



1. Introduction to Acids and Bases
Acids and bases are fundamental concepts in chemistry and play a crucial role in chemical reactions, biological systems, industrial processes, and environmental chemistry. The study of acids and bases is known as acid–base chemistry, which forms a core part of physical chemistry and analytical chemistry.
Acids and bases were recognized long before modern chemistry developed. Early chemists classified substances based on their observable properties such as taste, reactivity, and effects on indicators. For example:
- Acids typically have a sour taste
- Bases usually have a bitter taste and slippery feel
However, modern chemistry defines acids and bases in terms of their behavior in chemical reactions and their ability to donate or accept protons or electrons.
Acid–base reactions are extremely common and are involved in many processes including:
- Digestion in the human body
- Industrial chemical production
- Soil chemistry and agriculture
- Water treatment
- Pharmaceutical synthesis
- Environmental processes such as acid rain
Understanding acids and bases helps scientists predict chemical behavior, control reactions, and maintain balance in biological and environmental systems.
2. Historical Development of Acid–Base Concepts
The understanding of acids and bases evolved over time. Several scientists proposed theories explaining their behavior.
The most important acid–base theories include:
- Arrhenius theory
- Brønsted–Lowry theory
- Lewis theory
Each theory expanded the concept of acids and bases.
3. Arrhenius Theory of Acids and Bases
The Arrhenius theory was proposed by the Swedish chemist Svante Arrhenius in 1884.
According to Arrhenius:
Acids are substances that produce hydrogen ions (H⁺) in aqueous solution.
Examples of Arrhenius acids:
- Hydrochloric acid (HCl)
- Sulfuric acid (H₂SO₄)
- Nitric acid (HNO₃)
Example reaction:
HCl → H⁺ + Cl⁻
Bases are substances that produce hydroxide ions (OH⁻) in aqueous solution.
Examples:
- Sodium hydroxide (NaOH)
- Potassium hydroxide (KOH)
- Calcium hydroxide (Ca(OH)₂)
Example:
NaOH → Na⁺ + OH⁻
Limitations of Arrhenius Theory
Although useful, Arrhenius theory has several limitations:
- It applies only to aqueous solutions.
- It cannot explain reactions occurring without water.
- It does not include substances like ammonia as bases.
Because of these limitations, more general theories were developed.
4. Brønsted–Lowry Theory
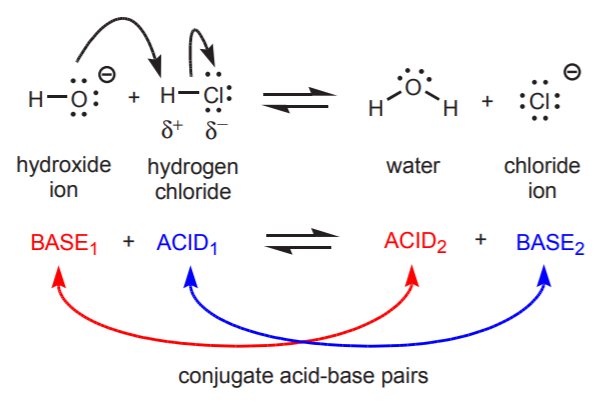



The Brønsted–Lowry theory was proposed in 1923 by Johannes Brønsted and Thomas Lowry.
According to this theory:
An acid is a proton donor.
A base is a proton acceptor.
Example reaction:
HCl + H₂O → H₃O⁺ + Cl⁻
Here:
- HCl donates a proton → acid
- H₂O accepts a proton → base
Conjugate Acid–Base Pairs
In Brønsted–Lowry reactions, acids and bases exist as conjugate pairs.
Example:
NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
Pairs:
NH₃ / NH₄⁺
H₂O / OH⁻
Each acid has a conjugate base, and each base has a conjugate acid.
5. Lewis Theory of Acids and Bases
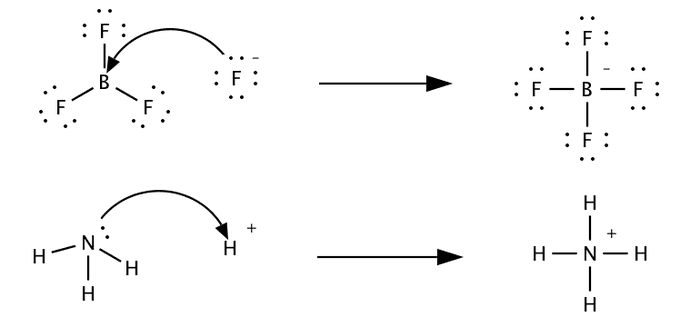

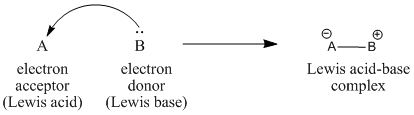
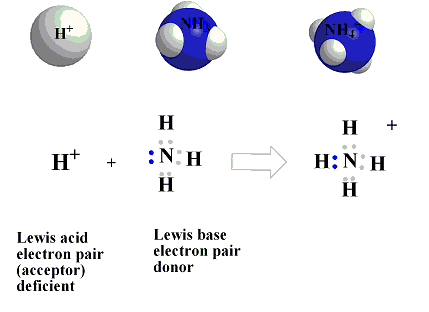
The Lewis theory was proposed by Gilbert N. Lewis.
According to Lewis:
A Lewis acid is an electron pair acceptor.
A Lewis base is an electron pair donor.
Example reaction:
BF₃ + NH₃ → F₃B–NH₃
Here:
- BF₃ accepts electron pair → Lewis acid
- NH₃ donates electron pair → Lewis base
Importance of Lewis Theory
Lewis theory explains reactions that cannot be described by proton transfer, such as:
- Metal complex formation
- Catalytic reactions
- Organic reactions
6. Properties of Acids
Acids exhibit several characteristic properties.
1. Sour Taste
Examples include citric acid in lemons and acetic acid in vinegar.
2. Turn Blue Litmus Red
Acids change the color of litmus indicator.
3. React with Metals
Acids react with metals to produce hydrogen gas.
Example:
Zn + 2HCl → ZnCl₂ + H₂
4. Conduct Electricity
Acids form ions in solution and conduct electricity.
5. React with Bases
Acids react with bases to form salt and water.
7. Properties of Bases
Bases also have distinctive properties.
1. Bitter Taste
2. Slippery Texture
Soap and detergents feel slippery.
3. Turn Red Litmus Blue
4. Conduct Electricity in Solution
5. Neutralize Acids
Bases react with acids to produce salt and water.
8. Acid–Base Neutralization




Neutralization is a reaction between an acid and a base.
General reaction:
Acid + Base → Salt + Water
Example:
HCl + NaOH → NaCl + H₂O
Neutralization reactions are widely used in:
- Medicine
- Agriculture
- Water treatment
- Industrial chemistry
9. Strength of Acids and Bases
Acids and bases are classified as strong or weak based on their ionization in water.
Strong Acids
Strong acids ionize completely in water.
Examples:
- HCl
- HNO₃
- H₂SO₄
Weak Acids
Weak acids ionize partially.
Examples:
- Acetic acid
- Carbonic acid
- Formic acid
Strong Bases
Examples:
- NaOH
- KOH
- Ca(OH)₂
Weak Bases
Examples:
- Ammonia
- Amines
10. pH Scale




The pH scale measures acidity or basicity.
Range:
0 – 14
pH < 7 → acidic
pH = 7 → neutral
pH > 7 → basic
Examples:
- Lemon juice pH ≈ 2
- Pure water pH = 7
- Soap pH ≈ 9
11. Acid–Base Indicators
Indicators are substances that change color depending on pH.
Common indicators include:
- Litmus
- Phenolphthalein
- Methyl orange
- Universal indicator
Indicators help detect acidity or alkalinity in chemical reactions.
12. Buffer Solutions
Buffers resist changes in pH when acids or bases are added.
They consist of:
- Weak acid + conjugate base
or - Weak base + conjugate acid
Example:
Acetic acid + sodium acetate.
Buffers maintain pH stability in biological systems.
13. Acid–Base Titration
Titration is an analytical technique used to determine the concentration of acids or bases.
Equipment used:
- Burette
- Pipette
- Indicator
During titration, acid and base react until the equivalence point is reached.
14. Acid–Base Reactions in Biology
Acid–base balance is crucial in biological systems.
Examples include:
- Blood pH regulation
- Enzyme activity
- Cellular metabolism
The human body maintains blood pH around 7.4 using buffer systems.
15. Environmental Importance
Acid–base chemistry influences many environmental processes.
Examples include:
- Acid rain formation
- Ocean acidity
- Soil chemistry
- Water purification
Understanding acid–base reactions helps scientists address environmental challenges.
16. Industrial Applications
Acids and bases are widely used in industry.
Examples:
- Sulfuric acid production
- Fertilizer manufacturing
- Petroleum refining
- Pharmaceutical synthesis
- Food processing
17. Importance of Acid–Base Chemistry
Acid–base chemistry is essential for understanding:
- Chemical reactions
- Biological processes
- Environmental systems
- Industrial chemistry
It provides a framework for studying chemical behavior and predicting reaction outcomes.
Conclusion
Acids and bases are fundamental chemical substances that play a vital role in chemistry and everyday life. Their behavior has been explained through several theories including Arrhenius, Brønsted–Lowry, and Lewis theories. Acid–base reactions such as neutralization, titration, and buffer systems are essential in laboratory analysis, industrial processes, and biological systems. The pH scale provides a quantitative measure of acidity and basicity, allowing scientists to monitor and control chemical environments. Understanding acids and bases is therefore crucial for advancing chemistry, medicine, environmental science, and technology.
