


1. Introduction to Polymers
Polymers are large molecules composed of repeating structural units known as monomers. These monomers are chemically bonded together to form long chains or networks, resulting in extremely large molecules called macromolecules. Polymer chemistry is an important branch of chemistry that studies the synthesis, structure, properties, and applications of polymeric materials.
The word polymer originates from the Greek words:
- poly meaning many
- meros meaning parts
Thus, polymers are molecules made of many repeating parts.
Polymers occur naturally in many biological systems and are also manufactured synthetically in industries. Natural polymers include proteins, DNA, cellulose, and starch, while synthetic polymers include plastics, nylon, polyester, and rubber.
Polymers play an essential role in modern life. They are used in:
- Packaging materials
- Clothing fibers
- Medical devices
- Electronic components
- Automotive parts
- Construction materials
Because of their versatility and unique properties, polymers have become one of the most important classes of materials in science and technology.
2. Structure of Polymers
A polymer molecule consists of repeating units called monomers that are linked together by covalent bonds.
Example:
Ethene monomer:
CH₂ = CH₂
Polymerization forms polyethylene:
–CH₂–CH₂–CH₂–CH₂–CH₂–
The repeating unit is called the repeat unit.
Polymers can have extremely large molecular weights, often containing thousands or even millions of atoms.
3. Classification of Polymers
Polymers can be classified in several ways depending on their origin, structure, and polymerization mechanism.
Major classifications include:
- Natural polymers
- Synthetic polymers
- Semi-synthetic polymers
4. Natural Polymers




Natural polymers are polymers that occur naturally in living organisms.
Examples include:
Proteins
Proteins are polymers of amino acids linked by peptide bonds.
Functions include:
- Enzymes
- Structural components
- Hormones
Carbohydrates
Carbohydrates such as starch and cellulose are polymers of glucose.
Starch serves as energy storage in plants.
Cellulose forms plant cell walls.
DNA and RNA
DNA and RNA are polymers of nucleotides.
They store and transmit genetic information.
5. Synthetic Polymers




Synthetic polymers are man-made materials produced through chemical reactions.
Examples include:
Polyethylene
Used in plastic bags and containers.
Polypropylene
Used in packaging and automotive parts.
Nylon
Used in textiles and ropes.
Polyester
Used in clothing fibers.
Synthetic polymers have revolutionized modern manufacturing due to their durability and versatility.
6. Semi-Synthetic Polymers
Semi-synthetic polymers are chemically modified natural polymers.
Example:
Cellulose acetate.
Applications include:
- Photographic films
- Textile fibers
- Coatings
7. Polymerization


Polymerization is the chemical process through which monomers combine to form polymers.
Two main types exist:
- Addition polymerization
- Condensation polymerization
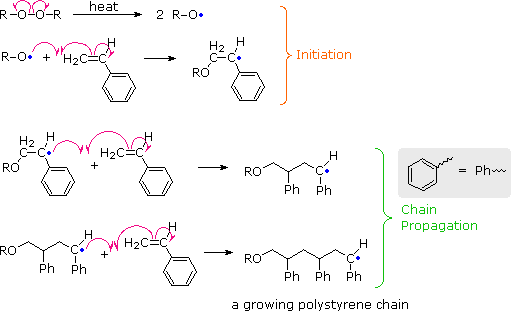
Addition Polymerization
In addition polymerization, monomers join together without the loss of small molecules.
Example:
Ethene → Polyethylene
Steps include:
- Initiation
- Propagation
- Termination
Addition polymerization is common in plastics.
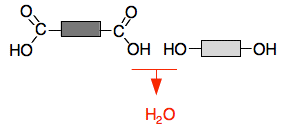
Condensation Polymerization
In condensation polymerization, monomers combine with the elimination of small molecules such as water or ammonia.
Example:
Nylon formation from diamine and dicarboxylic acid.
8. Polymer Structure
Polymers may have different structural arrangements.
Linear Polymers
Monomers are linked in straight chains.
Example:
Polyethylene.
Branched Polymers
Side chains branch from the main chain.
Example:
Low-density polyethylene.
Cross-Linked Polymers
Chains are interconnected forming a network.
Example:
Vulcanized rubber.
9. Types of Polymer Materials



Polymers can also be classified based on their mechanical properties.
Thermoplastics
Thermoplastics soften when heated and harden when cooled.
Examples:
- Polyethylene
- Polystyrene
- PVC
Used in packaging and containers.
Thermosetting Polymers
Thermosetting polymers become permanently hard after heating.
Examples:
- Bakelite
- Epoxy resins
Used in electrical components.
Elastomers
Elastomers are flexible polymers with rubber-like properties.
Example:
Natural rubber.
Used in tires and elastic materials.
10. Properties of Polymers
Polymers exhibit a wide range of properties.
Mechanical Properties
- Strength
- Elasticity
- Toughness
Thermal Properties
- Glass transition temperature
- Melting point
Chemical Resistance
Many polymers resist chemical corrosion.
Electrical Properties
Some polymers are electrical insulators.
11. Applications of Polymers
Polymers are used in many industries.
Packaging
Plastic containers and films.
Textiles
Nylon, polyester, acrylic fibers.
Medicine
Artificial organs, sutures, drug delivery systems.
Electronics
Insulating materials and circuit components.
Automotive Industry
Lightweight materials for vehicles.
12. Biodegradable Polymers
Biodegradable polymers can be broken down by microorganisms.
Examples:
- Polylactic acid
- Polyhydroxyalkanoates
These materials help reduce plastic pollution.
13. Environmental Impact of Polymers
The widespread use of plastics has led to environmental challenges.
Problems include:
- Plastic waste accumulation
- Ocean pollution
- Microplastics
Recycling and biodegradable polymers are being developed to address these issues.
14. Future of Polymer Science
Polymer science continues to evolve with new developments such as:
- Smart polymers
- Conducting polymers
- Biopolymers
- Nanocomposites
These materials have applications in medicine, electronics, and renewable energy.
15. Importance of Polymers
Polymers are among the most important materials in modern society.
They provide:
- Lightweight materials
- Durable products
- Versatile manufacturing options
Polymer science continues to drive innovation in many technological fields.
Conclusion
Polymers are macromolecules composed of repeating monomer units linked together through chemical bonds. They occur naturally in biological systems and can also be synthesized artificially in laboratories and industries. Polymers can be classified as natural, synthetic, or semi-synthetic and may exhibit linear, branched, or cross-linked structures. Polymerization processes such as addition and condensation reactions form these macromolecules. Polymers are used in countless applications including packaging, textiles, electronics, medicine, and construction. Despite their usefulness, environmental concerns related to plastic waste have prompted research into biodegradable and sustainable polymer materials. Polymer chemistry continues to be a rapidly advancing field with enormous scientific and industrial significance.
