
Introduction
Electron spin is a fundamental quantum property of electrons that describes an intrinsic form of angular momentum carried by the particle. Unlike classical rotation, electron spin does not correspond to the electron physically spinning around its axis. Instead, it is a purely quantum mechanical property that arises from the mathematical structure of quantum theory.
Electron spin plays a crucial role in many areas of physics, including atomic structure, magnetism, spectroscopy, and quantum mechanics. The concept was introduced in 1925 by physicists George Uhlenbeck and Samuel Goudsmit to explain certain spectral phenomena that could not be accounted for by earlier atomic models.
The discovery of electron spin significantly improved the understanding of atomic structure and led to the development of the Pauli exclusion principle, proposed by Wolfgang Pauli. This principle explains the arrangement of electrons in atoms and the structure of the periodic table.
Electron spin also contributes to magnetic properties of materials and is essential in modern technologies such as magnetic resonance imaging (MRI), spintronics, and quantum computing.
Nature of Electron Spin
Electron spin is a type of intrinsic angular momentum possessed by electrons. It differs from classical angular momentum in several ways:
- It is intrinsic, meaning it exists even when the electron is not moving.
- It is quantized, meaning it can take only certain discrete values.
- It is described mathematically by quantum mechanics rather than classical physics.
The magnitude of electron spin angular momentum is given by:
[
S = \sqrt{s(s+1)}\hbar
]
where
- (s = \frac{1}{2}) for electrons
- (\hbar = \frac{h}{2\pi})
Thus the spin angular momentum magnitude becomes:
[
S = \sqrt{\frac{3}{4}}\hbar
]
This shows that electron spin is a fixed property of the electron.
Spin Quantum Number
The spin of an electron is characterized by the spin quantum number.
[
s = \frac{1}{2}
]
The orientation of spin is described by the magnetic spin quantum number:
[
m_s = +\frac{1}{2}, -\frac{1}{2}
]
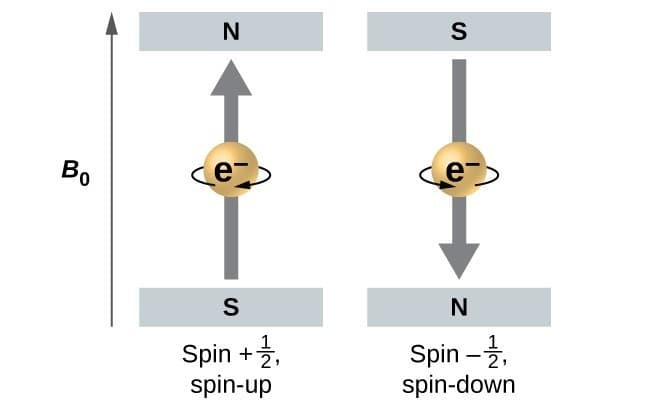
These two values correspond to the two possible spin orientations:
- Spin-up
- Spin-down
These orientations are often represented using arrows:
↑ (spin up)
↓ (spin down)
Historical Discovery of Electron Spin
The concept of electron spin emerged from attempts to explain anomalies in atomic spectra.
Scientists observed that certain spectral lines were split into multiple components, a phenomenon known as fine structure. Classical atomic models could not explain these observations.
In 1925, George Uhlenbeck and Samuel Goudsmit proposed that electrons possess intrinsic angular momentum.
Their hypothesis successfully explained many spectroscopic observations and was later confirmed experimentally.
Stern–Gerlach Experiment


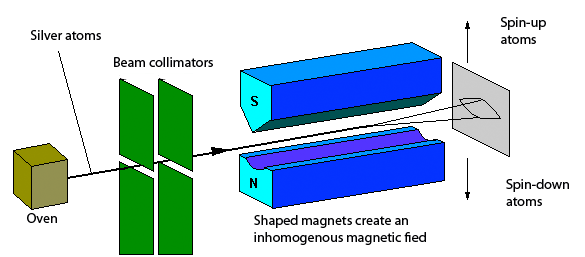
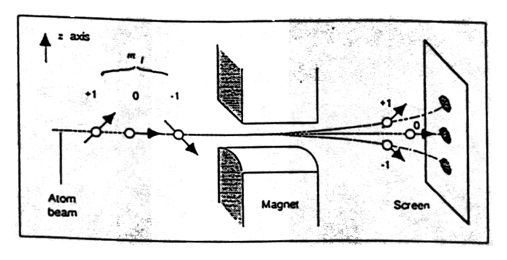
One of the most important experiments demonstrating quantum spin is the Stern–Gerlach experiment, conducted by Otto Stern and Walther Gerlach.
Experimental Setup
The experiment involved:
- A beam of silver atoms
- A non-uniform magnetic field
- A detector screen
Observations
If angular momentum were continuous, the beam would spread out continuously. However, the beam split into two distinct components.
Conclusion
The result showed that angular momentum is quantized.
The two beam components correspond to the two possible spin orientations of electrons.
Spin Magnetic Moment
Electron spin produces a magnetic moment, meaning that electrons behave like tiny magnets.
The magnetic moment associated with spin is given by:
[
\mu_s = g \frac{e}{2m_e} S
]
where:
- (g) = gyromagnetic ratio
- (e) = electron charge
- (m_e) = electron mass
This magnetic moment explains why electrons interact with magnetic fields.
Spin and the Pauli Exclusion Principle
Electron spin is central to the Pauli exclusion principle, proposed by Wolfgang Pauli.
The principle states:
No two electrons in an atom can have the same set of four quantum numbers.
As a result:
- Each atomic orbital can hold at most two electrons
- The two electrons must have opposite spins
This rule determines how electrons fill atomic orbitals.
Spin-Orbit Interaction
Electron spin interacts with its orbital motion around the nucleus.
This interaction is known as spin–orbit coupling.
Spin–orbit coupling causes:
- Splitting of energy levels
- Fine structure in atomic spectra
The strength of this interaction increases with atomic number.
Fine Structure of Atomic Spectra
Fine structure refers to the small splitting of spectral lines observed in atomic spectra.
The splitting arises from:
- Spin–orbit interaction
- Relativistic effects
- Electron spin contributions
Fine structure provides evidence for electron spin and quantum mechanics.
Electron Spin in Magnetic Fields
When electrons are placed in an external magnetic field, their spin magnetic moments interact with the field.
This interaction leads to phenomena such as:
- Zeeman effect
- Magnetic resonance
The energy difference between spin states in a magnetic field is given by:
[
\Delta E = g \mu_B B
]
where:
- (\mu_B) = Bohr magneton
- (B) = magnetic field strength
Electron Spin Resonance
Electron spin resonance (ESR), also called electron paramagnetic resonance (EPR), is a technique used to study materials containing unpaired electrons.
In ESR:
- A magnetic field splits spin energy levels.
- Microwave radiation induces transitions between spin states.
This technique is used to study:
- Chemical radicals
- Transition metal complexes
- Defects in solids
Spin and Magnetism
Electron spin is responsible for many magnetic properties of materials.
Magnetic behavior arises from electron spins in atoms.
Types of magnetism include:
Diamagnetism
Occurs when all electron spins are paired.
Paramagnetism
Occurs when atoms have unpaired electrons.
Ferromagnetism
Occurs when electron spins align collectively.
Ferromagnetic materials include iron, cobalt, and nickel.
Spin in Quantum Mechanics
In quantum mechanics, spin is described using spin operators.
The spin components along different axes are represented by matrices known as Pauli matrices.
These matrices were introduced by Wolfgang Pauli.
The three Pauli matrices are:
[
\sigma_x, \sigma_y, \sigma_z
]
They describe the quantum behavior of spin systems.
Spin and Quantum Statistics
Electrons belong to a class of particles called fermions.
Fermions have half-integer spin values.
Particles with half-integer spin obey Fermi–Dirac statistics.
This statistical behavior explains many properties of matter, including:
- Electron configurations
- Stability of matter
- Structure of solids
Spintronics
Electron spin has become an important resource in modern technology.
Spintronics is a field that uses both the charge and spin of electrons in electronic devices.
Spintronic devices include:
- Magnetic memory
- Spin transistors
- Magnetic sensors
Spintronics has potential applications in faster and more efficient electronics.
Electron Spin in Quantum Computing
Electron spin is used as a quantum bit (qubit) in quantum computing.
Spin states:
- Spin-up
- Spin-down
represent the two states of a qubit.
Quantum computers use superposition and entanglement of spin states to perform complex computations.
Measurement of Electron Spin
Electron spin can be measured using several experimental techniques.
Examples include:
- Stern–Gerlach experiments
- Electron spin resonance
- Magnetic resonance techniques
These methods detect the magnetic moment associated with electron spin.
Importance of Electron Spin
Electron spin is one of the most important quantum properties of matter.
It influences:
- Atomic structure
- Chemical bonding
- Magnetic properties
- Spectral lines
- Electronic materials
Without electron spin, many features of modern physics and chemistry would remain unexplained.
Modern Research on Electron Spin
Scientists continue to study electron spin in advanced fields such as:
- Quantum information science
- Condensed matter physics
- Nanotechnology
- Topological materials
Understanding spin behavior may lead to breakthroughs in future technologies.
Conclusion
Electron spin is a fundamental quantum property that plays a central role in atomic physics and quantum mechanics. Although it is often described as a type of angular momentum, spin is not a classical rotation but an intrinsic property of particles.
The discovery of electron spin helped explain fine structure in atomic spectra and led to the development of the Pauli exclusion principle, which governs electron configurations in atoms. Spin interactions with magnetic fields give rise to phenomena such as the Zeeman effect and electron spin resonance.
Electron spin is also responsible for many magnetic properties of materials and has become a key concept in modern technologies such as spintronics and quantum computing. The study of electron spin continues to be an active and important area of research in physics, offering insights into both fundamental science and technological innovation.
