



1. Introduction to Acid–Base Titrations
Acid–base titration is one of the most widely used analytical techniques in chemistry for determining the concentration of an unknown acid or base solution. It is a type of volumetric analysis, where the volume of a solution with known concentration is used to determine the concentration of another solution.
In an acid–base titration, an acid reacts with a base in a neutralization reaction to produce salt and water. By carefully measuring the volume of the titrant added until the reaction reaches completion, chemists can calculate the concentration of the unknown solution.
The technique is essential in:
- Analytical chemistry
- Pharmaceutical industries
- Food chemistry
- Environmental monitoring
- Water quality testing
- Chemical manufacturing
Acid–base titrations are widely taught in chemistry laboratories because they demonstrate important principles of acid–base reactions, stoichiometry, and solution chemistry.
2. Principle of Acid–Base Titration
The principle of acid–base titration is based on the neutralization reaction between an acid and a base.
General reaction:
Acid + Base → Salt + Water
Example:
HCl + NaOH → NaCl + H₂O
During titration:
- One solution of known concentration (titrant) is slowly added to another solution of unknown concentration.
- The reaction continues until the equivalence point, where the amount of acid equals the amount of base.
At this point:
moles of acid = moles of base (according to stoichiometry)
Indicators are often used to detect when the reaction reaches completion.
3. Components of a Titration Experiment




A typical acid–base titration experiment requires several laboratory instruments.
1. Burette
A burette is a long graduated glass tube used to deliver precise volumes of liquid.
It contains the titrant (solution of known concentration).
2. Pipette
A pipette is used to measure a fixed volume of the analyte solution (unknown concentration).
3. Conical Flask
The analyte solution is placed in a conical flask where the titration reaction occurs.
4. Indicator
An indicator is added to detect the endpoint of the titration.
5. Stand and Clamp
Used to hold the burette securely during titration.
4. Terminology Used in Titration
Several important terms are commonly used in acid–base titration.
Titrant
The solution of known concentration placed in the burette.
Analyte
The solution of unknown concentration being analyzed.
Equivalence Point
The point at which the number of moles of acid equals the number of moles of base.
Endpoint
The point at which the indicator changes color.
Ideally, endpoint should be very close to the equivalence point.
Standard Solution
A solution whose concentration is accurately known.
5. Types of Acid–Base Titrations
Acid–base titrations are classified based on the strength of the acid and base involved.
Four main types exist:
- Strong acid – strong base
- Weak acid – strong base
- Strong acid – weak base
- Weak acid – weak base
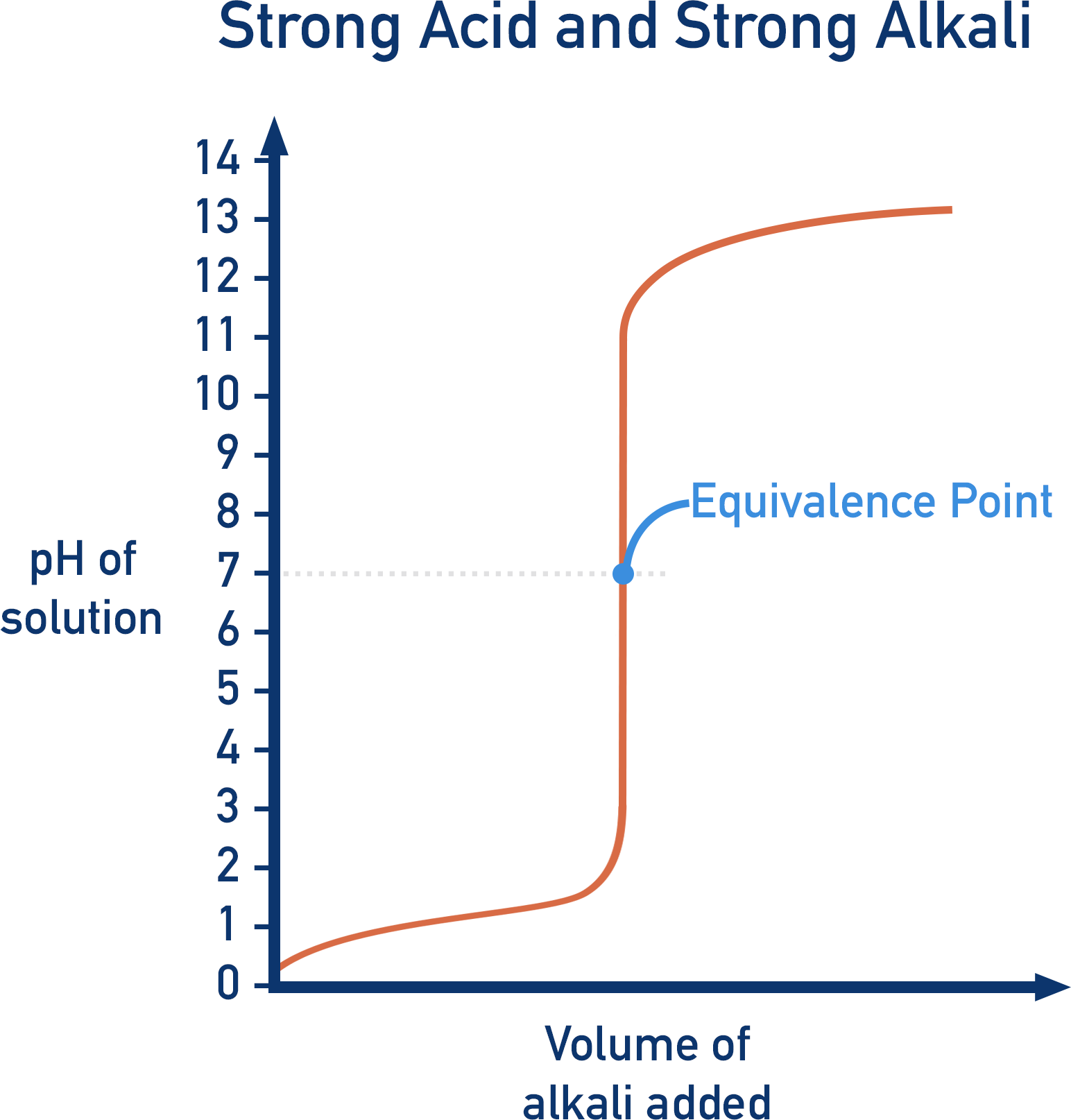
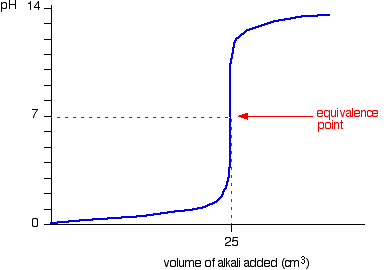
6. Strong Acid–Strong Base Titration


Example:
HCl + NaOH → NaCl + H₂O
Characteristics:
- Sharp pH change near equivalence point
- Equivalence point occurs at pH 7
- Suitable indicators include phenolphthalein and methyl orange.
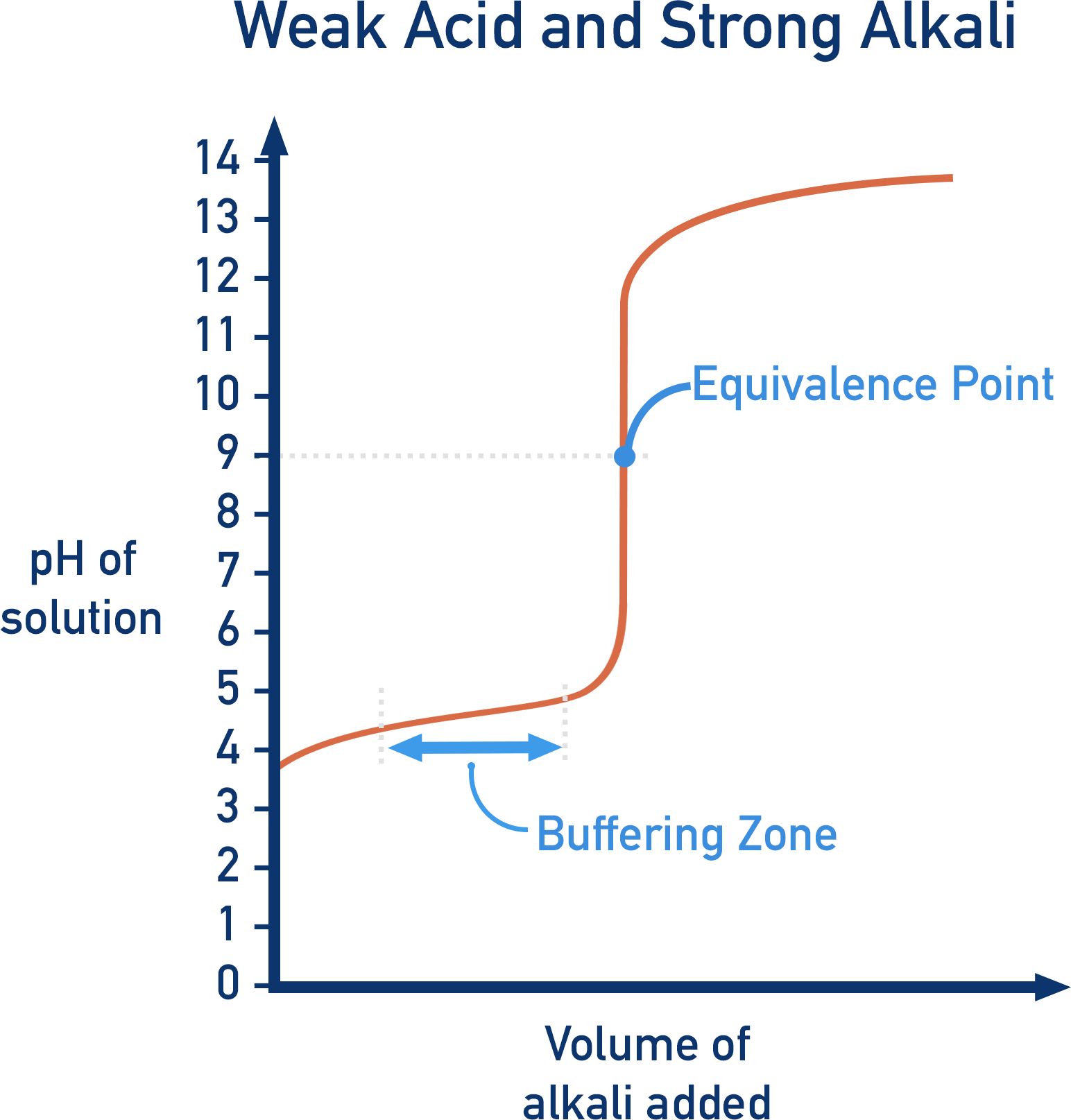
7. Weak Acid–Strong Base Titration


Example:
CH₃COOH + NaOH → CH₃COONa + H₂O
Characteristics:
- Initial pH is higher than strong acid
- Buffer region present
- Equivalence point occurs above pH 7
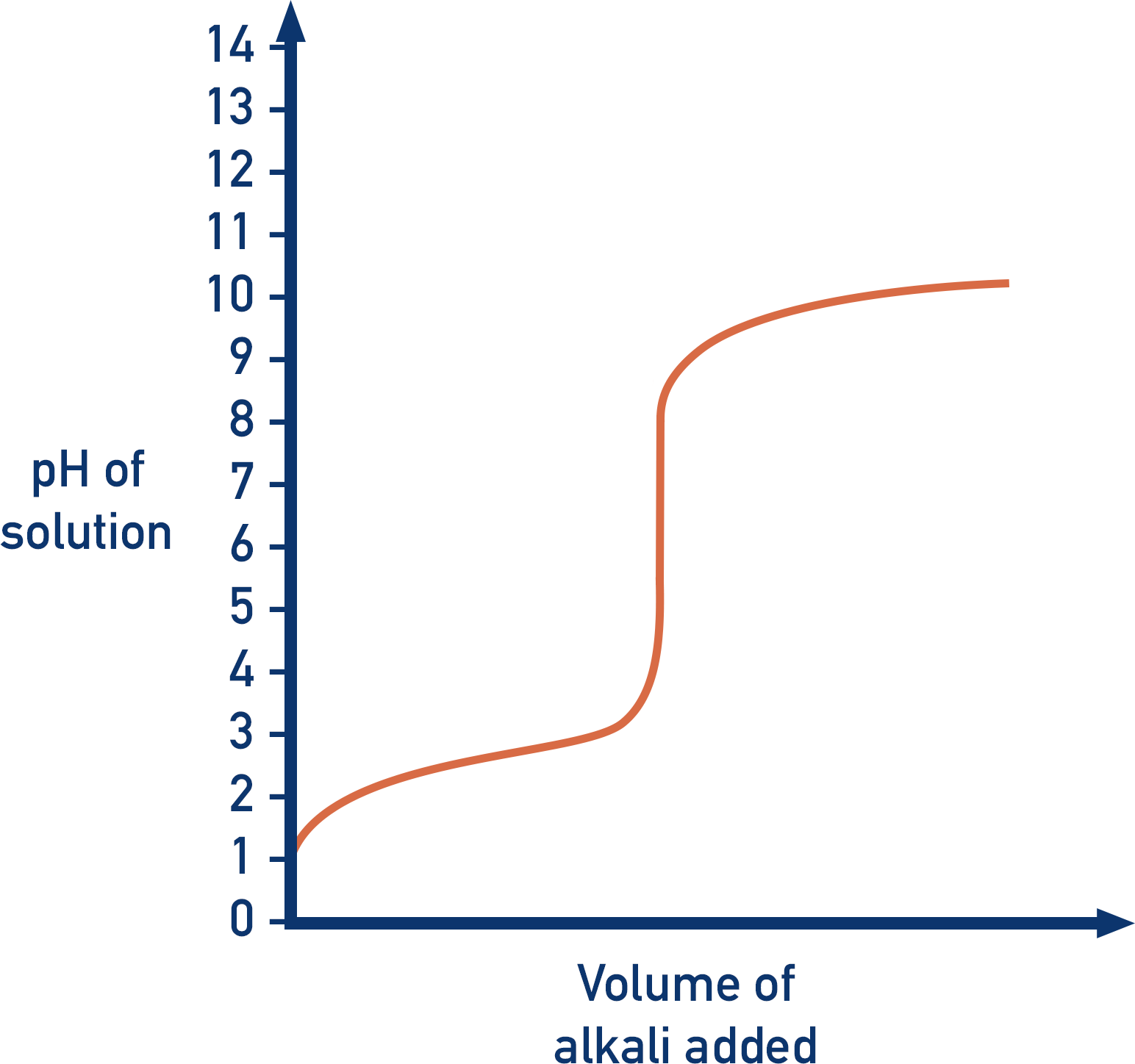
8. Strong Acid–Weak Base Titration
Example:
HCl + NH₃ → NH₄Cl
Characteristics:
- Equivalence point below pH 7
- pH change less steep
Indicators such as methyl orange are commonly used.
9. Weak Acid–Weak Base Titration
These titrations are more complex.
Characteristics:
- No sharp pH change
- Difficult to determine endpoint
- Indicators often ineffective
Potentiometric methods are sometimes used instead.
10. Titration Curves




A titration curve is a graph showing pH versus volume of titrant added.
The curve illustrates several important regions:
- Initial pH
- Buffer region
- Equivalence point
- Endpoint
Titration curves help determine the strength and properties of acids and bases.
11. Indicators Used in Titrations
Indicators are substances that change color at specific pH ranges.
Common indicators include:
Phenolphthalein
Colorless in acid
Pink in base
pH range: 8.2–10
Methyl Orange
Red in acid
Yellow in base
pH range: 3.1–4.4
Bromothymol Blue
Yellow in acid
Blue in base
pH range: 6.0–7.6
12. Selection of Indicators
Choosing the correct indicator is important.
The indicator must change color near the equivalence point.
Example:
Strong acid–strong base titration → phenolphthalein or methyl orange.
13. Calculations in Acid–Base Titrations
The main calculation is based on stoichiometry.
For a reaction:
aA + bB → products
The formula:
[
M_a V_a = M_b V_b
]
Where:
- M = molarity
- V = volume
This equation is used to calculate unknown concentration.
14. Applications of Acid–Base Titrations
Acid–base titration has many applications.
Pharmaceutical Industry
Determining drug purity.
Food Industry
Measuring acidity in foods.
Example:
Acidity of vinegar.
Environmental Monitoring
Testing water acidity.
Agriculture
Soil acidity analysis.
Chemical Manufacturing
Quality control in production processes.
15. Advantages of Acid–Base Titrations
- High accuracy
- Simple experimental setup
- Cost-effective
- Applicable to many chemical systems
16. Limitations of Acid–Base Titrations
Some limitations include:
- Indicator error
- Human observation error
- Weak acid–weak base titrations are difficult
Despite these limitations, titration remains one of the most reliable analytical techniques.
17. Importance of Acid–Base Titrations
Acid–base titration is an essential technique in analytical chemistry.
It allows scientists to:
- Determine unknown concentrations
- Study acid–base reactions
- Analyze chemical purity
- Monitor industrial processes
The method continues to be widely used in research laboratories and industrial applications.
Conclusion
Acid–base titrations are a fundamental analytical method used to determine the concentration of acids and bases through neutralization reactions. By carefully measuring the volume of titrant required to reach the equivalence point, chemists can calculate the concentration of unknown solutions with high accuracy. Different types of titrations exist depending on the strengths of acids and bases involved, and titration curves help illustrate changes in pH during the reaction. Indicators play a crucial role in detecting the endpoint of titration. Acid–base titrations are widely applied in industries, environmental monitoring, pharmaceutical analysis, and scientific research, making them an essential tool in chemistry.
