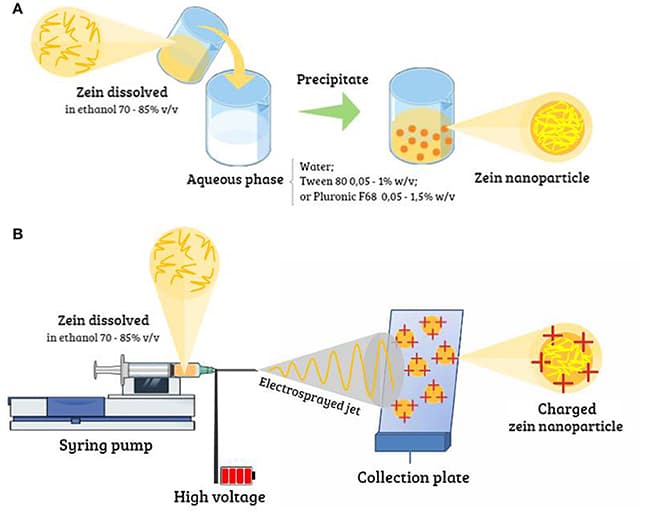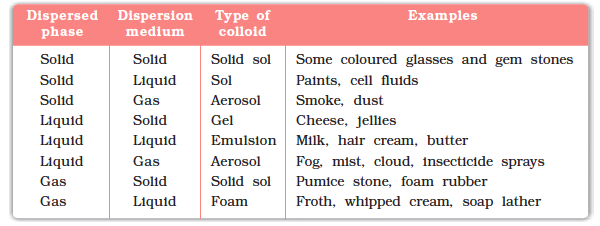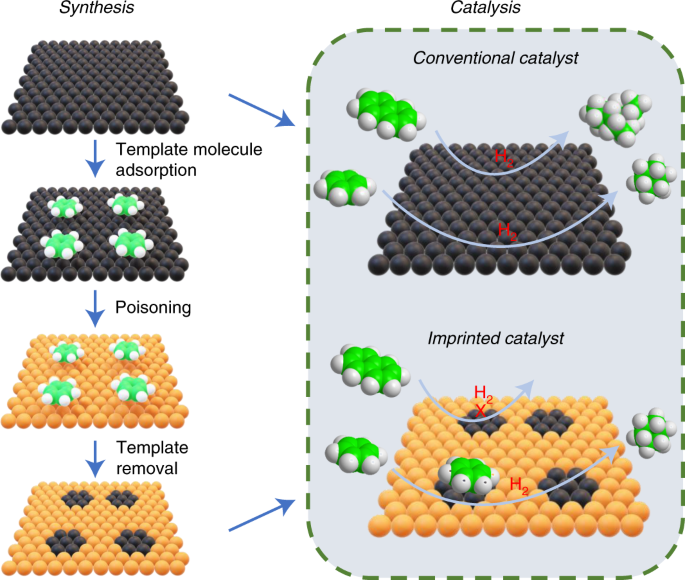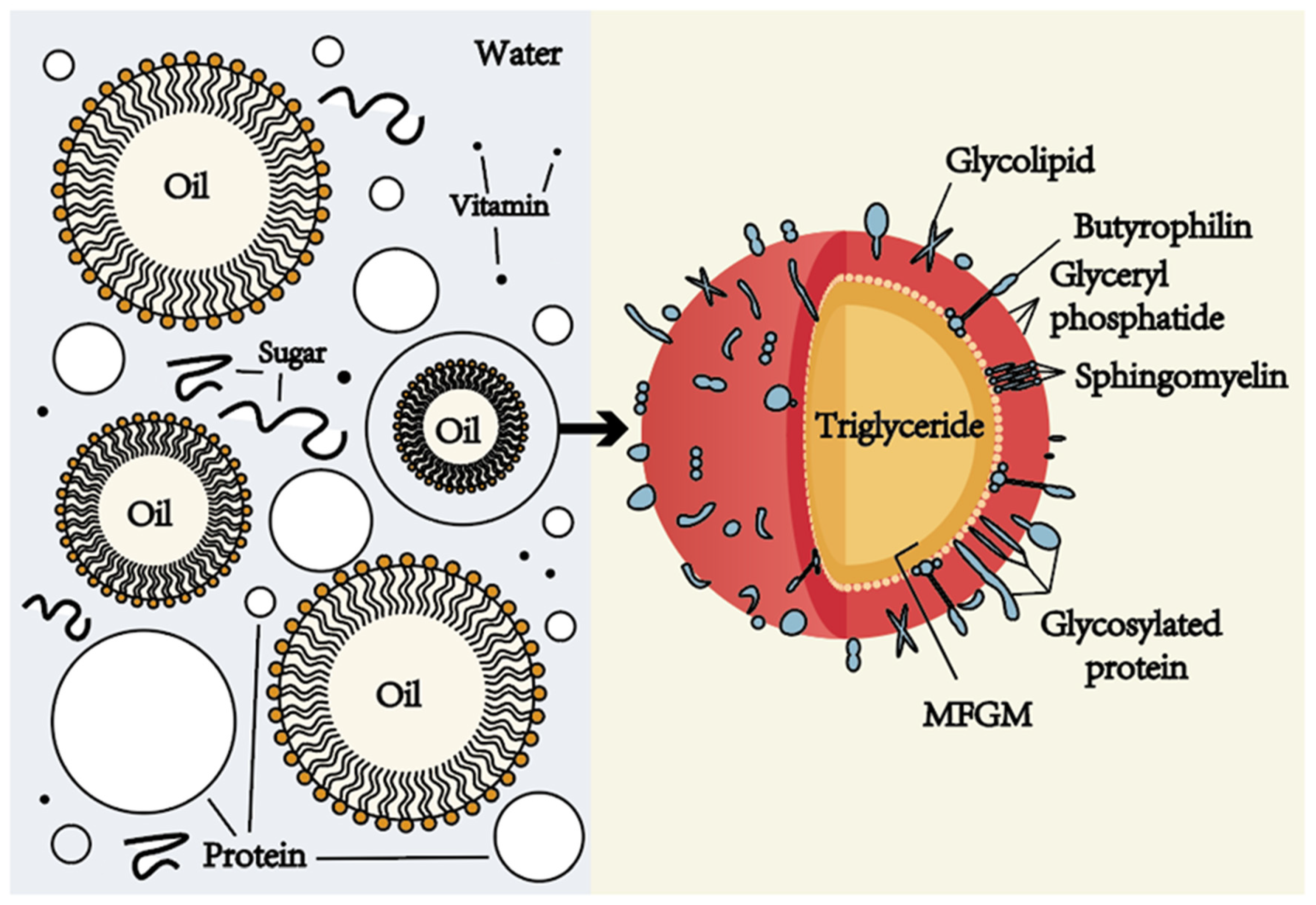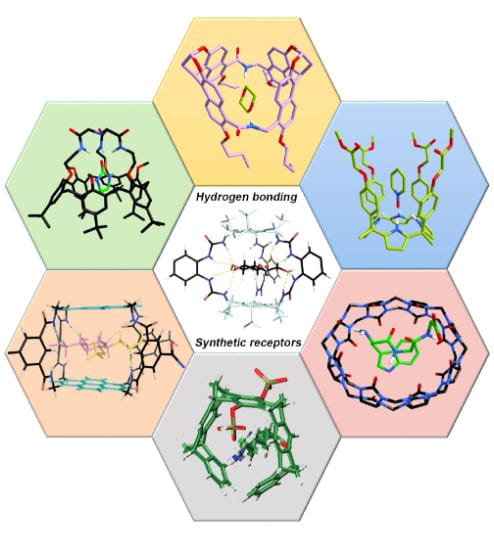
1. Introduction to Supramolecular Chemistry
Supramolecular chemistry is the branch of chemistry that studies molecular assemblies formed through non-covalent interactions between molecules. While traditional chemistry focuses on covalent bonds within molecules, supramolecular chemistry focuses on the interactions between molecules, often referred to as “chemistry beyond the molecule.”
The term supramolecular refers to structures formed by the association of two or more molecules through weak intermolecular forces such as hydrogen bonding, van der Waals interactions, electrostatic interactions, and hydrophobic effects.
Supramolecular chemistry was popularized in the late 20th century, particularly through the work of scientists such as Jean-Marie Lehn, Donald J. Cram, and Charles J. Pedersen, who received the Nobel Prize in Chemistry in 1987 for their work on molecular recognition and host–guest chemistry.
This field has become extremely important in modern chemistry because it helps explain many biological processes and enables the development of advanced technologies.
Supramolecular chemistry is used in:
- Molecular recognition systems
- Drug delivery technologies
- Nanotechnology
- Sensors and molecular devices
- Materials science
Because supramolecular structures rely on weak interactions, they can assemble and disassemble dynamically, allowing scientists to create adaptive and responsive molecular systems.
2. Concept of Molecular Recognition
Molecular recognition is a fundamental concept in supramolecular chemistry.
It refers to the specific interaction between two or more molecules through non-covalent bonding.
These interactions are highly selective, meaning certain molecules recognize and bind only to specific partners.
Examples of molecular recognition include:
- Enzyme–substrate interactions
- Antibody–antigen binding
- DNA base pairing
Molecular recognition plays a crucial role in biological systems and is widely studied in supramolecular chemistry.
3. Non-Covalent Interactions


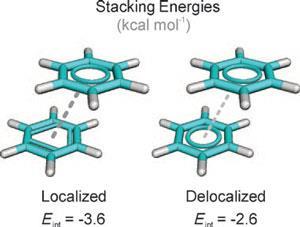

Supramolecular chemistry relies on several types of weak intermolecular forces.
Hydrogen Bonding
Hydrogen bonding occurs when a hydrogen atom bonded to an electronegative atom interacts with another electronegative atom.
This interaction plays an important role in:
- DNA structure
- Protein folding
- Molecular recognition
Van der Waals Forces
These are weak attractive forces between molecules due to temporary dipoles.
Although individually weak, they become significant when many interactions occur simultaneously.
π–π Interactions
These interactions occur between aromatic rings.
They are important in molecular stacking and supramolecular assemblies.
Electrostatic Interactions
Attraction between oppositely charged ions or molecules.
These interactions are important in host–guest chemistry.
4. Host–Guest Chemistry




Host–guest chemistry involves the interaction between a host molecule and a guest molecule.
The host molecule contains a cavity or binding site that can accommodate the guest molecule.
Examples include:
- Crown ethers binding metal ions
- Cyclodextrins forming inclusion complexes
These complexes are held together by non-covalent interactions.
Host–guest systems are widely used in drug delivery and molecular sensing.
5. Self-Assembly in Supramolecular Chemistry




Self-assembly is the spontaneous organization of molecules into ordered structures.
This process occurs without external direction and is driven by non-covalent interactions.
Examples include:
- Formation of micelles
- Lipid bilayers in cell membranes
- DNA double helix structure
Self-assembly is widely used in nanotechnology to create complex molecular structures.
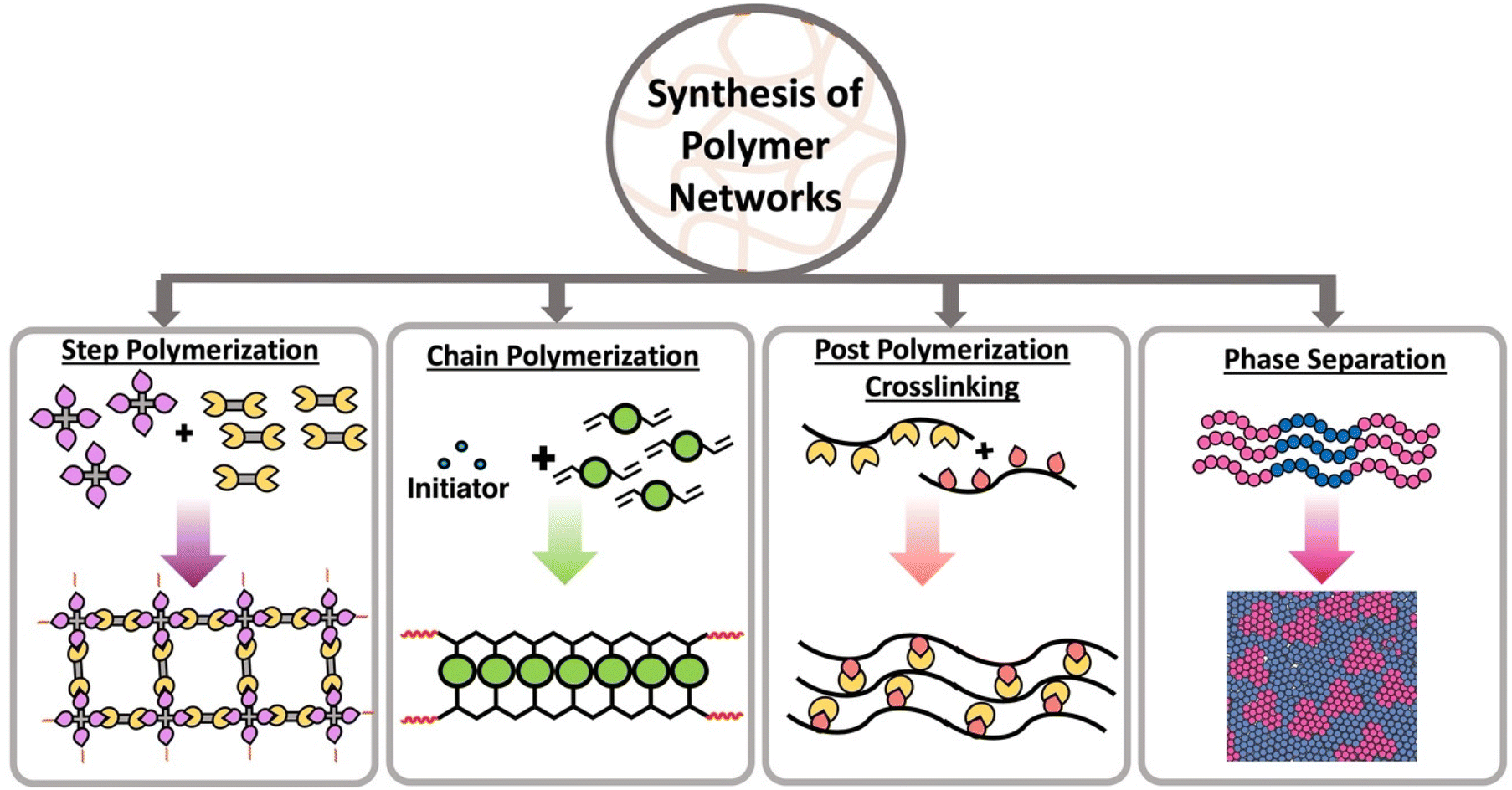
6. Supramolecular Polymers
Supramolecular polymers are polymers formed through reversible non-covalent interactions rather than covalent bonds.
These polymers have unique properties such as:
- Self-healing ability
- Reversible assembly
- Stimulus responsiveness
Applications include smart materials and adaptive systems.
7. Molecular Machines

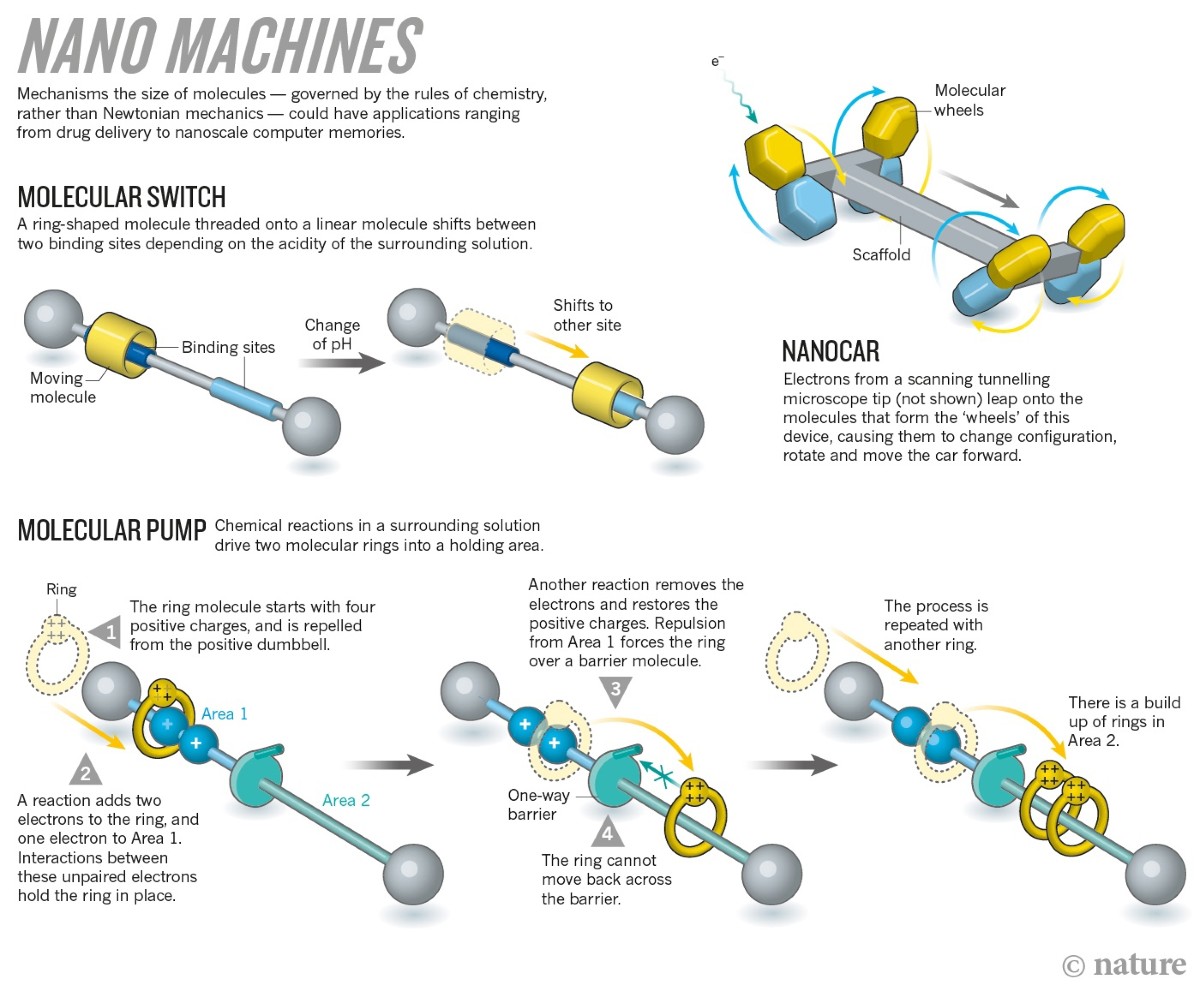
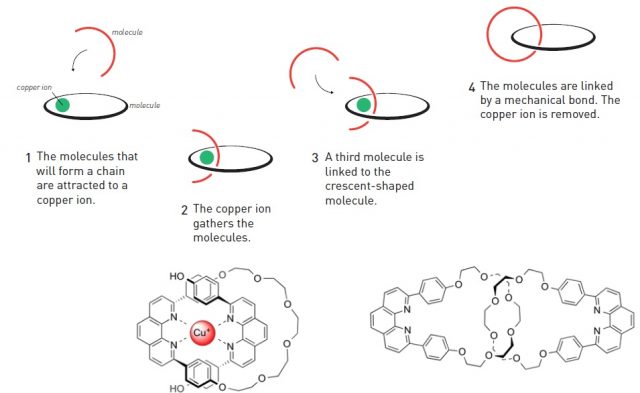
Molecular machines are nanoscale systems capable of performing mechanical movements.
Examples include:
- Rotaxanes
- Catenanes
These molecules contain components that move relative to each other.
In 2016, the Nobel Prize in Chemistry was awarded for the development of molecular machines.
Potential applications include nanoscale robotics and molecular electronics.
8. Supramolecular Materials
Supramolecular chemistry enables the creation of advanced materials with unique properties.
Examples include:
Smart Materials
Materials that respond to environmental changes such as temperature or pH.
Self-Healing Materials
Materials that repair themselves after damage.
Molecular Sensors
Devices that detect specific molecules using molecular recognition.
9. Supramolecular Chemistry in Biology
Many biological processes rely on supramolecular interactions.
Examples include:
DNA Structure
DNA strands are held together by hydrogen bonding between base pairs.
Enzyme–Substrate Interactions
Enzymes recognize and bind specific molecules through molecular recognition.
Cell Membranes
Phospholipid molecules self-assemble into bilayer structures.
These biological systems demonstrate the importance of supramolecular chemistry in life processes.
10. Applications of Supramolecular Chemistry
Supramolecular chemistry has numerous technological applications.
Drug Delivery
Host molecules can transport drugs to specific locations in the body.
Chemical Sensors
Supramolecular systems can detect pollutants or biological molecules.
Nanotechnology
Supramolecular structures are used to construct nanoscale devices.
Materials Science
Self-assembling materials are used in coatings, electronics, and smart materials.
11. Supramolecular Catalysis
Supramolecular catalysts mimic enzyme activity.
These catalysts create specific environments that enhance reaction rates and selectivity.
Such systems are used in advanced chemical synthesis.
12. Advantages of Supramolecular Systems
Supramolecular chemistry offers several advantages.
- Dynamic and reversible interactions
- High selectivity in molecular recognition
- Ability to create complex structures through self-assembly
These properties allow scientists to design adaptable molecular systems.
13. Challenges in Supramolecular Chemistry
Despite its potential, supramolecular chemistry faces challenges.
Examples include:
- Weak interaction stability
- Complex system design
- Difficulty in controlling molecular assembly
Researchers continue to develop methods to overcome these challenges.
14. Future of Supramolecular Chemistry
The future of supramolecular chemistry includes exciting possibilities.
Examples include:
- Molecular robots
- Artificial molecular systems
- Smart nanomaterials
- Advanced drug delivery technologies
These developments could revolutionize medicine, electronics, and materials science.
15. Importance of Supramolecular Chemistry
Supramolecular chemistry is essential for understanding interactions between molecules.
It bridges chemistry with biology, nanotechnology, and materials science.
By studying non-covalent interactions, scientists can design complex molecular systems with advanced functions.
Conclusion
Supramolecular chemistry focuses on molecular assemblies formed through non-covalent interactions such as hydrogen bonding, electrostatic attraction, and van der Waals forces. Unlike traditional chemistry, which focuses on individual molecules, supramolecular chemistry studies how molecules interact to form larger organized systems. Concepts such as molecular recognition, host–guest chemistry, and self-assembly are fundamental to this field. Supramolecular chemistry plays a critical role in biological processes and modern technologies including drug delivery, molecular sensors, nanotechnology, and smart materials. As research continues to advance, supramolecular chemistry is expected to contribute significantly to future developments in science and technology.