


1. Introduction to Hybridization
Hybridization is a fundamental concept in chemistry that explains how atomic orbitals combine to form new orbitals suitable for bonding in molecules. The concept was introduced by the scientist Linus Pauling to explain the shapes and bonding patterns of molecules that could not be adequately described by simple atomic orbital theory.
In atoms, electrons occupy atomic orbitals such as s, p, d, and f orbitals. However, when atoms form molecules, these orbitals can mix or combine to produce new orbitals known as hybrid orbitals. These hybrid orbitals have different shapes, orientations, and energies compared to the original atomic orbitals.
Hybridization allows atoms to form stronger and more directional bonds, which helps explain molecular geometry and chemical bonding patterns. It is closely related to molecular geometry and valence bond theory.
For example, carbon atoms in methane form four identical bonds arranged in a tetrahedral shape. Without hybridization, carbon would have two unpaired electrons and could form only two bonds. Hybridization explains how carbon reorganizes its orbitals to create four equivalent bonding orbitals.
Hybridization is particularly important in:
- Organic chemistry
- Molecular geometry prediction
- Chemical bonding theory
- Materials science
- Biological molecules
Understanding hybridization helps chemists explain the structures of molecules such as methane, ethene, benzene, and many complex organic compounds.
2. Atomic Orbitals and Their Shapes




Before understanding hybridization, it is important to understand atomic orbitals.
Atomic orbitals are regions around the nucleus where electrons are most likely to be found. Each orbital has a characteristic shape.
s Orbitals
- Spherical shape
- Only one orientation
- Found in every energy level
p Orbitals
- Dumbbell-shaped
- Three orientations (px, py, pz)
- Found from the second energy level onward
d Orbitals
- Complex shapes
- Five orientations
- Found from the third energy level onward
f Orbitals
- Even more complex shapes
- Seven orientations
- Found in higher energy levels
Hybridization occurs when these orbitals combine to form hybrid orbitals.
3. Concept of Hybridization




Hybridization involves the mixing of atomic orbitals of similar energy to produce new hybrid orbitals.
These hybrid orbitals:
- Have identical energy levels
- Are oriented in specific directions
- Form stronger covalent bonds
The number of hybrid orbitals formed equals the number of atomic orbitals mixed.
For example:
1 s orbital + 3 p orbitals → 4 sp³ hybrid orbitals
Hybridization occurs in the valence shell of atoms during bond formation.
Hybrid orbitals explain the orientation of bonds and the shapes of molecules.
4. Types of Hybridization




Several types of hybridization exist depending on the number of orbitals involved.
sp Hybridization
sp hybridization occurs when one s orbital mixes with one p orbital.
Total hybrid orbitals formed: 2
Geometry: Linear
Bond angle: 180°
Example:
Acetylene (C₂H₂)
In this molecule, carbon atoms form two sp hybrid orbitals.
The remaining p orbitals form π bonds.
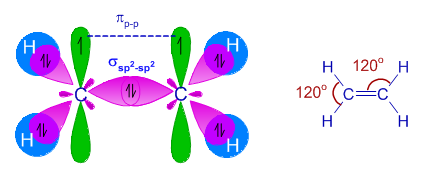
sp² Hybridization
sp² hybridization occurs when one s orbital mixes with two p orbitals.
Total hybrid orbitals formed: 3
Geometry: Trigonal planar
Bond angle: 120°
Example:
Ethene (C₂H₄)
Each carbon atom forms three sp² hybrid orbitals.
The remaining p orbital forms a π bond between carbon atoms.
sp³ Hybridization
sp³ hybridization occurs when one s orbital mixes with three p orbitals.
Total hybrid orbitals formed: 4
Geometry: Tetrahedral
Bond angle: 109.5°
Example:
Methane (CH₄)
The carbon atom forms four equivalent sp³ hybrid orbitals that bond with hydrogen atoms.
5. Advanced Hybridization Types




Some molecules involve hybridization with d orbitals.
sp³d Hybridization
Involves one s orbital, three p orbitals, and one d orbital.
Total hybrid orbitals: 5
Geometry: Trigonal bipyramidal
Bond angles: 90° and 120°
Example:
Phosphorus pentachloride (PCl₅)
sp³d² Hybridization
Involves one s orbital, three p orbitals, and two d orbitals.
Total hybrid orbitals: 6
Geometry: Octahedral
Bond angle: 90°
Example:
Sulfur hexafluoride (SF₆)
6. Hybridization and Sigma and Pi Bonds




Hybridization helps explain the formation of two types of covalent bonds.
Sigma (σ) Bonds
Sigma bonds form through head-on overlap of orbitals.
They are the strongest type of covalent bond.
Examples:
sp³–s overlap in methane.
Pi (π) Bonds
Pi bonds form through side-by-side overlap of p orbitals.
They occur in double and triple bonds.
Example:
Ethene contains one σ bond and one π bond between carbon atoms.
7. Hybridization in Organic Molecules




Hybridization is especially important in organic chemistry.
Methane (CH₄)
Carbon is sp³ hybridized.
Geometry: tetrahedral.
Ethene (C₂H₄)
Carbon atoms are sp² hybridized.
Contains a carbon-carbon double bond.
Acetylene (C₂H₂)
Carbon atoms are sp hybridized.
Contains a carbon-carbon triple bond.
Benzene (C₆H₆)
Carbon atoms are sp² hybridized.
The molecule has a delocalized π electron system.
8. Importance of Hybridization
Hybridization explains many important chemical phenomena.
It helps explain:
- Molecular shapes
- Bond angles
- Bond strength
- Reactivity of molecules
- Structure of organic compounds
Hybridization is also important in understanding:
- Catalysis
- Molecular spectroscopy
- Chemical reactions
9. Applications in Modern Chemistry
Hybridization concepts are widely used in many scientific fields.
Organic Chemistry
Understanding carbon bonding patterns.
Drug Design
Molecular shapes influence biological activity.
Materials Science
Carbon hybridization leads to materials such as diamond and graphene.
Nanotechnology
Hybridized orbitals determine the structure of nanomaterials.
10. Limitations of Hybridization Theory
Although hybridization is very useful, it has limitations.
It is primarily part of valence bond theory and may not accurately describe some molecules.
For complex molecules, molecular orbital theory provides a more accurate description.
Despite these limitations, hybridization remains one of the most widely used concepts for explaining molecular structure.
11. Conclusion
Hybridization is a key concept in chemistry that explains how atomic orbitals combine to form hybrid orbitals suitable for chemical bonding. By mixing s, p, and sometimes d orbitals, atoms can form hybrid orbitals that produce specific molecular shapes and bond angles.
Different types of hybridization, including sp, sp², sp³, sp³d, and sp³d², correspond to different molecular geometries such as linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral structures.
Hybridization plays a crucial role in understanding molecular geometry, chemical bonding, and the behavior of organic and inorganic molecules. It helps explain the structure of countless chemical compounds, from simple molecules to complex biological macromolecules.
Because of its ability to connect atomic structure with molecular shape, hybridization remains one of the most important and widely used concepts in modern chemistry.
