



Introduction
Nuclear structure refers to the internal arrangement and properties of particles within the atomic nucleus. The nucleus is the dense central region of an atom that contains most of the atom’s mass and is composed of protons and neutrons, collectively known as nucleons. Understanding nuclear structure is fundamental to nuclear physics because it explains the stability, behavior, and interactions of atomic nuclei.
The nucleus is extremely small compared to the size of the atom, yet it contains nearly all of the atom’s mass. Nuclear structure determines many important physical phenomena such as nuclear reactions, radioactive decay, nuclear energy production, and the formation of elements in stars.
The development of nuclear structure theory involved major contributions from scientists such as Ernest Rutherford, James Chadwick, Maria Goeppert Mayer, and Hans Jensen.
Nuclear structure is studied using theoretical models and experimental techniques that investigate how nucleons interact and arrange themselves inside the nucleus.
Discovery of the Atomic Nucleus
The concept of the nucleus was established through the famous gold foil experiment conducted by Ernest Rutherford in 1911.
In this experiment:
- Alpha particles were directed toward a thin gold foil.
- Most particles passed through the foil.
- Some particles were deflected at large angles.
These observations led to the conclusion that atoms contain a small, dense, positively charged nucleus.
Later, in 1932, James Chadwick discovered the neutron, completing the picture of nuclear composition.
Composition of the Nucleus
The atomic nucleus consists of two types of particles:
Protons
Protons are positively charged particles located in the nucleus.
Properties:
- Charge: (+1.602 \times 10^{-19}) C
- Mass: (1.67 \times 10^{-27}) kg
The number of protons determines the atomic number and the identity of the element.
Neutrons
Neutrons are electrically neutral particles also found in the nucleus.
Properties:
- No electric charge
- Mass slightly greater than proton
Neutrons play an important role in stabilizing the nucleus.
Nuclear Size
The nucleus is extremely small compared to the atom.
Typical nuclear radius is given by:
[
R = R_0 A^{1/3}
]
where
- (R_0 \approx 1.2 \times 10^{-15} m)
- (A) = mass number
This relationship shows that nuclear size increases with the cube root of the mass number.
Nuclear Density
One remarkable property of nuclei is their constant density.
Because the radius depends on (A^{1/3}), the density remains approximately constant for all nuclei.
Typical nuclear density:
[
\rho \approx 2.3 \times 10^{17} , kg/m^3
]
This density is extremely high compared to ordinary matter.
Nuclear Forces
The stability of the nucleus is due to the strong nuclear force.
This force binds protons and neutrons together.
Characteristics of the nuclear force:
- Very strong
- Short range
- Attractive between nucleons
- Independent of electric charge
The strong force overcomes the electrostatic repulsion between protons.
Binding Energy
Binding energy is the energy required to separate a nucleus into its individual nucleons.
It represents the strength of the nuclear force holding the nucleus together.
The binding energy is related to mass through Einstein’s equation:
[
E = mc^2
]
The difference between the mass of individual nucleons and the mass of the nucleus is called the mass defect.
Binding Energy Curve


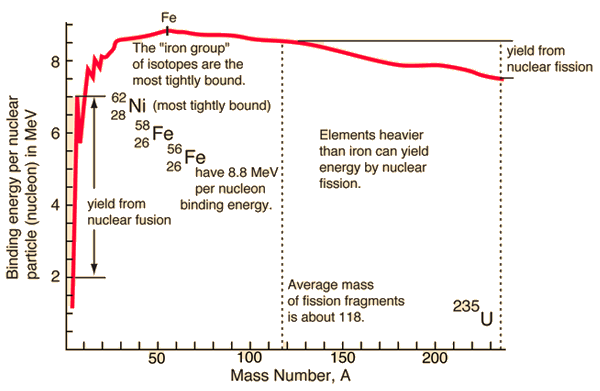
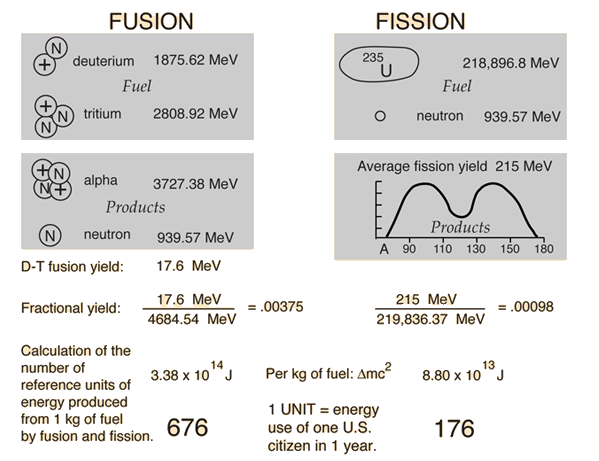
The binding energy per nucleon varies with mass number.
Key features:
- Increases for light nuclei
- Peaks near iron
- Decreases for heavy nuclei
This explains why:
- Light nuclei undergo fusion
- Heavy nuclei undergo fission
Both processes release energy.
Nuclear Models
Several models have been developed to describe nuclear structure.
Major models include:
- Liquid drop model
- Shell model
- Collective model
Each model explains different aspects of nuclear behavior.
Liquid Drop Model



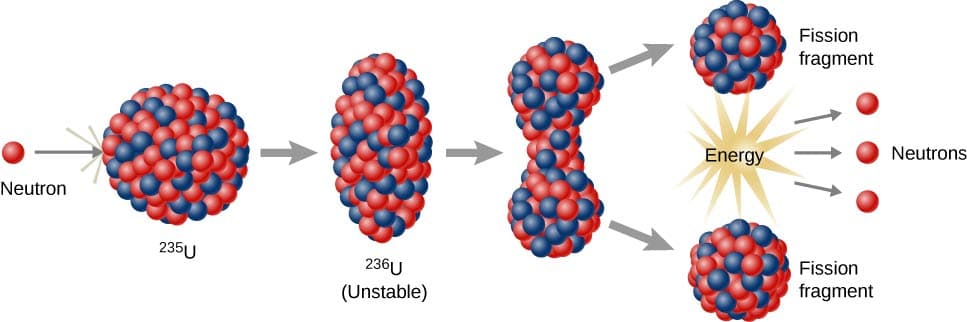
The liquid drop model treats the nucleus as a drop of incompressible nuclear fluid.
In this model:
- Nucleons interact with neighboring particles
- Nuclear forces resemble surface tension in liquids
This model explains:
- Nuclear binding energy
- Nuclear fission
- Nuclear stability
Semi-Empirical Mass Formula
The liquid drop model leads to the semi-empirical mass formula, also called the Weizsäcker formula.
It describes nuclear binding energy using several terms:
- Volume energy
- Surface energy
- Coulomb energy
- Asymmetry energy
- Pairing energy
These terms account for different nuclear effects.
Nuclear Shell Model

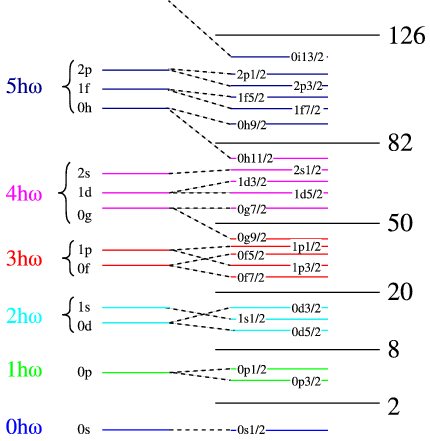

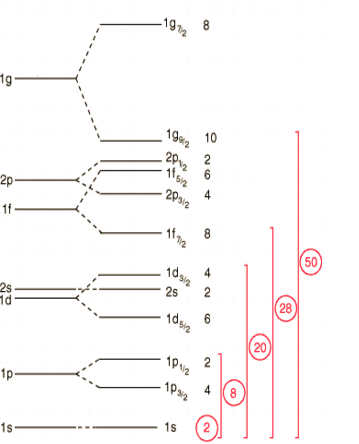
The nuclear shell model describes nucleons moving in quantized energy levels within the nucleus.
This model was developed by Maria Goeppert Mayer and Hans Jensen.
Magic Numbers
The shell model predicts certain numbers of nucleons that produce especially stable nuclei.
These are called magic numbers:
2, 8, 20, 28, 50, 82, 126
Nuclei with these numbers of protons or neutrons are more stable.
Collective Model
The collective model combines features of the liquid drop and shell models.
It describes the nucleus as a system where nucleons move individually but also participate in collective motion such as:
- Vibrations
- Rotations
This model explains nuclear excitation spectra.
Nuclear Spin and Magnetic Moment
Nuclei possess angular momentum known as nuclear spin.
Nuclear spin arises from the combined spins and orbital motion of nucleons.
The nuclear magnetic moment is associated with this spin and interacts with magnetic fields.
These properties are important in techniques such as nuclear magnetic resonance (NMR).
Nuclear Stability
Nuclear stability depends on the balance between:
- Nuclear forces
- Electrostatic repulsion
The neutron-to-proton ratio plays a key role.
Light nuclei are stable when:
[
N \approx Z
]
Heavy nuclei require more neutrons to remain stable.
Radioactivity
Unstable nuclei undergo radioactive decay.
Common types of decay include:
Alpha Decay
Emission of helium nucleus.
Beta Decay
Transformation of neutron to proton or vice versa.
Gamma Decay
Emission of high-energy photons.
Radioactivity changes the nuclear structure.
Nuclear Reactions
Nuclear reactions involve changes in nuclear structure.
Two major types include:
Nuclear Fusion
Light nuclei combine to form heavier nuclei.
Occurs in stars.
Nuclear Fission
Heavy nuclei split into smaller nuclei.
Used in nuclear reactors.
Both processes release enormous energy.
Experimental Methods for Studying Nuclear Structure
Scientists study nuclear structure using several experimental techniques:
- Particle accelerators
- Scattering experiments
- Nuclear spectroscopy
- Radioactive decay measurements
These methods reveal information about nuclear energy levels and interactions.
Applications of Nuclear Structure
Understanding nuclear structure has many practical applications.
Nuclear Energy
Used in power plants and reactors.
Medical Imaging
Techniques such as PET scans use nuclear processes.
Radiation Therapy
Radioactive isotopes treat cancer.
Astrophysics
Nuclear reactions explain stellar evolution.
Nuclear Structure in Astrophysics
Nuclear processes play a crucial role in the universe.
Stars generate energy through nuclear fusion reactions.
Heavier elements are formed through processes such as:
- Stellar nucleosynthesis
- Supernova explosions
These processes depend on nuclear structure.
Modern Research in Nuclear Physics
Modern nuclear physics explores topics such as:
- Exotic nuclei
- Neutron-rich nuclei
- Nuclear matter in neutron stars
- Quark structure of nucleons
Advanced facilities use particle accelerators and detectors to study these phenomena.
Conclusion
Nuclear structure is a fundamental concept in nuclear physics that describes the arrangement and interactions of protons and neutrons within the atomic nucleus. The nucleus is a tiny but extremely dense region that contains most of the atom’s mass and is held together by powerful nuclear forces.
Various theoretical models such as the liquid drop model, shell model, and collective model have been developed to explain nuclear behavior. These models help scientists understand nuclear stability, binding energy, and the processes involved in nuclear reactions.
The study of nuclear structure has led to important discoveries in physics and has numerous practical applications, including nuclear energy production, medical technology, and astrophysics. As research continues, scientists aim to gain deeper insights into the fundamental properties of nuclear matter and the forces that govern the atomic nucleus.
