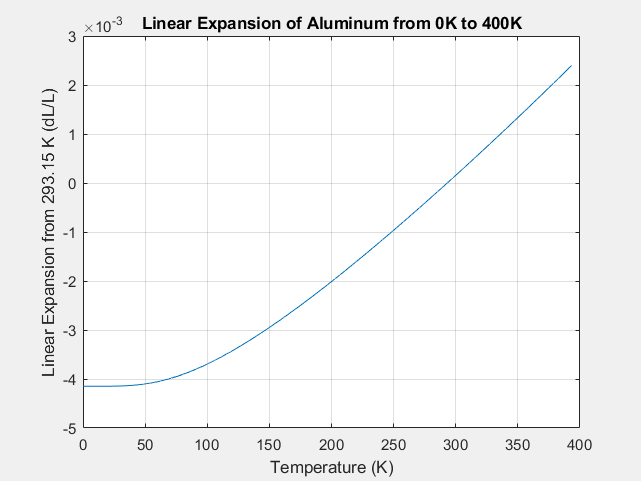
The coefficient of thermal expansion describes how much a material expands when temperature changes.
Different materials expand at different rates.
There are three coefficients:
- Coefficient of linear expansion
- Coefficient of area expansion
- Coefficient of volume expansion
Coefficient of Linear Expansion
Defined as the fractional increase in length per degree temperature increase.
Unit:
per degree Celsius (°C⁻¹)
Metals such as aluminum and copper have relatively large expansion coefficients, while materials such as glass and ceramics expand less.
5. Thermal Expansion in Solids
Solids expand when heated because their particles vibrate with greater amplitude.
However, the expansion in solids is relatively small because particles are tightly bound.
Examples include:
- Metal rods expanding when heated
- Bridge structures expanding in hot weather
- Electrical wires sagging during summer
To prevent structural damage, engineers design structures with expansion allowances.
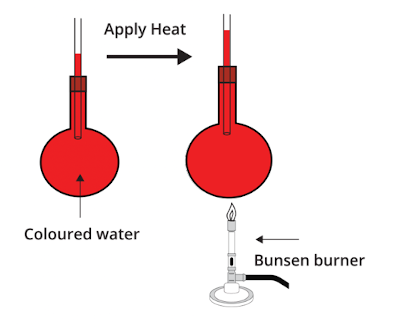
6. Thermal Expansion in Liquids

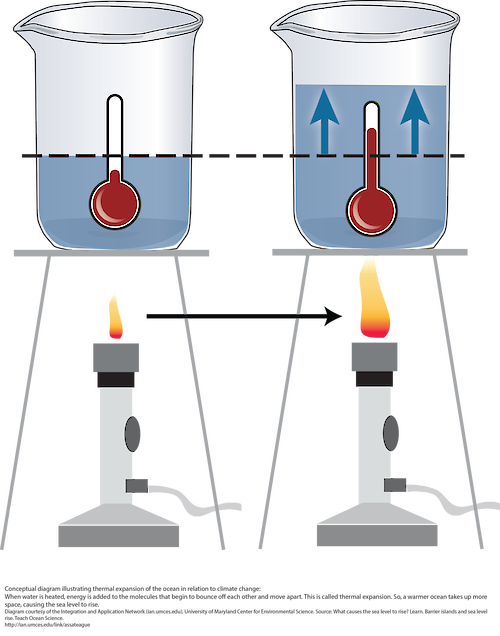

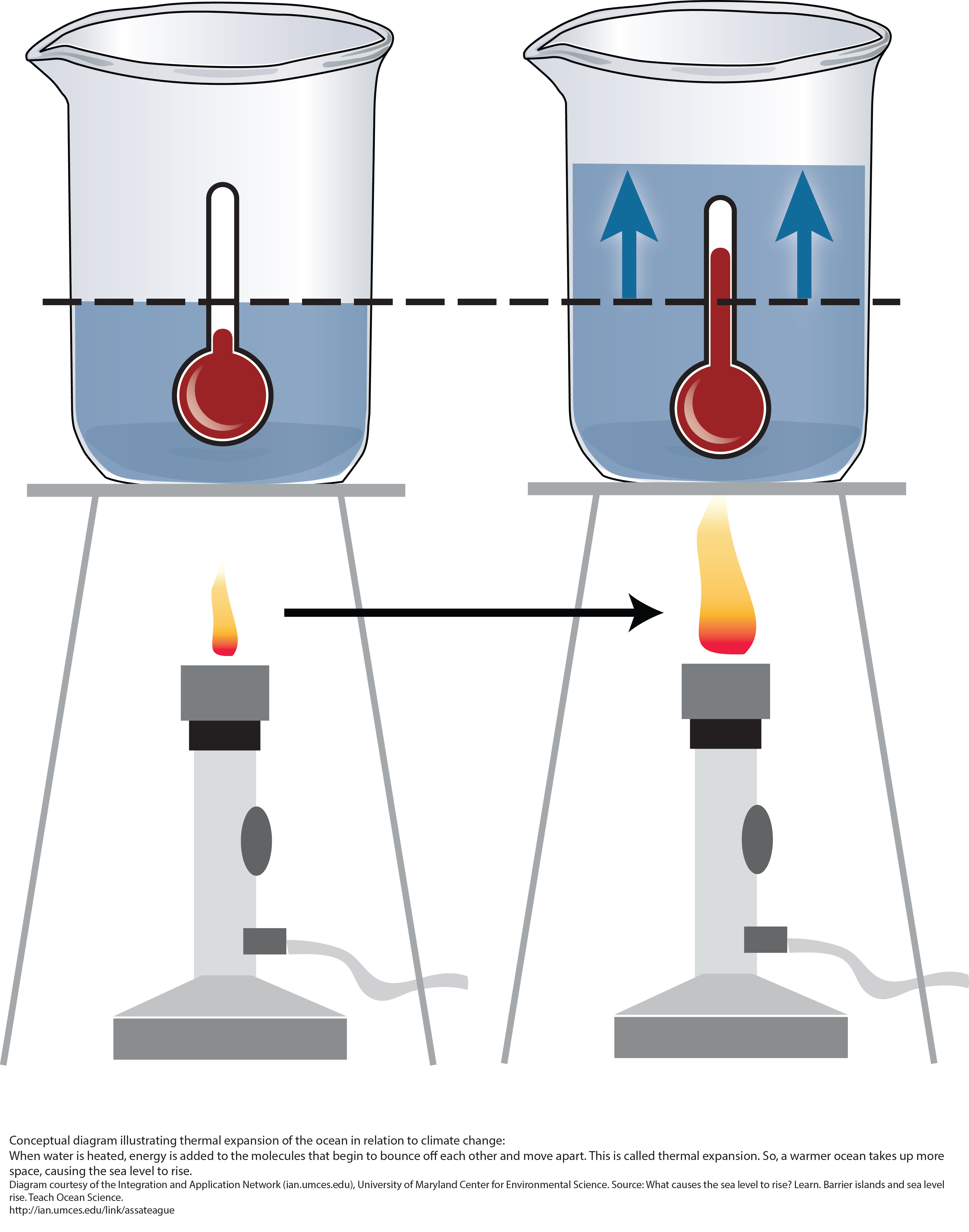
Liquids expand more than solids when heated.
This happens because molecules in liquids are less tightly bound.
Examples include:
- Mercury expanding in thermometers
- Gasoline expanding in fuel tanks
- Water expanding when heated
Because of thermal expansion, fuel tanks are not completely filled to allow room for expansion.
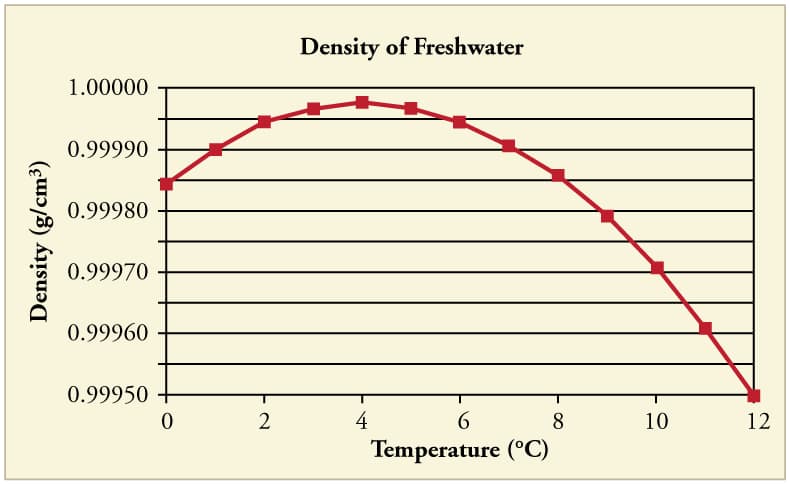
7. Anomalous Expansion of Water
Water exhibits unusual expansion behavior between 0°C and 4°C.
Normally substances contract when cooled.
However, water expands when cooled below 4°C.
At 4°C water has maximum density.
Below this temperature, water molecules form a more open structure, causing expansion.
This phenomenon is called anomalous expansion of water.
It is very important for aquatic life.
Because ice floats on water, lakes freeze from the top while water below remains liquid, allowing fish and other organisms to survive.
8. Thermal Expansion in Gases
Gases expand significantly when heated.
Because gas molecules are far apart and move freely, temperature changes strongly affect their volume.
According to Charles’s Law:
Volume is directly proportional to temperature at constant pressure.
V ∝ T
This means that heating a gas causes it to expand, while cooling causes it to contract.
Gas expansion is important in many applications including engines, balloons, and atmospheric science.
9. Applications of Thermal Expansion
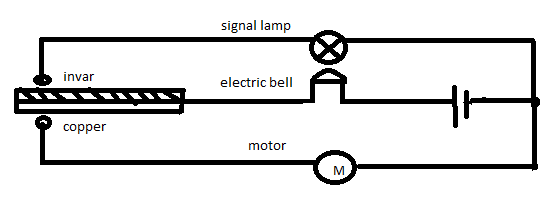


Thermal expansion has many practical applications in engineering and technology.
Thermometers
Thermometers operate based on expansion of liquids such as mercury or alcohol when temperature changes.
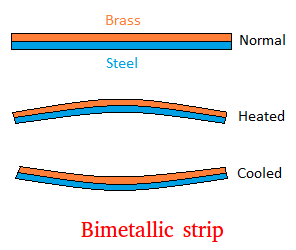
Bimetallic Strips
Bimetallic strips consist of two metals with different expansion rates.
When heated, the strip bends and can activate switches in thermostats.
Bridge Expansion Joints
Bridges include expansion joints that allow structures to expand and contract with temperature changes.
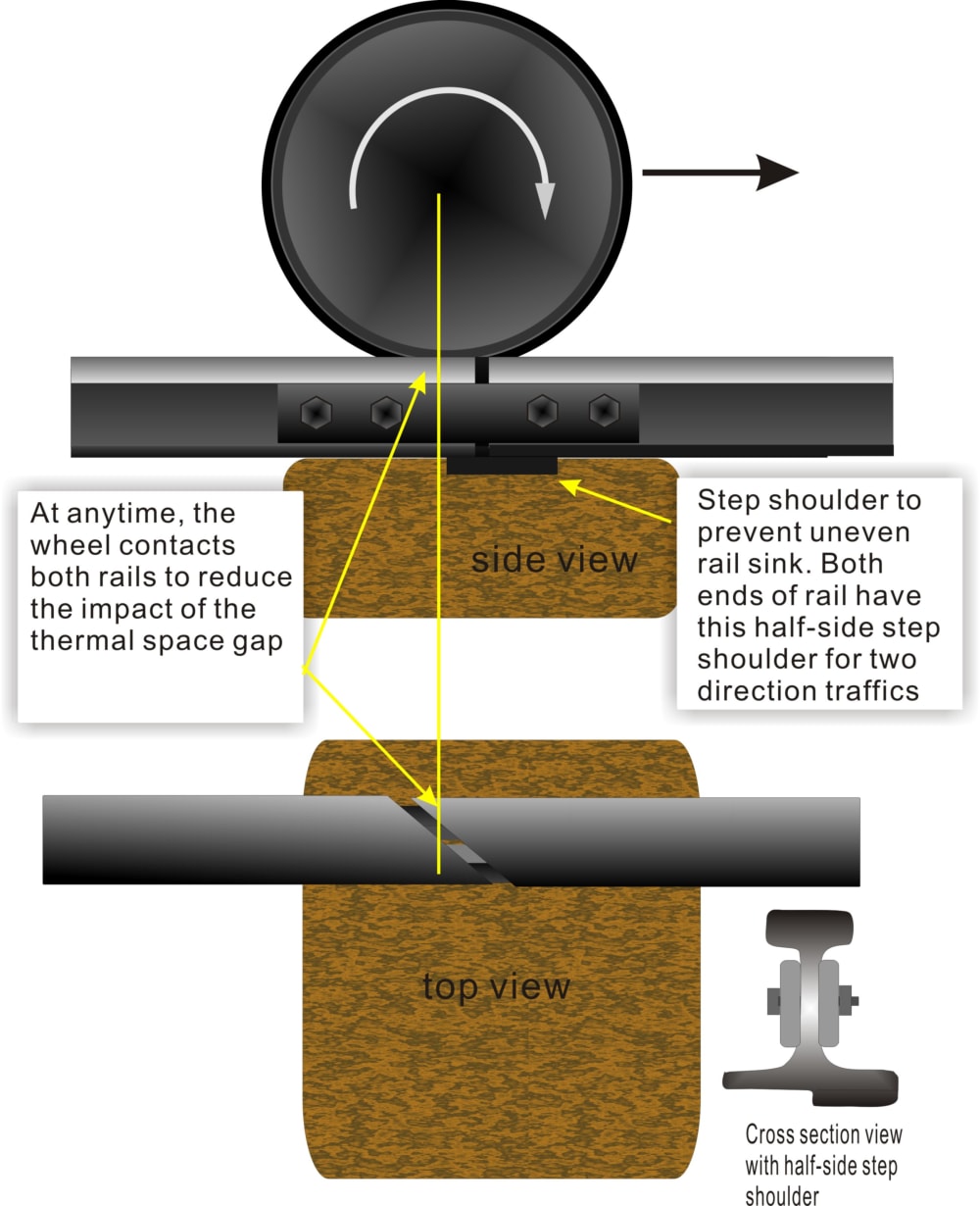
Railway Tracks
Small gaps are left between railway tracks to allow expansion during hot weather.
Overhead Power Lines
Electrical wires are installed with slack to allow expansion and contraction.
10. Thermal Stress
When expansion is restricted, thermal stress develops.
If a material is prevented from expanding or contracting freely, internal forces arise.
These forces can cause deformation or structural damage.
Examples include:
- Cracks in buildings due to temperature changes
- Buckling of railway tracks in hot weather
- Damage to pipelines exposed to heat
Engineers must consider thermal stress when designing structures.
11. Thermal Expansion in Nature
Thermal expansion also occurs in natural environments.
Examples include:
- Rocks expanding during hot daytime temperatures
- Cracking of rocks due to repeated heating and cooling
- Expansion of ocean water contributing to sea level rise
- Atmospheric expansion influencing weather patterns
These natural processes shape landscapes and influence climate systems.
12. Importance of Thermal Expansion
Thermal expansion is important for many reasons.
It helps explain how materials behave under temperature changes.
It influences engineering design, industrial processes, and natural phenomena.
Without understanding thermal expansion, structures could fail due to temperature variations.
Thermal expansion also plays a role in scientific instruments, climate studies, and mechanical systems.
Conclusion
Thermal expansion is the increase in size or volume of a material when its temperature rises. This phenomenon occurs because heating increases the kinetic energy of particles, causing them to move farther apart. Thermal expansion occurs in solids, liquids, and gases, although the magnitude varies depending on the material.
There are three main types of expansion: linear expansion, area expansion, and volume expansion. These are described using expansion coefficients that measure how much a material expands for each degree of temperature change.
Thermal expansion has numerous practical applications including thermometers, thermostats, bridge construction, railway track design, and industrial machinery. Engineers must consider thermal expansion to prevent structural damage and ensure safety.
In nature, thermal expansion influences geological processes, atmospheric circulation, and ocean dynamics. It also explains the unique behavior of water between 0°C and 4°C, which plays an essential role in sustaining aquatic ecosystems.
Understanding thermal expansion helps scientists and engineers design reliable systems and better understand how materials behave under changing temperature conditions.
Tags: