


Introduction
Coulomb’s Law is a fundamental principle of electrostatics that describes the force between two electrically charged particles. It explains how charged objects interact with each other through attractive or repulsive forces. This law forms the foundation of the study of electric fields, electrostatic interactions, and electromagnetic phenomena.
Coulomb’s Law was formulated in 1785 by the French physicist Charles-Augustin de Coulomb, who conducted precise experiments using a torsion balance to measure the force between charged objects. His experiments revealed that the electrostatic force between two charges depends on the magnitude of the charges and the distance between them.
Coulomb’s Law is extremely important in physics because it provides a quantitative description of electric forces similar to how Newton’s law of gravitation describes gravitational forces.
The law plays a crucial role in many areas such as:
- Atomic physics
- Electrical engineering
- Electronics
- Plasma physics
- Material science
- Electromagnetism
Understanding Coulomb’s Law allows scientists and engineers to predict how charged particles interact in various physical systems.
Historical Background




The study of electrostatic forces began centuries ago when scientists observed that certain materials such as amber could attract small objects after being rubbed.
However, a precise mathematical description of the electric force was developed in the late 18th century by Charles-Augustin de Coulomb.
Coulomb’s Experiment
Coulomb used a device called a torsion balance, which consisted of:
- A thin fiber
- Two small charged spheres
- A scale for measuring rotation
When two charged spheres were placed near each other, the electrostatic force caused the fiber to twist. By measuring the angle of twist, Coulomb determined the magnitude of the electric force.
His experiments showed that:
- The force between charges is proportional to the product of the charges.
- The force decreases with the square of the distance between them.
These observations led to the formulation of Coulomb’s Law.
Statement of Coulomb’s Law




Coulomb’s Law states that:
The electrostatic force between two point charges is directly proportional to the product of their charges and inversely proportional to the square of the distance between them.
Mathematical Expression
[
F = k \frac{|q_1 q_2|}{r^2}
]
Where:
- (F) = Electrostatic force between charges
- (q_1) = First charge
- (q_2) = Second charge
- (r) = Distance between the charges
- (k) = Coulomb’s constant
Coulomb’s Constant
[
k = 9 \times 10^9 , N,m^2/C^2
]
The value of the constant depends on the medium between the charges.
Nature of the Electrostatic Force



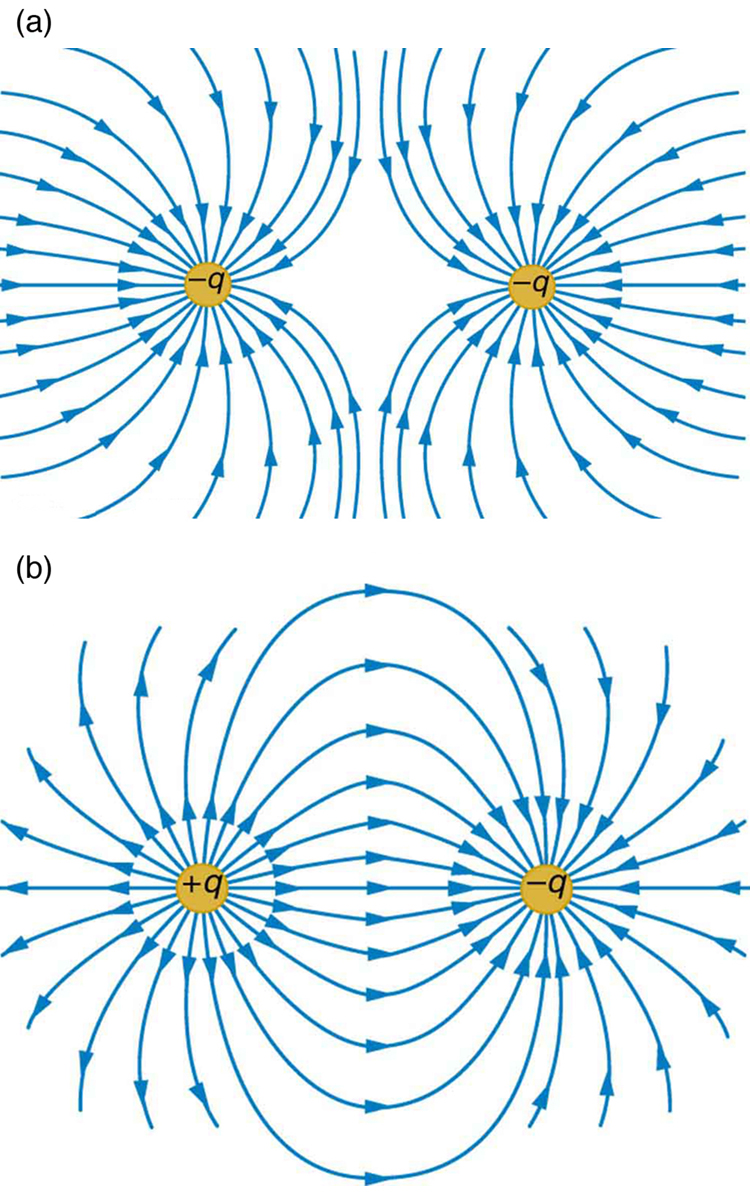
The electrostatic force described by Coulomb’s Law has specific characteristics.
Attractive Force
When charges have opposite signs, they attract each other.
Example:
- Electron and proton interaction.
Repulsive Force
When charges have same signs, they repel each other.
Example:
- Two electrons repel each other.
Line of Action
The force acts along the straight line joining the two charges.
Action and Reaction
The forces on the two charges are equal in magnitude and opposite in direction, according to Newton’s Third Law of Motion.
Inverse Square Law




Coulomb’s Law follows an inverse square relationship.
[
F \propto \frac{1}{r^2}
]
This means that if the distance between charges doubles, the force becomes one-fourth of its original value.
Example:
| Distance | Force |
|---|---|
| r | F |
| 2r | F/4 |
| 3r | F/9 |
This relationship is similar to gravitational force.
Vector Form of Coulomb’s Law


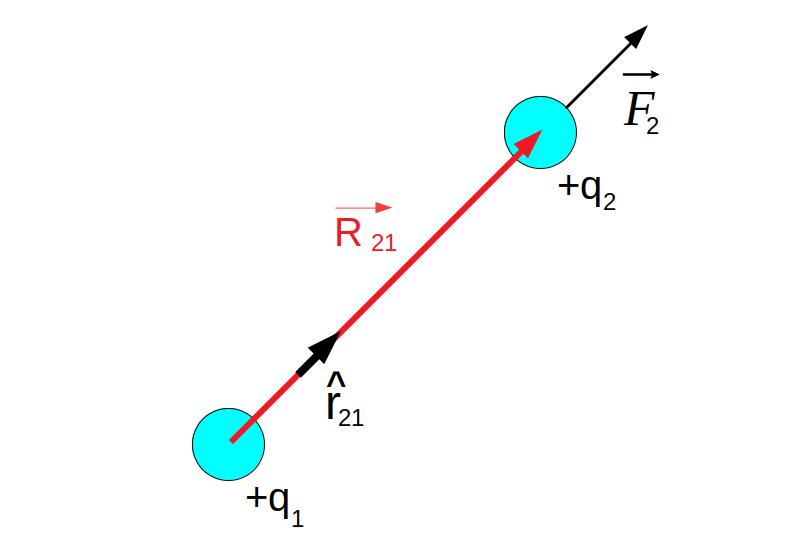
Coulomb’s Law can also be expressed in vector form.
[
\vec{F} = k \frac{q_1 q_2}{r^2} \hat{r}
]
Where:
- (\vec{F}) = force vector
- (\hat{r}) = unit vector along the line joining the charges
This form describes both the magnitude and direction of the force.
Principle of Superposition



In systems with multiple charges, the total force is calculated using the principle of superposition.
Definition
The total force on a charge is the vector sum of all individual forces acting on it.
Example:
If three charges act on a particle:
[
F_{total} = F_1 + F_2 + F_3
]
Each force is calculated using Coulomb’s Law.
Electric Field and Coulomb’s Law




Coulomb’s Law leads to the concept of the electric field.
Electric Field Definition
The electric field is the force experienced per unit charge.
[
E = \frac{F}{q}
]
Substituting Coulomb’s Law:
[
E = k \frac{Q}{r^2}
]
This equation describes the electric field produced by a point charge.
Electric Potential and Coulomb’s Law
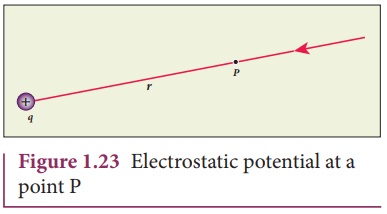


Electric potential is related to Coulomb’s Law.
Electric Potential
[
V = k \frac{Q}{r}
]
Where:
- (V) = electric potential
- (Q) = source charge
- (r) = distance from charge
Electric potential determines how much work is required to move a charge in an electric field.
Coulomb’s Law in Different Media

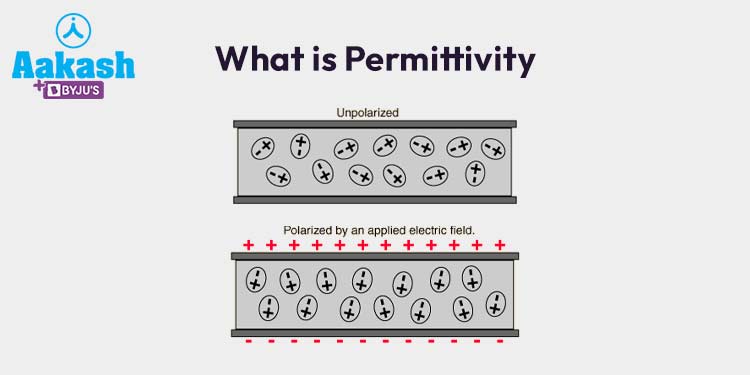


The electrostatic force depends on the medium between charges.
Permittivity
The constant (k) can be expressed using permittivity of free space.
[
k = \frac{1}{4\pi \varepsilon_0}
]
Where:
[
\varepsilon_0 = 8.85 \times 10^{-12} , C^2/Nm^2
]
In materials:
[
F = \frac{1}{4\pi \varepsilon} \frac{q_1 q_2}{r^2}
]
Different materials reduce electrostatic force due to dielectric effects.
Applications of Coulomb’s Law




Coulomb’s Law has many practical applications.
Atomic Structure
Electrostatic attraction between electrons and nuclei determines atomic structure.
Capacitors
Capacitors store electrical energy using electric charges on plates.
Electrostatic Precipitators
Used to remove dust and pollutants from industrial exhaust gases.
Photocopiers and Printers
Electrostatic forces help transfer toner particles to paper.
Electrostatic Painting
Charged paint particles are attracted to metal surfaces.
Comparison with Gravitational Law



Coulomb’s Law is mathematically similar to Newton’s law of gravitation.
| Feature | Coulomb’s Law | Gravitational Law |
|---|---|---|
| Interaction | Electric charges | Masses |
| Force type | Attractive or repulsive | Always attractive |
| Strength | Very strong | Very weak |
| Equation form | Inverse square | Inverse square |
Electrostatic forces are much stronger than gravitational forces at atomic scales.
Limitations of Coulomb’s Law
Coulomb’s Law works accurately under certain conditions.
Limitations
- Valid only for point charges
- Applies when charges are stationary
- Not valid for moving charges (requires electromagnetic theory)
- Does not include relativistic effects
For moving charges, the full theory of electromagnetism must be used.
Importance of Coulomb’s Law
Coulomb’s Law is essential because it:
- Explains electrostatic interactions
- Forms the basis of electric field theory
- Helps understand atomic and molecular forces
- Supports electrical engineering and electronics
- Leads to the development of Maxwell’s equations
It is one of the fundamental laws governing electricity and magnetism.
Conclusion
Coulomb’s Law provides a mathematical description of the electrostatic force between charged particles. It states that the force between two charges depends on the product of their charges and the square of the distance separating them.
This law explains the attraction and repulsion of electric charges and forms the foundation for understanding electric fields, electric potential, and many other electromagnetic phenomena.
From atomic interactions to modern technologies such as capacitors, printers, and pollution control systems, Coulomb’s Law plays a critical role in science and engineering. Its principles continue to be essential for studying electricity, electronics, and advanced physical theories.
