


Introduction
The law of conservation of energy is one of the most fundamental principles in physics and science. It states that energy cannot be created or destroyed; it can only be transformed from one form to another. This law applies to all physical processes in nature, from the motion of planets and mechanical systems to chemical reactions and electrical circuits.
Energy is a central concept in physics because it describes the ability of a system to perform work. Every process in nature involves some form of energy transformation. For example, when a ball falls from a height, its gravitational potential energy converts into kinetic energy. When a car burns fuel, chemical energy transforms into mechanical energy and heat.
The conservation of energy principle helps scientists understand and analyze complex systems by tracking how energy moves and changes form. It plays a crucial role in fields such as mechanics, thermodynamics, electromagnetism, chemistry, biology, and engineering.
Because energy cannot disappear or be created spontaneously, this law allows physicists to predict how systems behave and ensures that physical processes follow consistent rules.
Historical Development of Energy Conservation
The concept of energy conservation developed gradually through the work of several scientists during the 18th and 19th centuries. Early thinkers observed that motion and heat were related, but it took many years before scientists fully understood the relationship between different forms of energy.
Scientists such as James Prescott Joule, Hermann von Helmholtz, Julius Robert Mayer, and others contributed to the development of the conservation of energy principle.
James Joule conducted experiments demonstrating that mechanical work could be converted into heat. His experiments showed that energy transformations follow precise quantitative relationships. These discoveries led to the formulation of the law of conservation of energy.
Today, this law is considered a universal principle that applies to all physical systems.
Statement of the Law of Conservation of Energy
The law of conservation of energy states:
Energy cannot be created or destroyed in an isolated system; it can only change from one form to another. The total energy of a closed system remains constant over time.
Mathematically, the total energy in a system is expressed as:
[
E_{total} = constant
]
This means that if energy changes form within a system, the sum of all forms of energy remains unchanged.
Forms of Energy
Energy exists in many different forms. Understanding these forms helps explain how energy transformations occur in physical systems.
Mechanical Energy
Mechanical energy is the energy associated with the motion or position of objects. It consists of two main components:
Kinetic energy
Potential energy
Mechanical energy can be written as:
[
E = KE + PE
]
Where:
KE = kinetic energy
PE = potential energy
Mechanical energy is commonly observed in moving machines, vehicles, and natural systems.
Kinetic Energy
Kinetic energy is the energy possessed by an object due to its motion.
The formula for kinetic energy is:
[
KE = \frac{1}{2}mv^2
]
Where:
(m) = mass
(v) = velocity
The faster an object moves, the greater its kinetic energy.
Examples of kinetic energy include:
- A moving car
- Flowing water
- Wind moving through the atmosphere
- A rolling ball
Potential Energy
Potential energy is the energy stored in an object because of its position or configuration.
Gravitational Potential Energy
This type of energy occurs due to the height of an object above the ground.
[
PE = mgh
]
Where:
(m) = mass
(g) = acceleration due to gravity
(h) = height
Examples include:
- Water stored in a dam
- A book placed on a shelf
- A roller coaster at the top of a hill
Elastic Potential Energy
Elastic potential energy is stored when objects such as springs or rubber bands are stretched or compressed.
Examples include:
- A stretched bow
- A compressed spring
- A stretched rubber band
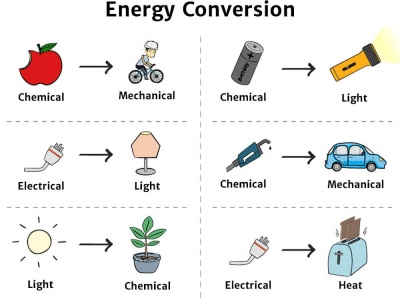
Energy Transformations




Energy transformations occur when energy changes from one form to another.
Examples include:
Chemical energy → mechanical energy in muscles
Electrical energy → light energy in a bulb
Mechanical energy → electrical energy in generators
Solar energy → chemical energy in plants
Despite these transformations, the total energy remains constant.
Conservation of Mechanical Energy
One of the most important applications of the conservation of energy is in mechanical systems.
When no external forces such as friction act on a system, the total mechanical energy remains constant.
[
KE + PE = constant
]
This means:
[
KE_1 + PE_1 = KE_2 + PE_2
]
Example: Falling Object



Consider a ball dropped from a height.
At the top:
Potential energy = maximum
Kinetic energy = zero
As the ball falls:
Potential energy decreases
Kinetic energy increases
Just before hitting the ground:
Potential energy = minimum
Kinetic energy = maximum
The total energy remains constant.
Conservation of Energy in Everyday Life
The principle of energy conservation can be observed in many everyday situations.
Roller Coasters
At the top of the track, potential energy is maximum. As the roller coaster descends, potential energy converts to kinetic energy.
Hydroelectric Power Plants
Water stored in dams has gravitational potential energy. As it flows downward, this energy converts to mechanical energy, which turbines convert to electrical energy.
Electric Bulbs
Electrical energy converts into light and heat.
Car Engines
Chemical energy in fuel converts into mechanical energy that moves the vehicle.
Energy Loss and Dissipation
In real-world systems, mechanical energy may appear to decrease because some energy converts into heat, sound, or other forms.
For example:
Friction converts mechanical energy into heat.
Although mechanical energy decreases, the total energy of the system remains constant.
This process is called energy dissipation.
Conservation of Energy in Different Fields
The conservation of energy principle applies across many scientific disciplines.
Thermodynamics
In thermodynamics, the conservation of energy appears as the first law of thermodynamics.
This law states that the change in internal energy of a system equals heat added minus work done.
Electromagnetism
Electrical circuits convert electrical energy into light, heat, or mechanical motion while conserving total energy.
Nuclear Physics
Nuclear reactions convert mass into energy according to Einstein’s equation:
[
E = mc^2
]
Even in nuclear processes, total energy remains conserved.
Astrophysics
Planetary motion and star formation obey energy conservation principles.
Gravitational potential energy converts into kinetic energy during the formation of stars.
Importance of Conservation of Energy
The conservation of energy principle is one of the most powerful tools in physics.
It allows scientists to:
Predict system behavior
Analyze complex mechanical systems
Design efficient machines
Understand natural processes
Study energy transfer in ecosystems
Without this principle, many physical calculations would be impossible.
Advantages of Using Energy Conservation
Using energy conservation simplifies many physics problems because it avoids complicated force calculations.
Instead of analyzing forces and accelerations, physicists can compare energy values at different points in a system.
This approach is widely used in mechanics, engineering, and physics research.
Limitations and Practical Considerations
Although energy is always conserved, some systems appear to lose energy because it converts into forms such as heat or sound that are difficult to measure.
For example:
Friction in machines
Air resistance in moving objects
Heat loss in engines
Engineers work to minimize these losses to improve efficiency.
Summary
The law of conservation of energy is a fundamental principle stating that energy cannot be created or destroyed but only transformed from one form to another. This law applies to all physical systems and ensures that the total energy of an isolated system remains constant.
Energy exists in many forms, including kinetic energy, potential energy, thermal energy, chemical energy, and electrical energy. During physical processes, energy continuously changes from one form to another, but the total energy remains conserved.
This principle plays a crucial role in understanding mechanical systems, thermodynamics, electricity, nuclear reactions, and astrophysical processes. By applying the conservation of energy, scientists and engineers can analyze complex systems and design technologies that use energy efficiently.
The conservation of energy remains one of the most powerful and universal laws in physics, helping explain the behavior of the natural world.
