


1. Introduction to Chemical Reactions
Chemical reactions are processes in which substances called reactants undergo chemical changes to form new substances known as products. During a chemical reaction, atoms are rearranged to create different molecular structures, resulting in substances with new properties.
Chemical reactions are fundamental to chemistry because they explain how matter transforms from one form to another. Every reaction involves the breaking of existing chemical bonds and the formation of new bonds between atoms.
Chemical reactions occur everywhere in nature and daily life. Examples include:
- Rusting of iron
- Burning of fuels
- Digestion of food
- Photosynthesis in plants
- Respiration in living organisms
Scientists classify chemical reactions into different types based on the patterns of reactant and product formation. Understanding these classifications helps chemists predict how substances react and allows them to design reactions for industrial and scientific applications.
The most common types of chemical reactions include:
- Synthesis reactions
- Decomposition reactions
- Single displacement reactions
- Double displacement reactions
- Combustion reactions
Additional classifications include acid–base reactions, redox reactions, and precipitation reactions.
2. Synthesis (Combination) Reactions




A synthesis reaction, also known as a combination reaction, occurs when two or more substances combine to form a single product.
General form:
A + B → AB
In this reaction type, multiple reactants join together to produce one compound.
Example:
2H₂ + O₂ → 2H₂O
In this reaction, hydrogen and oxygen combine to form water.
Another example:
2Na + Cl₂ → 2NaCl
Here, sodium reacts with chlorine to produce sodium chloride.
Synthesis reactions are common in nature and industrial processes. They are often used to produce compounds from simpler substances.
Characteristics of synthesis reactions include:
- Formation of a single product
- Combination of elements or compounds
- Often exothermic reactions
These reactions are important in chemical manufacturing and materials production.
3. Decomposition Reactions

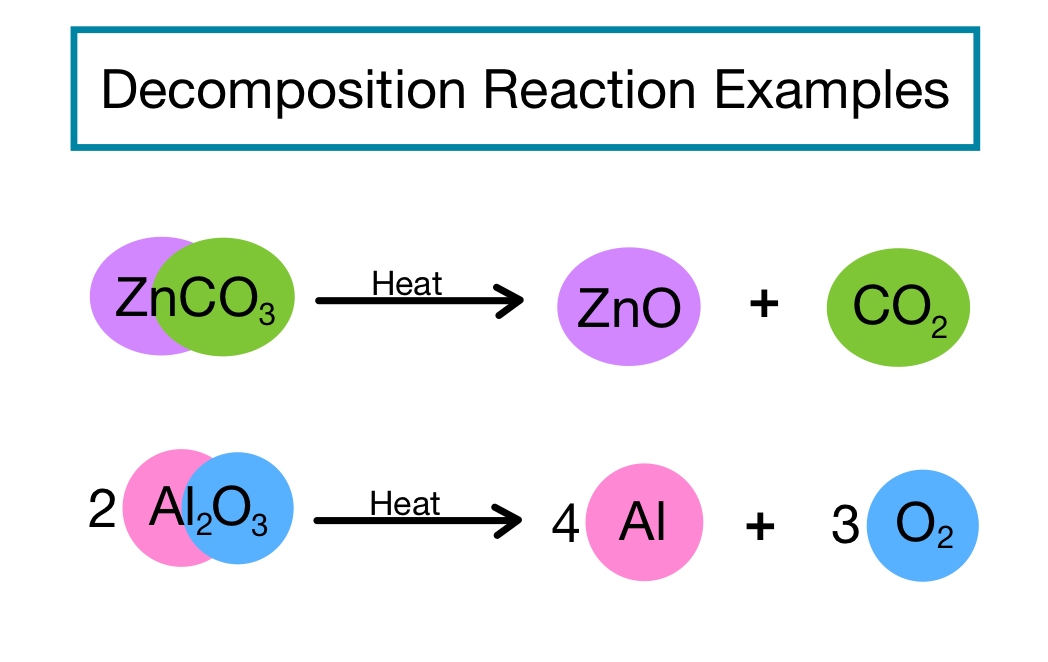


A decomposition reaction occurs when a single compound breaks down into two or more simpler substances.
General form:
AB → A + B
In these reactions, chemical bonds are broken, resulting in simpler products.
Example:
2H₂O₂ → 2H₂O + O₂
Hydrogen peroxide decomposes into water and oxygen.
Another example:
CaCO₃ → CaO + CO₂
Calcium carbonate decomposes into calcium oxide and carbon dioxide.
Decomposition reactions often require energy input, such as heat, light, or electricity.
Types of decomposition reactions include:
- Thermal decomposition
- Electrolytic decomposition
- Photodecomposition
These reactions play important roles in industrial processes and energy production.
4. Single Displacement Reactions


A single displacement reaction, also called a single replacement reaction, occurs when one element replaces another element in a compound.
General form:
A + BC → AC + B
In this reaction, a more reactive element displaces a less reactive element from a compound.
Example:
Zn + CuSO₄ → ZnSO₄ + Cu
Zinc replaces copper in copper sulfate.
Another example:
Cl₂ + 2NaBr → 2NaCl + Br₂
Chlorine displaces bromine from sodium bromide.
Single displacement reactions are governed by reactivity series, which ranks elements according to their ability to displace other elements.
These reactions are widely used in metallurgy and chemical synthesis.
5. Double Displacement Reactions




A double displacement reaction, also known as a double replacement reaction, occurs when two compounds exchange ions to form two new compounds.
General form:
AB + CD → AD + CB
These reactions typically occur in aqueous solutions.
Example:
AgNO₃ + NaCl → AgCl + NaNO₃
In this reaction, silver ions combine with chloride ions to form silver chloride.
Another example:
HCl + NaOH → NaCl + H₂O
This is an acid–base neutralization reaction.
Double displacement reactions often produce:
- Precipitates
- Water
- Gases
These reactions are commonly observed in laboratory experiments and industrial processes.
6. Combustion Reactions



A combustion reaction occurs when a substance reacts rapidly with oxygen to release energy in the form of heat and light.
Combustion reactions often involve hydrocarbons.
Example:
CH₄ + 2O₂ → CO₂ + 2H₂O
Methane reacts with oxygen to produce carbon dioxide and water.
Characteristics of combustion reactions include:
- Rapid oxidation
- Release of heat and light
- Formation of carbon dioxide and water
Combustion reactions are essential in:
- Energy production
- Internal combustion engines
- Industrial heating processes
These reactions power many modern technologies.
7. Acid–Base Reactions




Acid–base reactions involve the transfer of protons between substances.
In these reactions:
- Acids donate hydrogen ions (H⁺).
- Bases accept hydrogen ions.
Example:
HCl + NaOH → NaCl + H₂O
Hydrochloric acid reacts with sodium hydroxide to produce sodium chloride and water.
These reactions are called neutralization reactions.
Acid–base reactions play important roles in:
- Biological systems
- Environmental chemistry
- Industrial processes
- Pharmaceutical chemistry
8. Redox (Oxidation–Reduction) Reactions




Redox reactions involve the transfer of electrons between substances.
Oxidation refers to the loss of electrons, while reduction refers to the gain of electrons.
Example:
Zn + Cu²⁺ → Zn²⁺ + Cu
Zinc loses electrons (oxidation), while copper ions gain electrons (reduction).
Redox reactions occur in many processes such as:
- Corrosion
- Photosynthesis
- Cellular respiration
- Electrochemical batteries
These reactions are fundamental to energy transfer in chemistry.
9. Precipitation Reactions



A precipitation reaction occurs when two aqueous solutions react to form an insoluble solid called a precipitate.
Example:
AgNO₃ + NaCl → AgCl + NaNO₃
Silver chloride forms as a white solid precipitate.
Precipitation reactions are used in:
- Chemical analysis
- Water purification
- Industrial chemical processes
They are commonly used to identify specific ions in solutions.
10. Energy Changes in Chemical Reactions




Chemical reactions can also be classified based on energy changes.
Exothermic Reactions
Release energy as heat or light.
Example:
Combustion reactions.
Endothermic Reactions
Absorb energy from surroundings.
Example:
Photosynthesis.
Energy changes influence reaction rates and feasibility.
11. Importance of Chemical Reactions
Chemical reactions are essential for understanding the transformation of matter.
They explain:
- Biological processes
- Industrial manufacturing
- Environmental chemistry
- Energy production
- Material synthesis
By studying reaction types, chemists can predict how substances behave under different conditions.
12. Conclusion
Chemical reactions describe the transformation of substances through the rearrangement of atoms and the breaking and formation of chemical bonds. Classifying reactions into types such as synthesis, decomposition, displacement, combustion, and redox reactions helps chemists understand and predict chemical behavior.
These reactions occur constantly in nature, from biological processes inside living organisms to large-scale industrial chemical production. Understanding the types of chemical reactions allows scientists to control chemical processes, develop new materials, produce energy, and improve technologies.
Chemical reactions form the foundation of chemistry and play a central role in shaping the physical world and advancing scientific knowledge.
